Identify The Most Appropriate Base To Deprotonate The Following Compound:

Hey there, chemistry curious! Ever feel like your everyday life could use a little… oomph? Maybe a sprinkle of science to spice things up? Well, get ready, because today we’re diving into a topic that’s surprisingly relatable, even if it sounds a tad complex at first: identifying the most appropriate base to deprotonate a compound. Think of it like picking the perfect outfit for a specific occasion – you wouldn't wear stilettos to hike a mountain, right? Same principle applies in the lab, or in our imaginary, chemistry-infused lives.
Now, before you start picturing bubbling beakers and mad scientists (though that’s fun too!), let’s break this down. "Deprotonate" is just a fancy word for removing a hydrogen atom that’s attached to another atom in a molecule, and taking its electron with it. Think of that hydrogen as a tiny, energetic extrovert who’s just a little too eager to share its space. A "base" is essentially a molecule that’s really good at inviting that extrovert (and its electron) to leave. And "appropriate"? That’s where the real magic happens, just like choosing the right filter for your sunset selfie.
We’re going to be looking at a specific compound, and our mission, should we choose to accept it, is to figure out which base is the gentlest yet most effective one for the job. It’s all about finding that sweet spot – a base strong enough to do the deed, but not so aggressive that it causes a chaotic, unwanted reaction. It's the ultimate chemistry matchmaking.
The Art of the Chemical Connection
Imagine you're at a party, and you want to introduce two people. You wouldn’t just shove them together, right? You'd consider their personalities, their interests, what they might have in common. That’s what we're doing with our base and our compound. We’re looking for a mutual understanding, a chemical handshake that leads to a successful deprotonation.
The key player in this dance is something called acidity. Every hydrogen atom in a molecule has a certain level of "stickiness" or "looseness." Some are practically begging to leave, while others are pretty content to stay put. The easier it is for a hydrogen to leave (meaning it's more acidic), the weaker the base needs to be to snatch it away. Conversely, if a hydrogen is really clinging on, you’ll need a stronger, more determined base to convince it to depart.
Think of it like this: a weak acid is like a shy person who only whispers their opinion. A strong acid is like someone who’s ready to belt out a karaoke anthem. And a base? Well, the base is the audience. A strong base is a roaring crowd, ready to cheer for any hydrogen that steps up. A weak base is a polite clapper, only moved by the most courageous of hydrogen performers.
Meet Our Guest Star: The Compound
So, what’s our featured molecule today? Let’s paint a picture. Imagine a molecule that’s a bit like a cozy little cottage. It has a central structure, and on one side, there’s a hydrogen atom attached to an oxygen atom. This is our target hydrogen, the one we want to deprotonate. This oxygen atom is already a bit electronegative, meaning it has a slight pull on its electrons. This makes the hydrogen attached to it a tad more eager to leave than, say, a hydrogen stuck to a carbon atom in a more robust part of the molecule.
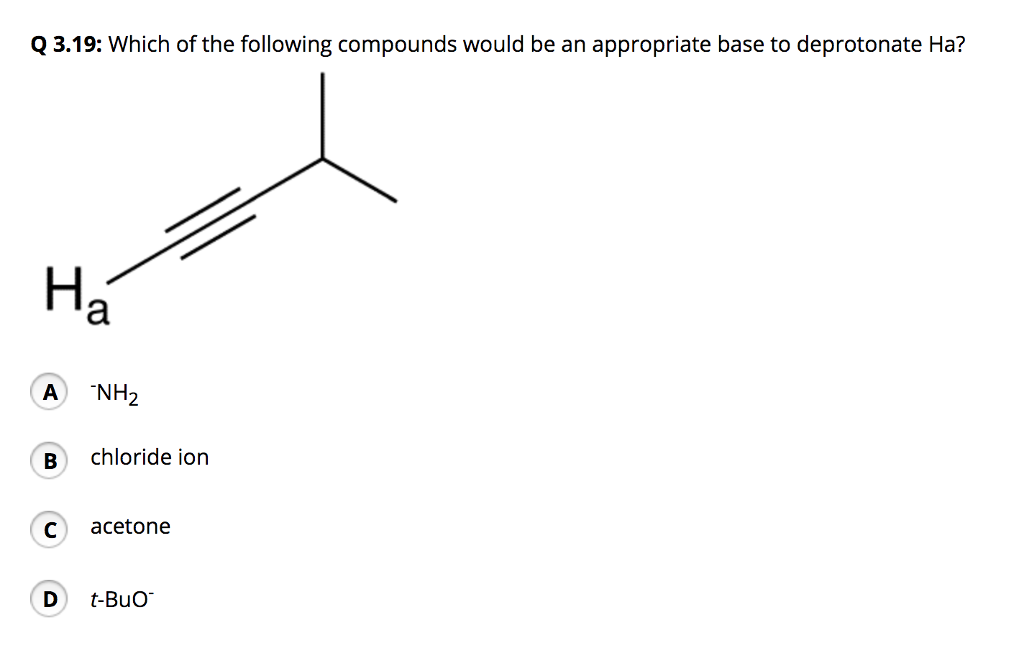
This specific hydrogen is like that friend who’s always on the verge of saying something witty, but just needs a little nudge. It’s moderately acidic. It’s not a rockstar demanding a spotlight, but it's definitely not hiding in the shadows.
The Lineup of Potential Bases
Now, for our potential bases! We have a few contenders, each with their own personality and strength. Let’s introduce them:
Base A: The Gentle Encourager
This base is like a warm hug. It’s a relatively weak base. Think of it as that friend who’s always there to offer a comforting word, but isn’t going to force you into anything. It’s good for situations where you want to be extra careful and avoid any unintended drama.
Base B: The Confident Conversationalist
This base is a bit more assertive, but still friendly. It’s a moderately strong base. Imagine someone who can steer a conversation with ease, making everyone feel comfortable while still getting their point across. It’s a great all-rounder.

Base C: The Powerhouse
This base is the life of the party, no doubt. It’s a very strong base. Think of a rock concert – it’s loud, it’s energetic, and it gets things done with a bang. But with great power comes… well, the potential for overkill.
The Acid-Base Dance: Making the Right Match
Here's where the strategy comes in. To deprotonate our moderately acidic hydrogen, we need a base that’s at least as strong, if not stronger, than the acid we're trying to form. This is a fundamental rule of thumb in chemistry, and it's as reliable as a good cup of coffee on a Monday morning.
Let’s think about it in terms of energy. Removing the hydrogen requires a certain amount of energy. A base provides the energy to make this happen. If the base isn't strong enough, it’s like trying to push a heavy door with a feather – it’s just not going to budge.
So, let’s analyze our options:
- Base A (Weak Base): This is probably not going to be strong enough to pull away our moderately acidic hydrogen. It’s like trying to have a serious heart-to-heart with someone who’s barely listening. You might get a few nods, but no real change.
- Base B (Moderately Strong Base): This looks promising! It’s strong enough to engage our moderately acidic hydrogen in a meaningful chemical conversation and successfully remove it. It’s the perfect balance of power and finesse.
- Base C (Very Strong Base): This base could deprotonate our target hydrogen, but it might be a bit much. Imagine using a sledgehammer to crack a walnut. It’ll work, but you might end up damaging more than you intended. A very strong base might react with other parts of the molecule that we don't want it to touch, leading to unwanted side reactions. It’s like inviting a bulldozer to your tea party – effective, but potentially destructive.
The Winning Choice: Base B
Therefore, the most appropriate base to deprotonate our compound is Base B, the moderately strong base. It’s the Goldilocks choice: not too weak, not too strong, but just right for the job. It’s efficient, effective, and minimizes the risk of collateral damage to our lovely molecule.
This concept is super important in organic chemistry. When chemists are designing reactions, they’re constantly thinking about which reagents (that’s a fancy word for chemicals used in reactions) will interact in the desired way without causing chaos. It’s like being a skilled conductor, ensuring all the instruments play their part harmoniously.
Fun Facts and Chemical Culture
Did you know that the strength of a base is often measured by something called its pKb value? A lower pKb means a stronger base. It’s kind of the opposite of how we usually think about numbers and strength, but hey, chemistry loves to keep us on our toes!
And in pop culture? Think about your favorite heist movies. The crew has to pick the right tools for the job, right? A safecracker needs precision tools, not a crowbar. Similarly, chemists need the right base to achieve a clean and successful deprotonation. It’s all about the right tool for the right job.
Even in cooking, we see similar principles. You wouldn't use a blowtorch to melt butter for your toast, would you? You’d use a gentle heat. Or when you’re proofing yeast for bread, you need a specific temperature range – too hot, and you kill the yeast; too cold, and it won’t activate. It’s all about finding that perfect chemical environment.
A Practical Analogy for Everyday Life
So, how does this tie back to your daily grind? Think about managing your energy levels. You have days when you’re naturally bursting with energy, ready to tackle anything (that’s like a strong acid). Other days, you’re feeling a bit more mellow, needing a gentler approach (like a weaker acid).
And what about your "bases" – the people, activities, or even snacks that help you recharge and feel your best? Sometimes, you need a high-energy friend who pulls you out of your shell (a stronger base for a low-energy you). Other times, you need a quiet evening with a book (a gentler base for a stressed-out you).
Identifying the most appropriate base to deprotonate a compound is, in essence, about understanding the balance of forces and the specific needs of a situation. It’s about finding that perfect synergy that leads to a desired outcome without unnecessary collateral damage.
In our lives, this translates to knowing when to be assertive and when to be gentle, when to push and when to let go. It’s about recognizing the subtle signals and choosing the right approach to navigate our own personal chemistry. So, the next time you’re making a decision, big or small, remember our chemical friends and their careful dance. Sometimes, the most elegant solutions are the ones that are just right – not too much, not too little, but perfectly balanced. Now go forth and apply a little chemistry to your day!
