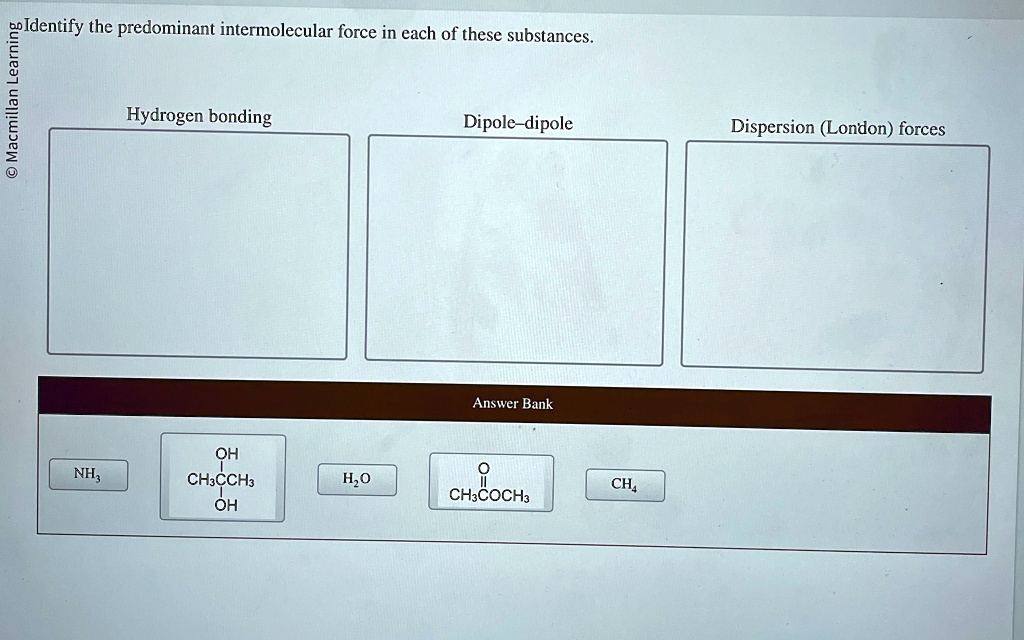
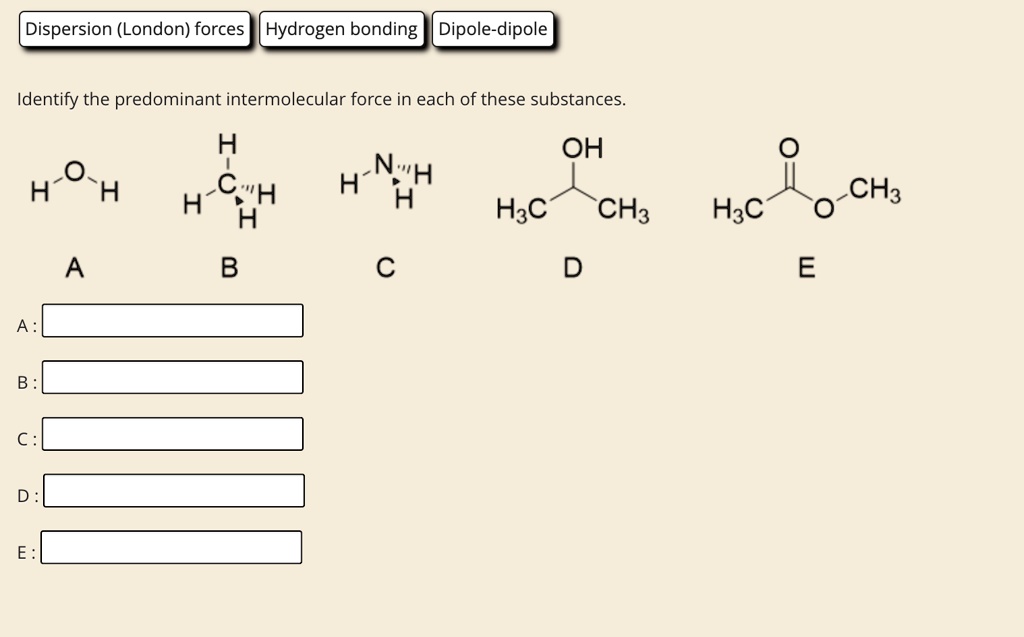
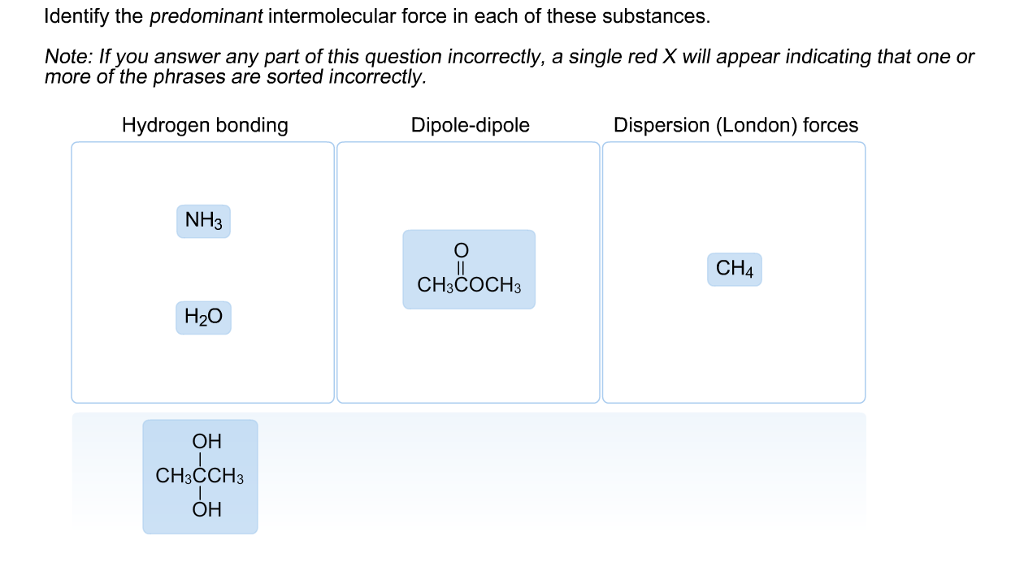
Identify The Predominant Intermolecular Forces In Each Of These Substances
You know, I was at a friend's barbecue last weekend, and it was one of those super casual, laid-back affairs. We were all lounging around, the grill was sizzling, and someone pulled out a bottle of water and a can of soda. My friend, bless his heart, was meticulously arranging the ice cubes in a cooler, making sure they were perfectly aligned. I, on the other hand, just chucked them in, not really giving it a second thought. It got me thinking, though, about why some things behave so differently, even when they seem like they should be on the same playing field. Like water and soda, right? They’re both liquids, both refreshing, but there's a whole world of difference in how they feel, how they interact. And it all boils down to… wait for it… intermolecular forces. Yep, sounds fancy, but stick with me, it’s actually pretty cool!
I know, I know, the term "intermolecular forces" probably conjures up images of dusty textbooks and tedious lectures. But honestly, it's like the secret handshake of molecules. It's what makes them stick together, or not stick together, and how they interact with their surroundings. Think of it as the invisible glue that holds the universe together, or sometimes, the little nudge that sends things flying apart. We're going to dive into some common substances and try to figure out what makes them tick, or rather, what makes their molecules attract.
So, let's get down to business. We've got a few substances to dissect, and our mission, should we choose to accept it (and we totally should, it's fun!), is to identify the predominant intermolecular forces at play in each. Ready to put on our molecular detective hats?
Substance 1: Water (H2O)
Ah, water. The elixir of life! It’s in our coffee, our tea, our blood, and pretty much everywhere. What makes water so… watery? And why does it do all those weird and wonderful things, like form droplets and climb up tiny tubes (capillary action, anyone?)?
First off, let's look at the molecule itself. Water is H2O. That means one oxygen atom and two hydrogen atoms. Now, oxygen is a bit of a hog when it comes to electrons. It’s much more electronegative than hydrogen. This means the electrons in the covalent bonds between oxygen and hydrogen are pulled closer to the oxygen atom. What does this create? A polar molecule. The oxygen end has a partial negative charge (δ-), and the hydrogen ends have partial positive charges (δ+).
And here's where the magic happens. Because water molecules are polar, the slightly positive hydrogen of one water molecule is attracted to the slightly negative oxygen of another water molecule. This specific, super-strong type of attraction between a hydrogen atom bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) and another electronegative atom is called hydrogen bonding. It’s like a really strong, directed hug between molecules.
This hydrogen bonding is the predominant intermolecular force in water. It’s responsible for water’s relatively high boiling point (compared to other molecules of similar size), its surface tension (that’s why insects can walk on water!), and its ability to dissolve so many other polar substances. So, when you see that perfect little bead of water on a leaf, thank hydrogen bonding!
Substance 2: Methane (CH4)
Now, let’s switch gears to methane. You know, the main ingredient in natural gas. Not exactly the life-giving stuff water is, but super important for heating our homes and cooking our food. How does methane behave, and what’s holding those methane molecules together (or allowing them to spread out, as in a gas)?

Let’s examine the methane molecule. It’s CH4. Carbon bonded to four hydrogen atoms. Now, carbon and hydrogen have very similar electronegativities. This means the electrons in the C-H bonds are shared pretty equally. There’s no significant pulling of electrons to one side or the other. So, even though the molecule has polar bonds, its overall shape (tetrahedral) makes it nonpolar. The positive and negative charges effectively cancel each other out.
Since methane molecules are nonpolar, they can't form hydrogen bonds. They also don't have any permanent dipoles to interact with. So, what’s left? The weakest, but still important, intermolecular forces: London dispersion forces (sometimes called Van der Waals forces, though technically that’s a broader category).
How do these work, you ask? Even in nonpolar molecules, electrons are constantly moving. At any given instant, there can be a temporary imbalance of electron density, creating a fleeting, instantaneous dipole. This temporary dipole can then induce a dipole in a neighboring molecule, leading to a weak, temporary attraction. Think of it as a very brief, accidental nudge.
The strength of London dispersion forces increases with the size and number of electrons in a molecule. Methane is a relatively small molecule, so these forces are quite weak. This is why methane exists as a gas at room temperature and pressure – its molecules don't have much reason to stick together. So, the predominant intermolecular force in methane is definitely London dispersion forces, and they’re pretty darn minimal!
Substance 3: Ammonia (NH3)
Next up, ammonia. Smells… interesting, right? Often used in cleaning products and as a fertilizer. It’s a bit like water in some ways – it's a liquid that can be quite reactive.
Let’s look at the molecule: NH3. One nitrogen atom and three hydrogen atoms. Nitrogen is a very electronegative atom, much more so than hydrogen. This means the electrons in the N-H bonds are pulled strongly towards the nitrogen. This creates a significant polar molecule, with a partial negative charge on the nitrogen and partial positive charges on the hydrogens.
Now, here’s the exciting part. Nitrogen is one of those key players (along with oxygen and fluorine) that can participate in hydrogen bonding. So, the partially positive hydrogen atoms on one ammonia molecule are strongly attracted to the partially negative nitrogen atoms on neighboring ammonia molecules. This is hydrogen bonding again!
Because nitrogen is even more electronegative than oxygen, the hydrogen bonds in ammonia are quite strong, though maybe slightly less so than in water. Still, they are definitely the predominant intermolecular forces in ammonia. This is why ammonia has a higher boiling point than methane, for example, and can exist as a liquid under certain conditions. It's the hydrogen bonds doing the heavy lifting!
Substance 4: Carbon Dioxide (CO2)
Carbon dioxide. The stuff we exhale, the stuff that makes soda fizzy, and a major player in climate change discussions. Interesting molecule, and its properties are a bit of a curveball for some.
CO2. One carbon atom double-bonded to two oxygen atoms. Now, oxygen is more electronegative than carbon. So, the C=O bonds are indeed polar. The electrons are pulled towards the oxygen atoms. This creates partial negative charges on the oxygens and a partial positive charge on the carbon.
But here’s the twist: CO2 has a linear shape. The two polar bonds are arranged on opposite sides of the carbon atom, pointing in exactly opposite directions. What does this mean? The bond polarities cancel each other out! So, even though it has polar bonds, the CO2 molecule as a whole is nonpolar. Neat, huh?

Since it’s a nonpolar molecule, CO2 can’t form hydrogen bonds. It doesn't have any permanent dipoles to interact. Therefore, the predominant intermolecular forces in carbon dioxide are London dispersion forces. These forces are relatively weak because CO2 is a small molecule. This is why CO2 is a gas at room temperature and pressure. At very low temperatures, however, these weak forces are enough to allow CO2 to solidify into "dry ice." So, while it’s a gas, those little London dispersion forces are still doing their job!
Substance 5: Sodium Chloride (NaCl)
Alright, time for something completely different: sodium chloride. You know, good old table salt! This one behaves way differently from our previous examples.
When we talk about NaCl, we're not really talking about discrete molecules in the same way we were with water or methane. Sodium chloride is an ionic compound. What does that mean? It’s formed from the transfer of electrons, not sharing. Sodium (Na) loses an electron to become a positively charged ion (Na+), and chlorine (Cl) gains that electron to become a negatively charged ion (Cl-). These oppositely charged ions are then attracted to each other very, very strongly.
These attractions aren't just between neighboring ions; they extend in all directions, forming a giant, ordered crystal lattice. This strong electrostatic attraction between positive and negative ions is called an ionic bond. And when we're talking about what holds the entire crystal together, we're talking about the overall network of these ionic bonds. There are no discrete molecules to form intermolecular attractions between!
So, while technically not "intermolecular forces" in the same sense as those between covalent molecules, the overwhelming force holding sodium chloride together is the ionic attraction within its crystal lattice. This is why salt has such a high melting point and dissolves in water (water's polarity is strong enough to pull the ions away from the lattice). If we had to pick a "predominant force," it's the powerful ionic bonding. It's on a whole other level of "stickiness" compared to the forces we've seen so far!
Substance 6: Ethanol (C2H5OH)
Let’s wrap things up with something a bit more familiar, perhaps something found in that soda or even a spirit (though let’s keep it about the chemistry, folks!). Ethanol, also known as ethyl alcohol.
Ethanol has the formula C2H5OH. Let’s break it down. We have a carbon chain (C2H5) and then an –OH group. The presence of that –OH group is a big clue. Oxygen is highly electronegative, and it's bonded to hydrogen. This means the O-H bond is very polar, with a partial negative charge on the oxygen and a partial positive charge on the hydrogen.
This polar O-H group allows ethanol molecules to participate in hydrogen bonding with each other. The partially positive hydrogen of one ethanol molecule is attracted to the partially negative oxygen of another. This is a significant force!
However, ethanol also has a nonpolar part – the ethyl group (C2H5). This nonpolar part can participate in weaker London dispersion forces with other ethanol molecules. And because the oxygen atom also has lone pairs of electrons, there are also weaker dipole-dipole interactions between the polar parts of ethanol molecules.
So, what's predominant? In ethanol, the hydrogen bonding is the strongest and therefore the most influential intermolecular force. It's what gives ethanol its relatively high boiling point compared to hydrocarbons of similar size and its ability to dissolve in water. But it’s a bit of a multi-force situation here, with London dispersion forces also playing a role!
And there you have it! A little peek into the fascinating world of what makes molecules stick together. It’s pretty amazing how these invisible forces dictate so much of the world around us, from the way water behaves to the properties of the fuels we use. Next time you’re enjoying a cold drink, maybe you’ll think about those molecules and their little dances!
