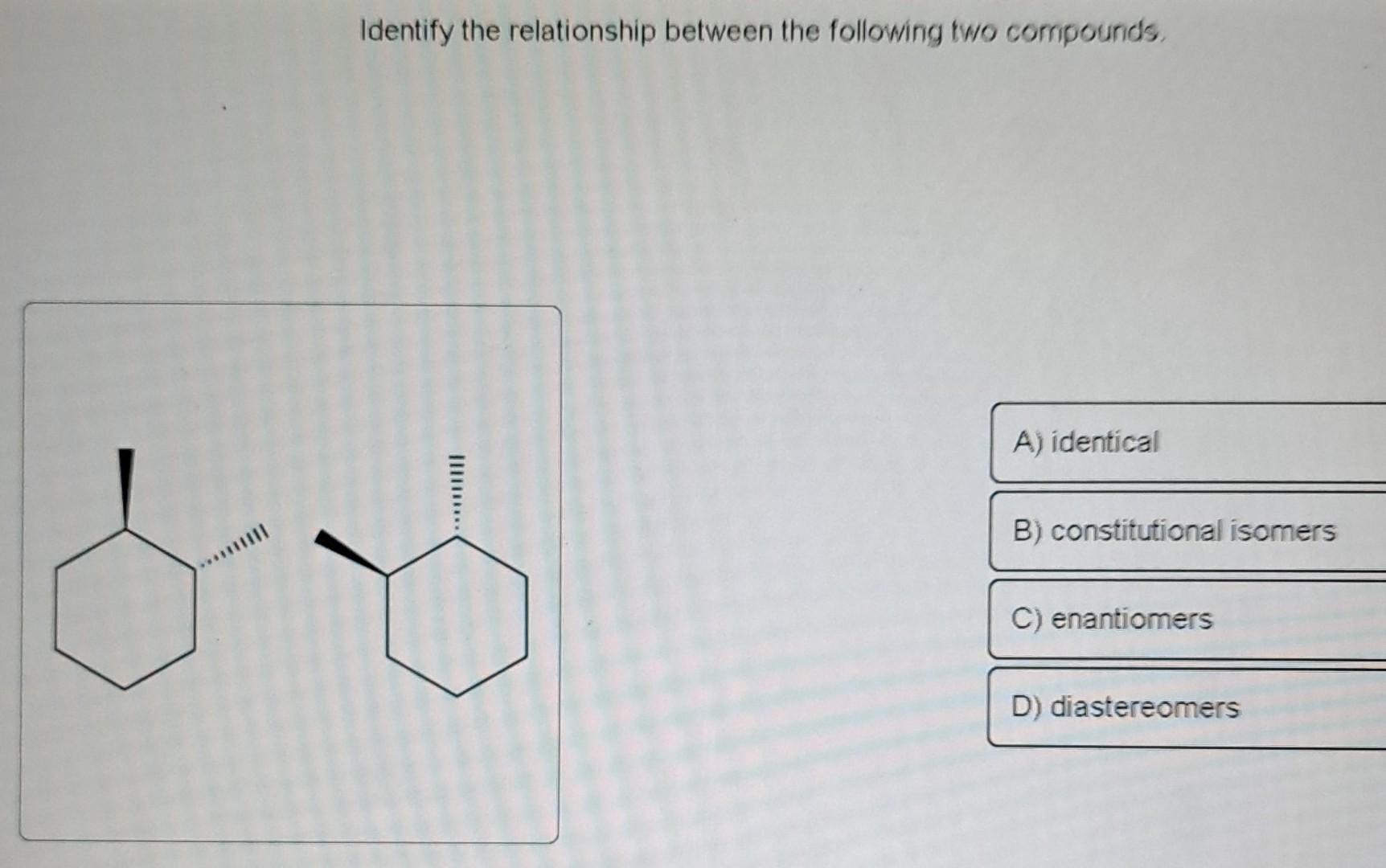
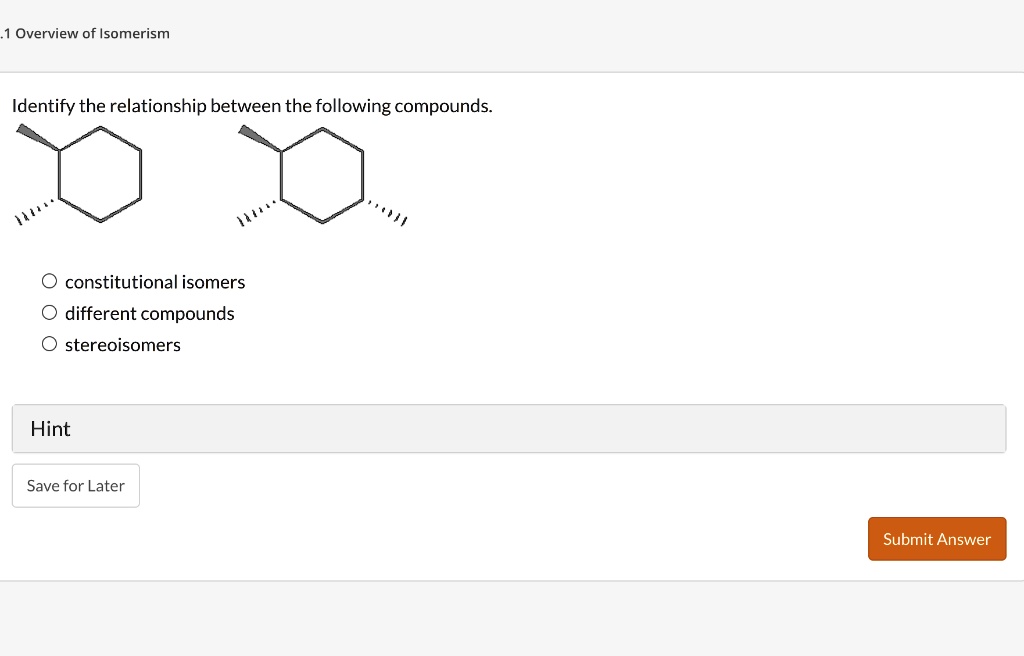
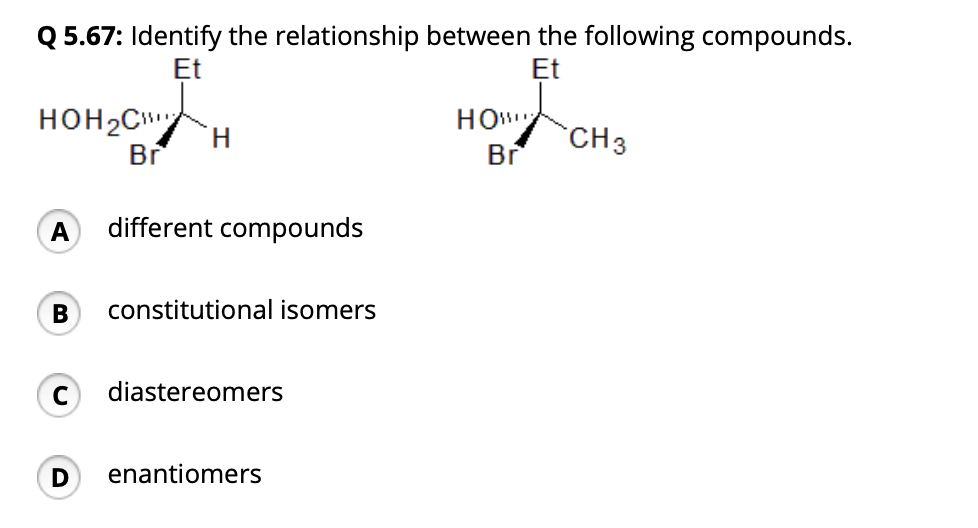
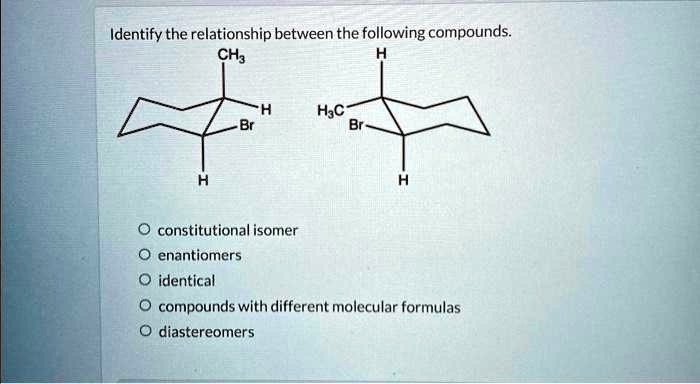
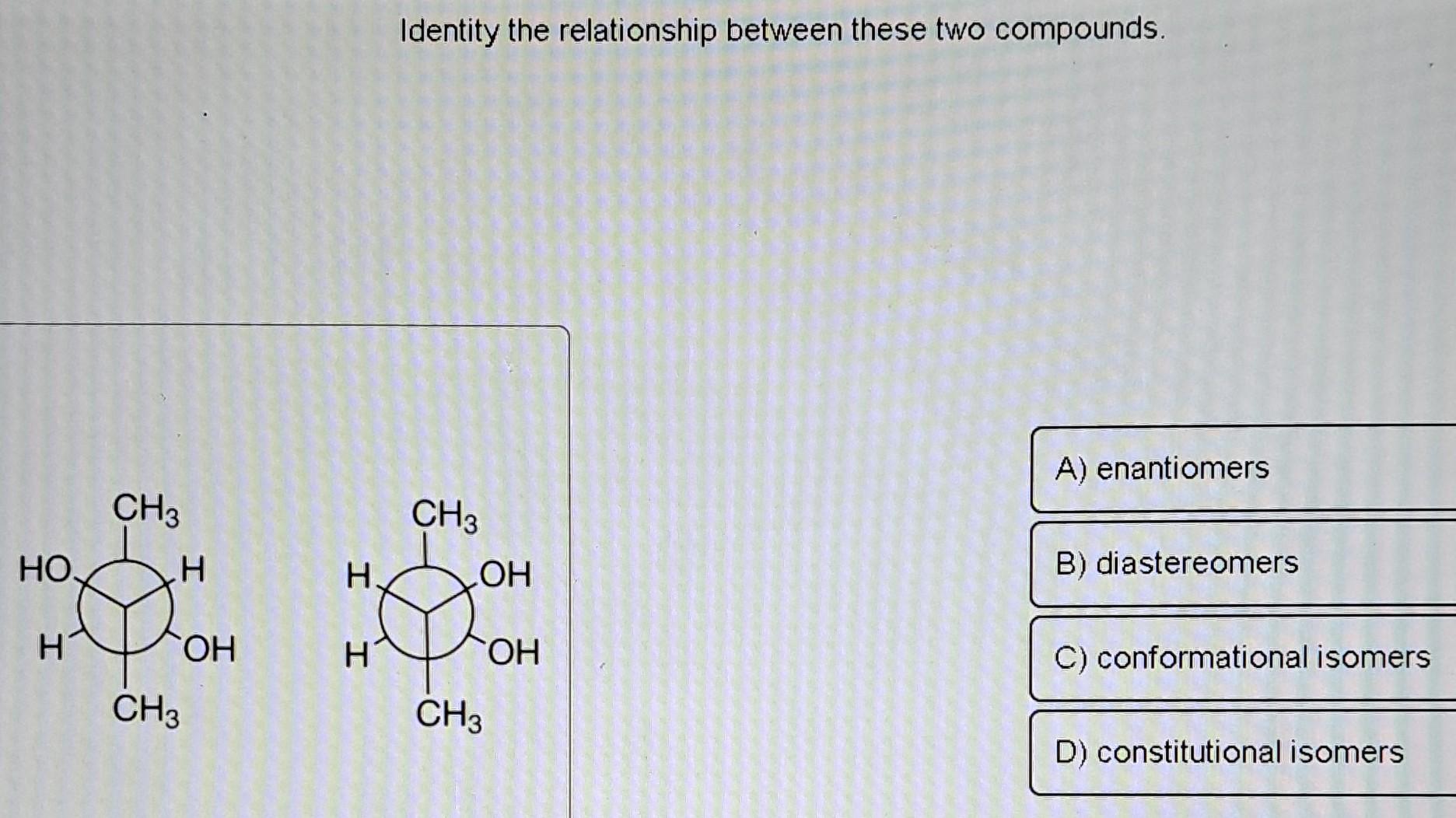
Identify The Relationship Between The Following Compounds
Oh, hello there, fellow curious minds! Get ready to dive into the utterly fascinating, sometimes mind-boggling, but always wonderful world of tiny things we call compounds. Think of them like ingredients in a cosmic recipe, each with its own special job and personality. We’re going to be playing a super fun game today: spotting the relationship between these amazing little building blocks of everything.
Imagine you’re at a party, and everyone's got a story. Some people are instant best friends, others are distant cousins, and some just share a common interest. That’s kind of what we’re doing here, but with molecules instead of people! We're looking for those secret connections, those shared histories, those “aha!” moments that make you go, “Of course! They’re practically twins!”
Our first duo, or maybe trio, or even a whole little squad, are the dynamic duo known as water and hydrogen peroxide. Now, these two might look a bit alike at first glance. They both have those zappy little hydrogen atoms, right? But oh boy, do they have different personalities!
Water: The Life of the Party
Water (H2O) is our ultimate chill friend. It’s the life of the party, the soothing balm, the universal solvent. Everything loves water! Plants drink it, we drink it, and it helps all sorts of chemical reactions happen without getting too worked up. It’s the ultimate go-with-the-flow compound.
Think of water like your favorite cozy sweater. It’s reliable, comfortable, and essential for survival. Without it, life as we know it would be… well, pretty darn dry! It’s the foundation of so many amazing things.
Hydrogen Peroxide: The Energetic Upstart
Then there’s hydrogen peroxide (H2O2). This one is like the energetic younger sibling who’s always buzzing with ideas. It’s got an extra oxygen atom hanging around, and that extra bit makes it a little more… reactive. It’s not as laid-back as water.
This extra oxygen makes hydrogen peroxide a bit of a superhero in disguise. It’s great for cleaning, for a little antiseptic boost, and it can even be used in some cool scientific experiments. It’s like water’s slightly more dramatic cousin, always ready for action!
The Family Connection
So, what’s their relationship? They are, dare I say, cousins in the chemical family tree! They both come from the same basic ingredients: hydrogen and oxygen. It’s like having two siblings who share parents but have wildly different interests. One loves to relax by the pool, and the other is off trying to climb Mount Everest!
The key difference, that extra oxygen, is what gives hydrogen peroxide its special powers. It’s like a tiny, almost imperceptible change that leads to a whole new set of behaviors. It's the difference between a gentle breeze and a playful gust of wind!
This is a prime example of how small tweaks in a compound's structure can lead to drastically different properties. It’s like adding a pinch of spice to a plain dish – suddenly, everything is more exciting! This relationship is all about shared ancestry and distinct destinies.
Let's Talk About Acids and Bases: The Ultimate Frenemies
Now, let’s switch gears and talk about another classic pairing: acids and bases. These two are like the ultimate frenemies of the chemical world. They can’t stand each other, but they also can’t exist without each other. It’s a relationship built on… well, a whole lot of fizzing and bubbling!
Think of acids like the grumpy one who’s always complaining. They tend to taste sour (please, don’t taste random chemicals!), and they can be pretty corrosive. They're the ones that can sting your tongue if you eat a lemon, for example. They’re all about giving away hydrogen ions, which is like them saying, “Here, take this!”
On the other hand, bases are the ones who are a bit more… slippery. They tend to taste bitter (again, don’t taste them!) and feel soapy. Bases are the ones who are happy to take those hydrogen ions. They’re like the accommodating friend who’s always saying, “Sure, I’ll hold that for you!”
The Big Reaction
When an acid and a base meet, it’s fireworks! They have a dramatic reaction where they neutralize each other. It’s like a heated debate that ends with a surprising agreement. The grumpy acid and the accommodating base come together to form something new and usually quite calm: salt and water.
This is one of the most fundamental relationships in chemistry. It’s all about the transfer of these little hydrogen guys. The acid gives, the base takes, and poof! You get a neutral compound, like your everyday table salt. It’s a beautifully balanced dance.
This dance of acids and bases is happening all around us, all the time. From your stomach digesting food to cleaning products working their magic, this relationship is essential for countless processes. It's the ultimate "opposites attract" scenario, leading to stability and everyday utility.
Simple Sugars: The Sweet Family
Let’s get sweet now! We're talking about simple sugars. These are like the energetic, happy-go-lucky members of the carbohydrate family. Think of them as the building blocks for longer, more complex sugary structures.
The most famous simple sugar is probably glucose. This is the sugar your body absolutely loves for energy. It’s like the fuel that powers your amazing brain and your incredible body. It's the immediate go-to for energy.
Another close relative is fructose. You find this one naturally in fruits, which is why fruits taste so wonderfully sweet! It’s like glucose’s slightly sweeter, fruitier sibling. They have similar structures but slightly different ways of interacting.
The Structural Link
The relationship between glucose and fructose is one of structural similarity with a twist. They are both monosaccharides, meaning they are single sugar units. They are both built from the same basic number of carbon, hydrogen, and oxygen atoms, but arranged just a little bit differently.
This slight rearrangement is like changing a few letters in a word – it can change the meaning or function. It’s the difference between saying “sweet” and “sweat”! For glucose and fructose, it means they are processed slightly differently by our bodies, even though they both contribute to sweetness and energy.
This family of simple sugars is crucial for life. They are the quick energy sources that keep everything running smoothly. Their relationship is one of close kinship, built on a shared foundation of atoms arranged in slightly different, but equally important, ways. They’re the foundational elements of sweetness and energy.
The Mighty Carbon Chains: The Backbone of Life
Finally, let’s marvel at the superstars of organic chemistry: compounds built around carbon. Carbon atoms are like the ultimate Lego bricks of life. They can connect to each other in long chains, rings, and branching structures, forming the basis of almost all living things.
Think of methane (CH4) as the simplest, smallest carbon compound. It's just one carbon atom with four hydrogens attached. It's like the single Lego brick, the absolute beginning of the carbon chain party. It’s the foundational concept.
Then you have things like ethane (C2H6), which is two carbon atoms linked together, each with a few hydrogens. This is like snapping two Lego bricks together. It’s the first step in building something bigger and more complex.
The Chain Reaction
The relationship here is all about chain length and complexity. Methane, ethane, propane, butane – these are all similar compounds called alkanes. They differ primarily in how many carbon atoms are linked end-to-end. It’s like graduating from a single brick to a small tower, then a bigger structure.
As the carbon chains get longer, the properties of the compounds change. They can become more solid, less volatile, and have different uses. This is the power of building with carbon – you can create an almost infinite variety of structures, each with its own unique characteristics. It's the foundation for the vast diversity of organic molecules.
This relationship, where increasing chain length leads to changing properties, is fundamental to understanding everything from fuels to plastics to the very molecules that make up our bodies. It's a powerful reminder that even small additions can lead to grand transformations. The potential for variation is truly astonishing.
Isn’t chemistry just the coolest? It’s like a giant puzzle where every piece fits together in the most ingenious ways. Keep your eyes peeled, and you’ll start spotting these relationships everywhere! Happy discovering!
