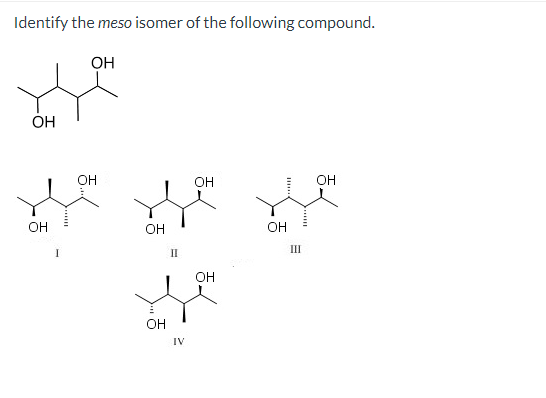
Identify The Meso Isomer Of The Following Compound.
Ever feel like you're looking at the same thing, but… just a little bit different? That’s the magic of stereoisomers, and today we’re diving into a particularly cool type: meso compounds. Think of it like a pair of hands. They’re both hands, but one is a left and the other is a right. They're related, but not quite identical. Similarly, some molecules can exist in different spatial arrangements, and figuring out these variations can be surprisingly fun and super useful in the world of chemistry!
Understanding these subtle spatial differences isn't just for super-smart scientists in labs. It's fundamental to how many things around us work. From the medicines we take to the flavors we taste, the specific 3D shape of a molecule can dramatically change its properties and how it interacts with other molecules. For instance, one form of a drug might be incredibly effective at treating an illness, while its mirror image could be inactive or even harmful! So, getting a handle on identifying these different forms, like our star of the show, the meso compound, is a bit like being a molecular detective. You’re piecing together clues to understand the true identity and behavior of a substance.
The purpose of identifying a meso compound is to recognize a specific type of stereoisomer that has a unique property: it's achiral, meaning it’s not chiral (like your hands are). This might sound contradictory because meso compounds often contain chiral centers (atoms with four different groups attached, making them "handed"). The trick with a meso compound is that it possesses an internal plane of symmetry that makes the entire molecule superimposable on its mirror image. This internal symmetry is what cancels out the "handedness" that the chiral centers would otherwise impart.
The benefits of being able to spot a meso compound are significant. For chemists, it helps predict and explain a molecule's physical and chemical properties. For example, chiral compounds often rotate plane-polarized light (they're optically active), but meso compounds, due to their symmetry, do not. This is a crucial distinction in experiments and analyses. Furthermore, in the synthesis of complex molecules, controlling the formation of specific stereoisomers is vital. Identifying whether a product is a meso compound or a chiral stereoisomer guides reaction design and purification strategies. It’s about precision and understanding the intricate dance of atoms in three dimensions.
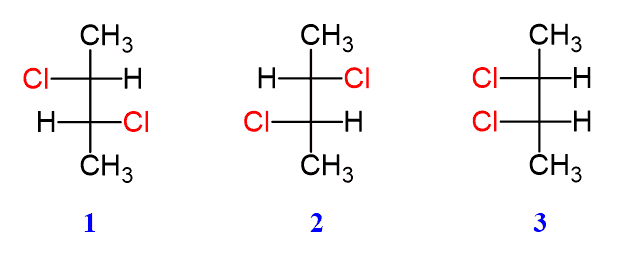
Now, let’s get to the fun part: identifying the meso isomer of a given compound. This often involves looking for specific structural features. We're usually dealing with molecules that have two or more chiral centers. The key clue for a meso compound is the presence of an internal plane of symmetry. Imagine drawing a line right through the middle of the molecule. If you can slice it in half such that one half is the perfect mirror image of the other, and the molecule can be rotated to perfectly overlap with its mirror image, then you've likely found a meso compound!

To illustrate, let's consider a hypothetical example. Imagine a molecule with two chiral centers. If the configuration of one chiral center is 'R' (right-handed) and the other is 'S' (left-handed), and there’s an internal plane of symmetry that bisects the molecule, it’s a meso compound. The 'R' and 'S' configurations effectively cancel each other out in terms of overall chirality due to the symmetry. It's like having a perfectly balanced seesaw – even though it has two ends, the overall state is balanced and symmetrical.
So, when you’re presented with a molecule and asked to identify its meso isomer, here’s your detective checklist:
- Look for multiple chiral centers: Meso compounds typically have at least two.
- Examine the symmetry: This is the most critical step. Can you find an internal plane of symmetry that divides the molecule into two mirror-image halves?
- Consider the configurations: If you know the configurations (R/S) of the chiral centers, a meso compound will often have opposite configurations (e.g., R and S) at its chiral centers, along with that crucial plane of symmetry.
- The mirror image test: Mentally (or by drawing) create the mirror image of the molecule. If the original molecule can be rotated to become identical to its mirror image, it’s a meso compound.
It's a bit like solving a 3D puzzle. The structure of the molecule gives you the pieces, and the presence (or absence) of a plane of symmetry tells you how they fit together in space to create a unique, symmetrical entity. It’s a concept that’s both intellectually stimulating and practically relevant, making the exploration of meso isomers a genuinely engaging journey into the fascinating world of molecular architecture.
Embrace the symmetry! That’s your golden ticket to identifying a meso compound. It's the hidden balance that makes these molecules special.
So, the next time you encounter a molecule with multiple potential chiral centers, don't just look at the atoms and bonds. Peer deeper into its spatial arrangement. Look for that elegant internal symmetry. It's the key to unlocking the secret identity of the meso isomer, a molecule that’s both chiral in its components and achiral in its entirety – a true testament to the surprising elegance of chemical structures!
