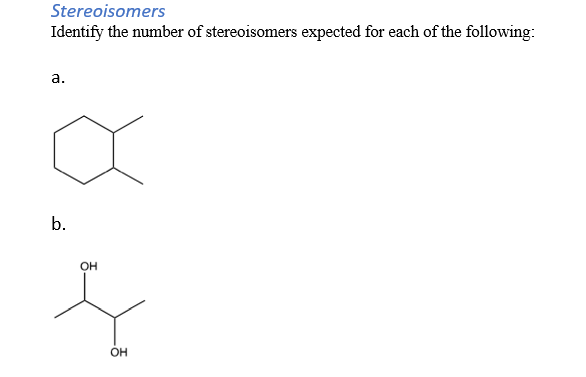
Identify The Number Of Stereoisomers Expected For The Following:
Ever found yourself staring at a complex puzzle, a challenging recipe, or maybe even a particularly tricky outfit combination, and felt that delightful buzz of mental engagement? That’s the magic of problem-solving, and today, we’re diving into a fascinating corner of it: identifying stereoisomers. It might sound like something out of a science lab, but understanding stereoisomers is like unlocking a secret level in how we perceive the world around us, from the taste of your favorite candy to the efficacy of life-saving medicines.
So, what exactly are stereoisomers? Think of them as molecules that have the same chemical formula, meaning they’re made of the same building blocks (atoms). However, the way these blocks are arranged in three-dimensional space is different. This subtle shift can lead to dramatically different properties, and that’s where the real fun – and utility – comes in! For everyday life, this concept underlies everything from the distinct flavors of enantiomers (a type of stereoisomer) – like how one can taste like mint and the other like caraway – to the precise shapes of drugs that fit perfectly into their target receptors in our bodies. Without understanding stereoisomers, developing effective medications or even creating specific food flavorings would be a matter of pure luck!
The beauty of this concept is its wide-ranging application. In organic chemistry, the cornerstone of so much modern science and technology, identifying stereoisomers is crucial for synthesizing new compounds, understanding reaction mechanisms, and ensuring the purity and efficacy of products. For instance, in the pharmaceutical industry, one stereoisomer of a drug might be therapeutically beneficial, while another could be inactive or even harmful. Remember the thalidomide tragedy? It’s a stark reminder of the critical importance of distinguishing between different stereoisomers. Beyond medicine, think about the fragrances you love; their unique scents often stem from specific stereoisomeric arrangements.
Now, how do we tackle the challenge of identifying the number of stereoisomers expected for a given molecule? This is where the "puzzle" aspect comes into play! It often involves looking for chiral centers – typically carbon atoms bonded to four different groups. For each chiral center, there are two possible spatial arrangements. A common rule of thumb is that for a molecule with n chiral centers, there can be up to 2n stereoisomers. However, it’s important to remember that sometimes symmetry can reduce this number. It’s a bit like a branching path; each chiral center presents a choice, and you need to count all the unique paths you can take.
To enjoy this mental exercise even more, consider breaking it down. Start with simpler molecules and gradually work your way up. Visual aids, like molecular modeling kits or online simulators, can be incredibly helpful. They allow you to physically (or virtually) manipulate the molecules and see the different spatial arrangements firsthand. Don’t be afraid to sketch things out! Drawing the molecules and their potential arrangements can clarify your thinking. The goal isn't just to get the right answer, but to develop an intuitive understanding of molecular architecture. So, the next time you encounter a chemical structure, embrace the challenge – you might just discover a whole new appreciation for the intricate, three-dimensional world of molecules!
