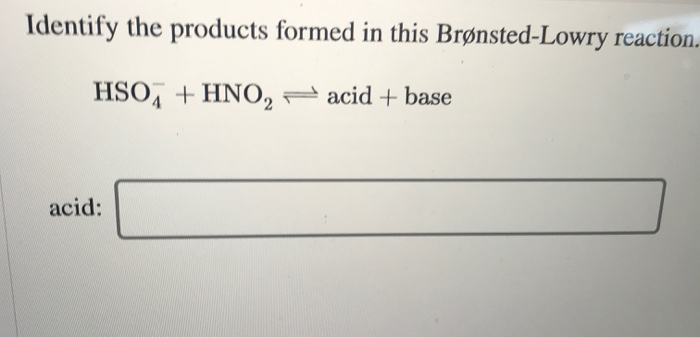
Identify The Products Formed In This Brønsted-lowry Reaction.
Ever wondered what happens when things mix and mingle in the world of chemistry? It’s not all bubbling beakers and dramatic explosions (though that can be fun too!). Sometimes, it’s about understanding the quieter, yet incredibly important, dance of molecules. Today, we’re diving into a concept called Brønsted-Lowry reactions and, more specifically, how we can figure out what’s formed during these interactions. It might sound a little technical, but trust us, it’s a fascinating peek into how our world works, from the water we drink to the way our bodies function.
So, why should you care about identifying products in a Brønsted-Lowry reaction? Think of it like being a detective, but instead of solving crimes, you’re solving chemical puzzles. Brønsted-Lowry theory helps us understand how acids and bases interact. In its simplest form, an acid is a proton donor (a hydrogen ion, H⁺), and a base is a proton acceptor. When they react, the acid gives away its proton to the base. Identifying the products means we’re figuring out what new substances are created after this proton exchange has happened. This is super useful because understanding these reactions helps us predict the outcome of mixing different chemicals, which is fundamental to countless scientific and industrial processes.
The benefits of grasping this are quite broad. In education, it’s a cornerstone of understanding acid-base chemistry, which is crucial for students in science, medicine, and engineering. Beyond the classroom, it has real-world applications. For instance, understanding how acids and bases react is vital in:
- Water treatment: Adjusting pH levels to make water safe and palatable.
- Food production: From the tang in your yogurt to the leavening in your bread, acid-base chemistry is at play.
- Medicine: Many drugs are designed to interact with biological systems based on their acidic or basic properties.
- Environmental science: Studying phenomena like acid rain and its impact.
Looking at a specific example, imagine hydrochloric acid (HCl) reacting with sodium hydroxide (NaOH). HCl is a strong acid, meaning it readily donates a proton. NaOH is a strong base, readily accepting one. When they meet, the HCl donates its H⁺ to the NaOH. The products formed would be water (H₂O) – the hydrogen from the acid and the hydroxide (OH⁻) from the base combine – and sodium chloride (NaCl), or table salt, which is the remaining ion from the base and the ion left after the acid donated its proton. So, the reaction is: HCl + NaOH → H₂O + NaCl. We’ve identified our products!
Ready to explore this a bit yourself? It’s simpler than you might think! You can start by looking up common household acids and bases. Think about vinegar (acetic acid) and baking soda (sodium bicarbonate). When you mix them, you see fizzing – that’s a reaction! While it’s a bit more complex than a simple Brønsted-Lowry proton transfer, the underlying principle of chemical interaction is there. For a more direct exploration, find simple acid-base reaction diagrams online. Pay attention to the arrows which show the movement of the proton (H⁺) from the acid to the base. Observing these simple exchanges can make the abstract concept of product formation much more tangible and, dare we say, fun!
