Identify The Most Acidic Proton On The Following Compound
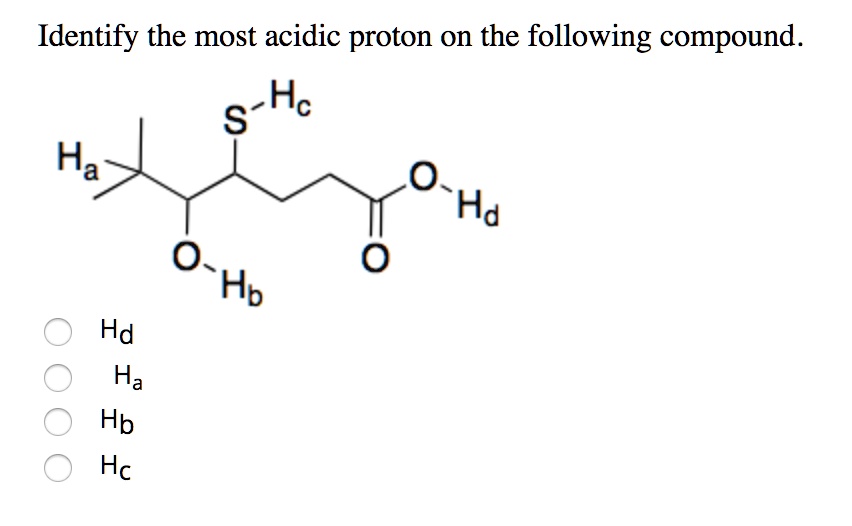
Ever stared at a molecule and felt like you were trying to solve a riddle? Yeah, me too. Especially when someone throws a bunch of atoms together and asks, "Which proton is the most acidic?" It sounds like something a superhero might ask before a big fight.
Honestly, sometimes I think these chemistry folks have a secret handshake for identifying these "acidic protons." It's like they've been practicing in a hidden lab, whispering the ancient incantations of electron pulling and resonance stabilization.
And then there's the compound itself. It’s a whole neighborhood of atoms, all hanging out. Some are chummy, some are a bit standoffish. We're basically trying to find the one atom that's really eager to get rid of its tiny hydrogen friend.
Think of it like this: you're at a party, and everyone has a balloon. Most people are happy holding onto their balloons. But one person is just itching to let theirs go, maybe because it’s tied to something really boring, or maybe they just want to see it float away really fast.
Our job, then, is to be the party pooper detective. We need to sniff out the balloon-releaser. And in the world of molecules, that means finding the most acidic proton.
Now, the compound we're looking at is quite the character. It’s got a bit of everything. There are some happy, stable little groups. And then there are some that seem a little… stressed. Like they’re constantly on edge.
We're on the hunt for that one special hydrogen. The one that’s thinking, "Get me outta here!" every single second. It's not just about being a proton; it's about being a proton with a serious case of wanderlust.
Imagine you have a bunch of friends, and you're all holding hands. Most of you are pretty comfortable. But one of you is secretly planning an escape. They're looking for the weakest link in the chain. That's our proton!
So, we scan our compound. We look at all the hydrogens. Are they just chilling, attached to a perfectly content atom? Or are they feeling the pressure? Are they next to something that’s just greedy for electrons?
This is where the fun really begins, right? It’s like a microscopic game of "Who's the Most Likely to Break Up?" We're looking for the bond that's most likely to snap.
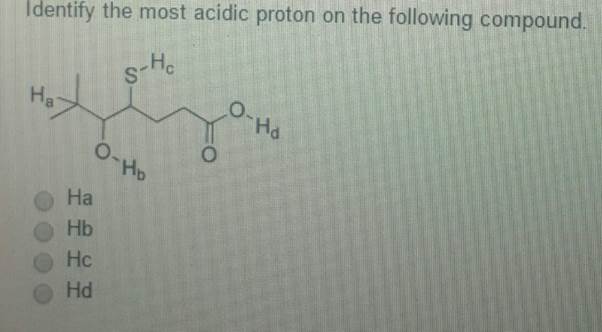
And what makes a proton more likely to pack its bags and leave? Well, it’s all about what’s left behind. If the atom that the proton leaves behind is super happy about it, then that proton was probably pretty eager to go.
Think of it like leaving a bad relationship. If your ex is thrilled that you’re gone and immediately finds someone better, well, you probably made the right decision, didn't you? It’s the same for our proton.
We're looking for the proton that, when it leaves, creates a really stable situation for the remaining atom. Like a perfectly balanced seesaw. Or a perfectly organized sock drawer.
And in our specific compound, there are a few places where a proton could potentially be. There are those friendly oxygen atoms, always willing to share. And then there are those carbon atoms, sometimes a bit more clingy.
But there’s one spot that just screams "escape route!" It’s like a tiny, molecular emergency exit. A proton’s personal ticket to freedom.
We have to consider how electronegative the neighboring atoms are. Electronegativity is just a fancy word for how much an atom likes to hog electrons. The more it hogs, the more it pulls on that proton.
And when an atom pulls really hard, it weakens the bond. It's like stretching a rubber band. Eventually, it’s going to snap!
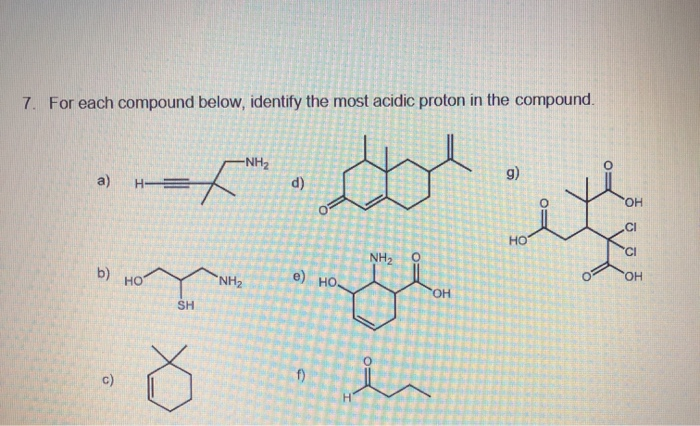
Our compound has some atoms that are champions at hogging electrons. They're the rock stars of the electron world. And where these electron-hogging superstars hang out, the protons nearby tend to get a little… antsy.
We also look at something called resonance. This is like having multiple personalities, but in a good way for stability. If the atom left behind can spread out its negative charge over a few different places, it’s much happier.
Imagine a rumor spreading through a small town versus a big city. In a small town, everyone knows the rumor. In a big city, it gets spread out, and no one person is burdened with it.
So, we're looking for a proton that, when it vacates its post, leaves behind an atom that can participate in this "rumor spreading" of negative charge. This makes the leftover atom super stable.
And in our particular molecule, there's a prime candidate. It’s attached to an atom that’s already feeling the electron-pulling pressure. It's like living on the edge of a cliff, even before you've made the jump.
This proton is probably having daily existential crises. "Am I going to stay? Am I going to go? What if I do go, and it's just… Awkward?"
But then, the magic happens. When this specific proton does leave, the resulting anion (that's the atom with the extra negative charge) is incredibly stable. It’s like it suddenly found its zen.

It’s not just about being next to a grumpy atom. It's about what happens after the break-up. Chemistry is all about the follow-through, apparently.
So, if you were to point a tiny, molecular spotlight on our compound, it would land squarely on this one particular hydrogen. It’s the one that’s most ready for its close-up as a free-floating proton.
It’s the proton that’s been practicing its dramatic exit. The one that’s been eyeing the escape hatch for a while now.
Think of it as the star of our "Most Acidic Proton" show. The one we’ve been waiting for. The one that’s going to make the biggest splash, or in this case, the most stable anion.
It’s not about who looks the most acidic. It's about who acts the most acidic. And this proton, my friends, is a natural performer.
So, when you see a molecule like this, don't get overwhelmed. Just look for the proton that’s got that "get me out of here" vibe. The one whose departure leads to a happy, stable ending.
It’s the proton that’s the most willing to embrace change. The one that’s ready to embark on its solo journey. It’s the hero of our little molecular story.
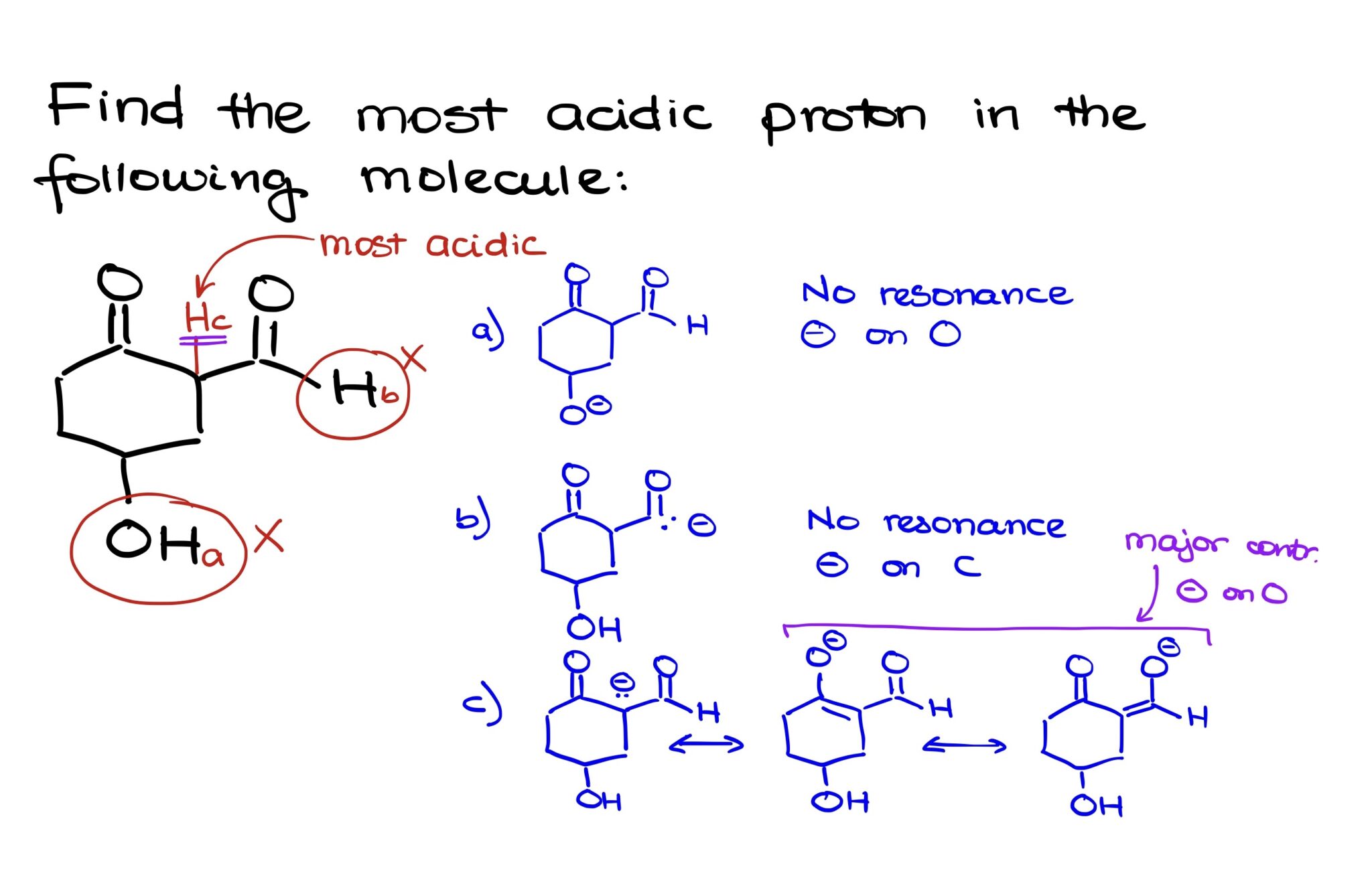
And in this specific compound, there's no doubt in my mind. There's one proton that is the undisputed champion. The most acidic proton.
It's the one that’s just begging to be set free. It’s the star of the show. The proton with the biggest dreams.
So next time you see a molecule, channel your inner chemistry detective. Look for the proton that’s got that sparkle in its eye. The one that’s ready for its adventure. It’s the most acidic proton, and it's waiting for you to identify it.
It's not rocket science, but it does involve understanding which atoms are a little bit of a diva about their electrons. And that, my friends, is the secret to finding our little proton superstar.
It's all about the stability game. The proton that can cause the most stable situation when it leaves is the winner. Simple, right? Well, as simple as we can make it.
And for this compound, the answer is clear. It's the proton that's got the best exit strategy. The one that's truly ready to fly.
So there you have it. The mystery of the most acidic proton. It's all about looking for that special hydrogen that's just itching for a change of scenery. The one that makes the rest of the molecule sigh with relief when it’s gone.
It’s the proton that’s the real deal. The one that’s not afraid to make a statement. The one that’s the undisputed most acidic proton.
