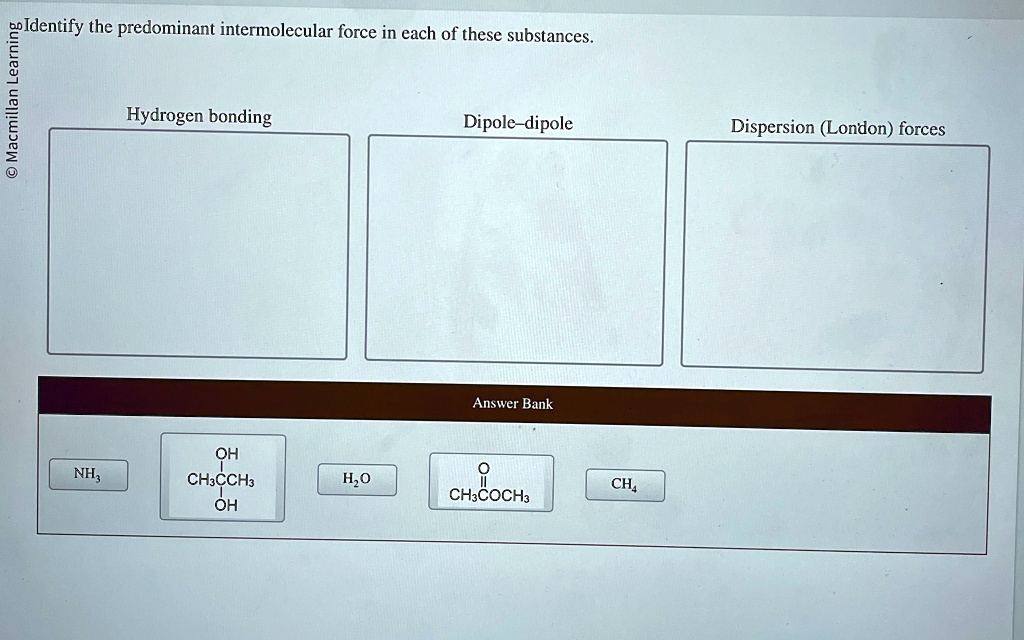
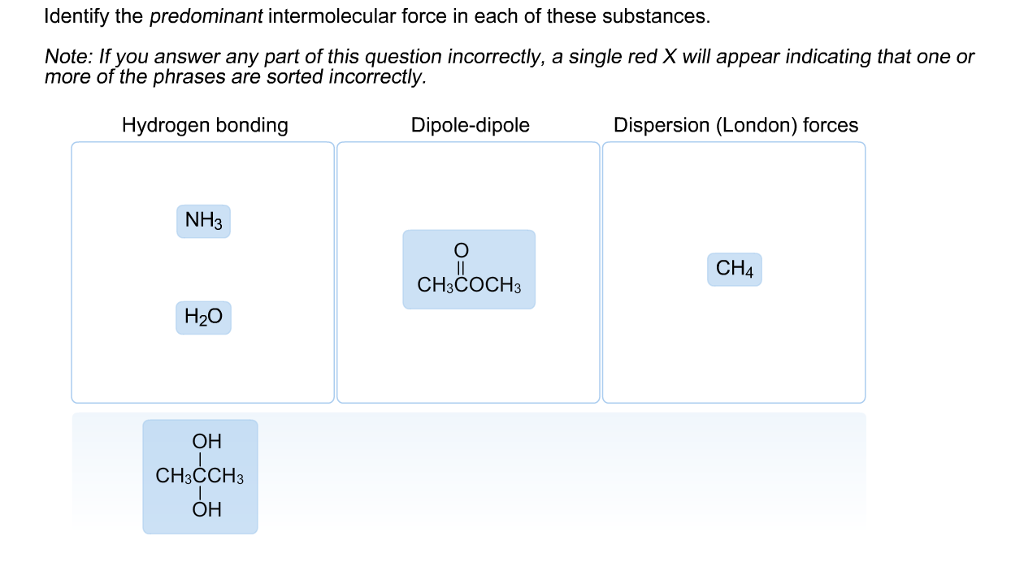
Identify The Predominant Intermolecular Force In Each Of These Substances
Ever wondered what makes different stuff stick together? It's like a secret handshake between tiny particles, and it's way cooler than you might think!
Let's dive into the wonderful world of what holds things together. It's all about these invisible forces, called intermolecular forces. They're the reason water can be liquid or ice, why some things dissolve and others don't, and even why your favorite snacks have their crunch.
Think of it like this: molecules are like little magnets, but not always. Some are more attracted to each other than others. The strength of this attraction is what we're talking about. It's what makes some substances really clingy and others a bit more aloof. It's pretty neat when you stop and think about it!
So, let's meet some of these substances and discover their signature intermolecular force. It's like giving them a personality test, and honestly, it's quite entertaining.
Water: The Ultimate Hugger!
First up, we have everyone's favorite: Water! H₂O, as the science buffs call it. You probably know water as that clear, life-giving liquid. But what makes it so special? It's all about how those water molecules love to hang out together.
The dominant force in water is called hydrogen bonding. Now, this isn't a real bond like you'd find within a single water molecule. It's more like a super strong attraction between molecules. Imagine the "H" (hydrogen) in one water molecule giving a big, warm hug to the "O" (oxygen) in another water molecule. These hugs are incredibly strong, and they're the reason water has such a high boiling point. It takes a lot of energy to pull those water molecules apart!

This hydrogen bonding is what gives water its amazing properties. It's why dew drops form on grass, why rain falls from the sky, and why ice floats (which is super important for aquatic life!). It's like water has a built-in social network, and everyone is invited to the party.
It's pretty cool, right? This one little force dictates so much about our world. It's the backbone of life as we know it, and it all comes down to these molecular hugs.
Methane: The Shy One
Now, let's look at something completely different: Methane. This is a simple molecule, CH₄. It’s the main ingredient in natural gas. Unlike water, methane molecules are pretty chill with each other. They don't really go in for big, enthusiastic hugs.
The main intermolecular force in methane is called London dispersion forces. These are the weakest of the bunch, and they're present in all molecules. But in methane, they're the star of the show because there are no stronger forces present.

Think of London dispersion forces as temporary, fleeting attractions. Imagine molecules are like a crowd of people. Every now and then, two people might accidentally bump into each other and feel a slight pull. That's kind of what's happening with methane molecules. These attractions are constantly forming and breaking, so the molecules don't stick together very tightly.
This is why methane is a gas at room temperature. The weak forces can't hold the molecules together against the constant jiggling. It's like they're all doing their own little dance and barely acknowledge each other. It’s a much more laid-back intermolecular vibe!
Ammonia: The Friendly Neighbor
Let's move on to Ammonia. You might know it for its strong smell, often found in cleaning products. The chemical formula is NH₃.

Just like water, ammonia can form hydrogen bonds! The nitrogen atom in ammonia is very electronegative, and the hydrogen atoms bonded to it are eager to find a negative neighbor. So, the hydrogen atoms in one ammonia molecule are attracted to the nitrogen atoms in other ammonia molecules. It's another form of that strong, friendly molecular interaction.
These hydrogen bonds make ammonia molecules stick together more than they would if they only had weaker forces. This is why ammonia is a liquid at room temperature, unlike methane. It's a bit more sociable, a bit more inclined to form connections. It's not quite as intensely social as water, but it definitely has a good grasp on its neighbors.
It’s this ability to form hydrogen bonds that gives ammonia its distinct properties. It shows how even small changes in molecular structure can lead to different intermolecular personalities.
Carbon Dioxide: The Diplomat
Finally, let's consider Carbon Dioxide (CO₂). This is the stuff we exhale and what plants use for photosynthesis. It's a linear molecule.
Carbon dioxide is a bit of a unique case. While it does have polar bonds (the bonds between carbon and oxygen aren't perfectly equal), the molecule itself is symmetrical. Imagine two people pulling equally in opposite directions on a rope – the rope doesn't move. That's kind of what happens with the polarity in CO₂. The molecule as a whole is nonpolar.
Therefore, the predominant intermolecular force in carbon dioxide is London dispersion forces, just like methane. These are the temporary, fleeting attractions. Because CO₂ molecules are relatively small and don't have any strong charges or polarity to speak of, these weak forces are all that's holding them together.
This is why CO₂ is a gas at room temperature and pressure. The weak London dispersion forces are easily overcome. It's like a crowd of people who are polite but don't really engage deeply with each other. They move past each other freely. It’s the essence of being a gas – a lot of space and little interaction!
So there you have it! A quick peek into the fascinating world of intermolecular forces. It’s amazing how these tiny, invisible interactions shape the world around us in such big ways. Next time you see water, methane, ammonia, or even just breathe out some CO₂, remember the invisible forces at play!
