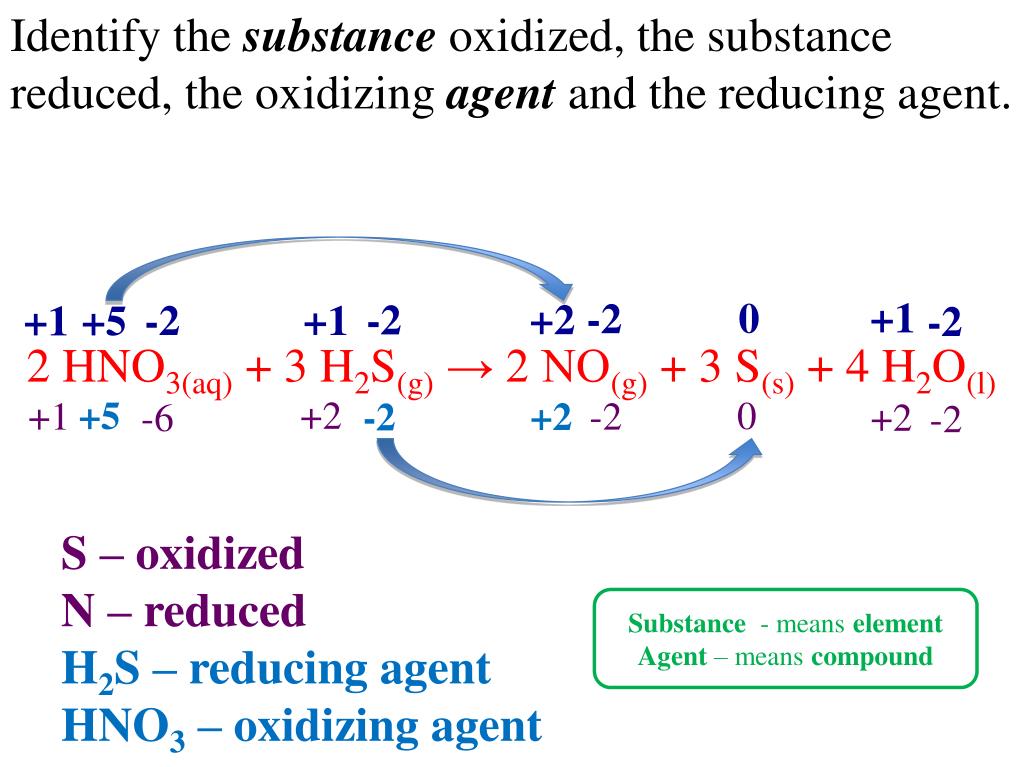
Identify The Oxidized Substance The Reduced Substance The Oxidizing Agent
Alright, let's talk about a concept that sounds a bit like something out of a mad scientist's lab, but is actually happening all around us, all the time. We're diving into the wonderfully weird world of redox reactions. Now, before you start picturing bubbling beakers and frantic scribbling on a chalkboard, let's break it down. Think of it less like a chemistry experiment and more like a really dramatic potluck dinner.
You know those moments when you're trying to figure out who brought what to the party? "Who brought the epic seven-layer dip? And who, for the love of all that is cheesy, forgot the napkins?" Redox reactions are kind of like that, but instead of food and party supplies, we're dealing with electrons. These tiny, zippy little things are the lifeblood of chemistry, and in redox reactions, they're being passed around like a hot potato at a toddler's birthday party.
The Great Electron Hand-Off
So, what's the deal? In a nutshell, a redox reaction is just a fancy way of saying that electrons have been transferred from one substance to another. It's like a cosmic game of "Got Your Electrons!"
Think about it like this: Imagine you've got two friends. One friend, let's call her Agnes, has way too many shiny marbles. She's practically drowning in them. The other friend, Bartholomew, has a gaping hole in his marble collection. He needs marbles. So, Agnes, being the generous (or perhaps slightly overwhelmed) soul she is, decides to share. She gives some marbles to Bartholomew.
That's your redox reaction right there! Agnes is the one losing marbles, and Bartholomew is the one gaining them. Simple, right? Now, let's sprinkle in some science-y terms, because, well, that's what we're here for.
Meet the Star Players: Oxidized and Reduced
In our marble analogy, Agnes, the one who lost marbles, is the one who got oxidized. And Bartholomew, the one who gained marbles, is the one who got reduced.
Now, I know what you're thinking. "Reduced? Does that mean he got smaller?" Not exactly. In chemistry, "reduced" has a very specific meaning related to those precious electrons. It means gaining electrons. So, Bartholomew, by gaining marbles (our stand-in for electrons), got reduced.
And "oxidized"? That means losing electrons. Agnes, by giving away her marbles, got oxidized.
It’s a bit of a quirky naming convention, I'll grant you. You’d think "oxidized" would mean something got more oxygen-y, and while it often does, the fundamental idea is the loss of electrons. It’s like saying your car got "detoxed" when it ran out of gas – it’s a bit of a misnomer, but we all understand what you mean.
The "OIL RIG" Cheat Sheet
To help you remember this, chemists often use a handy mnemonic: OIL RIG.

Oxidation Is Loss (of electrons).
Reduction Is Gain (of electrons).
So, whenever you're scratching your head, just think: "Is it OIL or RIG? Did they lose 'em, or did they gain 'em?" It’s like having a little chemical cheat sheet in your pocket.
When Things Get Spicy: The Oxidizing Agent
Now, back to our potluck. Agnes gave away marbles, and Bartholomew gained them. But who facilitated this whole exchange? Who was the catalyst for this electron (or marble) transfer?
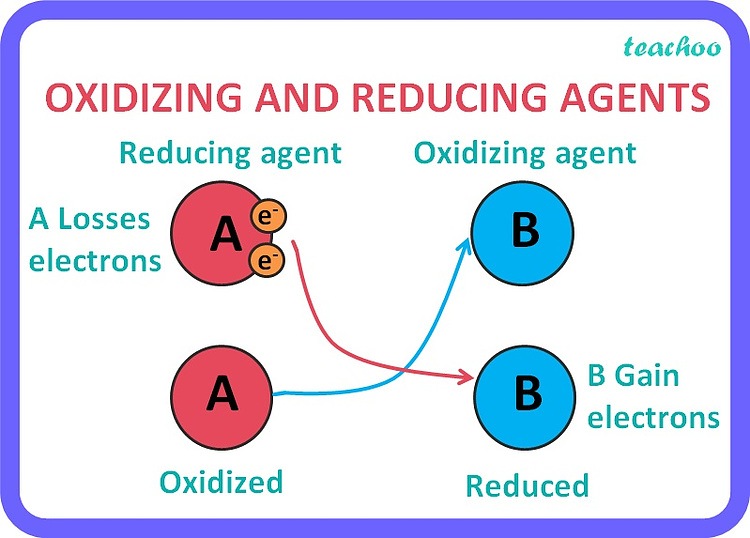
That's where the oxidizing agent comes in. The oxidizing agent is the substance that causes the other substance to be oxidized. In our marble scenario, Agnes was the one losing marbles, so she got oxidized. But what made her lose them?
Let's adjust the analogy a bit. Imagine Agnes has a really, really tempting pile of marbles. Bartholomew is practically drooling. Bartholomew, in his eagerness to gain marbles, essentially "attacks" Agnes's marbles. In doing so, he's the one taking them.
This is where it gets a little mind-bendy. The oxidizing agent is the substance that accepts electrons. It's the electron-grabber. So, in our potluck, the person who is most eager to take your amazing brownies is the oxidizing agent. They are making you (the brownie-maker) give them up, thus oxidizing you (of your brownies).
Think of it this way: the oxidizing agent is the superhero that swoops in and oxidizes something else. It's the one that makes the magic happen. It gets reduced in the process itself because it's gaining those electrons. It's like the person who takes the last slice of pizza – they’re causing you to not have that slice (oxidizing your pizza supply), and in return, they’re getting their fill (getting reduced, in a pizza sense).
So, the oxidizing agent is the one that oxidizes another substance. It’s a bit like calling a detective the "culprit finder" – they find the culprit, but they’re also involved in the situation. The oxidizing agent is the electron-taker, and by taking electrons, it causes the other guy to lose them (get oxidized). And in taking those electrons, the oxidizing agent itself gets reduced. It’s a win-win for the oxidizing agent, in a chemical sense!

And The Other Guy? The Reducing Agent
If we have an oxidizing agent, it stands to reason we need its opposite, right? Enter the reducing agent.
The reducing agent is the substance that causes the other substance to be reduced. It's the electron-giver. In our marble example, Bartholomew was the one gaining marbles, so he got reduced. But what enabled him to get reduced?
The reducing agent is the one that donates electrons. It's the electron-provider. It's the substance that is most eager to give away its electrons. Think of it as the generous friend who always has extra pens to lend.
The reducing agent is the one that reduces another substance. It’s making something else gain electrons. And in giving away its own electrons, the reducing agent itself gets oxidized. It’s like the person who brings way too much homemade jam to the party. They’re giving jam away (reducing the amount of jam they have), and in doing so, they’re making everyone else’s toast a lot tastier (oxidizing everyone else’s breakfast experience, in a delicious way!).
So, to recap the players:
- The Oxidized Substance: The one that LOSES electrons (like Agnes with her marbles).
- The Reduced Substance: The one that GAINS electrons (like Bartholomew with his marbles).
- The Oxidizing Agent: The substance that ACCEPTS electrons, causing another substance to be oxidized. It ITSELF gets REDUCED.
- The Reducing Agent: The substance that DONATES electrons, causing another substance to be reduced. It ITSELF gets OXIDIZED.
See the beautiful, albeit slightly confusing, symmetry? The oxidizing agent oxidizes something else but gets reduced. The reducing agent reduces something else but gets oxidized. It's like a chemical dance where everyone gets a turn in the spotlight, but also a little bit of a makeover.
Real-World Examples: Where the Electrons Flow
Okay, enough with the marbles. Let’s see this in action in the wild!
Rust Never Sleeps (But It Does Oxidize!)
Think about your car, or that old bicycle in the shed. What happens when they get wet and are left out in the elements? They rust! Rust is basically iron oxide, and it’s a classic example of oxidation.
In this case, iron (Fe) is the substance that gets oxidized. It loses electrons to the oxygen in the air, and often with the help of water, to form rust. So, iron is the oxidized substance.
And what's causing this iron to lose its electrons? That would be oxygen (O₂)! Oxygen is a notorious electron-grabber. It's like the ultimate oxidizing agent. It loves to steal electrons. So, in the rusting process, oxygen is the oxidizing agent. It causes the iron to oxidize, and in the process, the oxygen itself gets reduced (gains electrons).
So, iron is oxidized, and oxygen is the oxidizing agent. Simple as that. Your car is slowly but surely turning into a crumbly mess because of the electron-stealing power of oxygen!
Batteries: The Electron Highways
Ever wonder how your TV remote or your flashlight works? They rely on batteries, and batteries are all about controlled redox reactions!
Inside a battery, you have two different materials that are eager to play the electron game. One material will readily donate electrons (it’s the reducing agent), and the other material will readily accept electrons (it’s the oxidizing agent).
Let’s say you have a simple zinc-copper battery. Zinc (Zn) is a pretty good electron donor. It's the reducing agent. It readily loses electrons and gets oxidized.
On the other side, you have copper ions (Cu²⁺), which are itching to gain electrons. They are the oxidizing agent. They readily accept electrons and get reduced.
So, the zinc gives up its electrons, gets oxidized. The copper ions grab those electrons, get reduced. This flow of electrons through an external circuit is what powers your device. It's like a tiny, organized electron race track!

Without these redox reactions, your gadgets would be about as useful as a chocolate teapot.
Digestion: Your Inner Chemical Powerhouse
Even your own body is a hotbed of redox reactions! When you eat food, your body breaks it down to get energy, and this process involves a lot of electron shuffling.
Think about the process of cellular respiration, where your cells convert glucose (sugar) into energy. Glucose is oxidized – it loses electrons. Various molecules within your cells act as electron carriers, passing those electrons along.
And what's often the final electron acceptor in this process? Oxygen (O₂)! Yep, our old friend oxygen is back as the ultimate oxidizing agent, accepting electrons from the molecules that were originally part of the glucose. This helps to create ATP, the energy currency of your cells. So, you're basically a walking, talking redox reaction fueled by snacks and oxygen. Pretty cool, huh?
Why Should You Care?
You might be thinking, "Okay, this is neat, but why do I need to know this?" Well, understanding redox reactions helps you understand a whole lot about the world around you.
It explains why things rust, how batteries work, how we get energy from food, and even how the atmosphere stays balanced (to some extent). It's the hidden engine behind so many processes.
Plus, it’s a fantastic party trick. The next time someone’s looking confused about why their apple is turning brown, you can casually drop in, "Ah, that's just oxidation! The apple is losing electrons to the air, and it's getting oxidized. Pretty neat, right?" You’ll be the resident chemistry guru, or at least the person who knows slightly more than everyone else about browning fruit.
So, next time you see something changing color, or a battery powering up, remember the great electron hand-off. Remember OIL RIG. Remember the oxidized substance, the reduced substance, the oxidizing agent, and the reducing agent. They’re the unsung heroes of the chemical world, quietly making everything happen, one electron at a time. And that, my friends, is pretty darn cool.
