Identify The Location Of Oxidation In An Electrochemical Cell
Alright, let's chat about something that sounds fancy but is actually as common as forgetting where you put your keys: oxidation in an electrochemical cell. Think of it like a tiny, invisible drama playing out, but instead of actors forgetting their lines, it's all about electrons doing a disappearing act. And trust me, we’ve all been there, right? That moment of "Wait, where did that thing go?" Yep, that's kind of what oxidation is about.
So, picture this: you've got your trusty battery. Whether it's powering your remote control, your phone, or even those ridiculously bright bike lights, it's a mini-world of chemical reactions. And at the heart of this battery's magic is the tango between oxidation and its partner, reduction. They're like the Laurel and Hardy of the electro-chemical world – always together, always doing their thing. You can't have one without the other, it’s a package deal, like socks and shoes. Or, you know, coffee and the overwhelming desire to function before 9 AM.
Now, when we talk about an "electrochemical cell," it's just a fancy name for any setup where a chemical reaction creates electricity, or electricity drives a chemical reaction. Think of a car battery, or even the humble potato clock you might have made in school. Remember those? A rusty nail, a copper coin, a potato… and voilà! A faint glow from an LED. Those little guys were little electrochemical cells, and they were up to some serious oxidation business.
So, where does this "oxidation" business happen? It's all about the loss of electrons. Electrons are like tiny, energetic little sprites, and when an atom or molecule decides to shed one (or more) of these sprites, that's oxidation. Imagine an atom that's just a little too keen to share its belongings. It's like that friend who always leaves their charger at your place, except this is more permanent. They're giving away their electrons.
In our electrochemical cell, we've got two main players, usually two different metals or chemical compounds, hanging out in what we call "half-cells." Each half-cell is a little kingdom, and within these kingdoms, these oxidation and reduction reactions are taking place. The key to spotting oxidation is to look for the element that's getting electron-deprived. It's the one that's losing its electron-sprite.
Finding the Culprit: The Electron Loser
Let's make this even more relatable. Think about your kitchen. You've got a sink full of dirty dishes (reduction, maybe?) and a pantry full of food you haven't touched (oxidation, potentially!). You're the battery, and you’re either using energy (electricity) to clean those dishes, or the act of cooking (chemical reaction) is giving you the energy to do something else. It’s a bit of a stretch, I know, but stick with me!
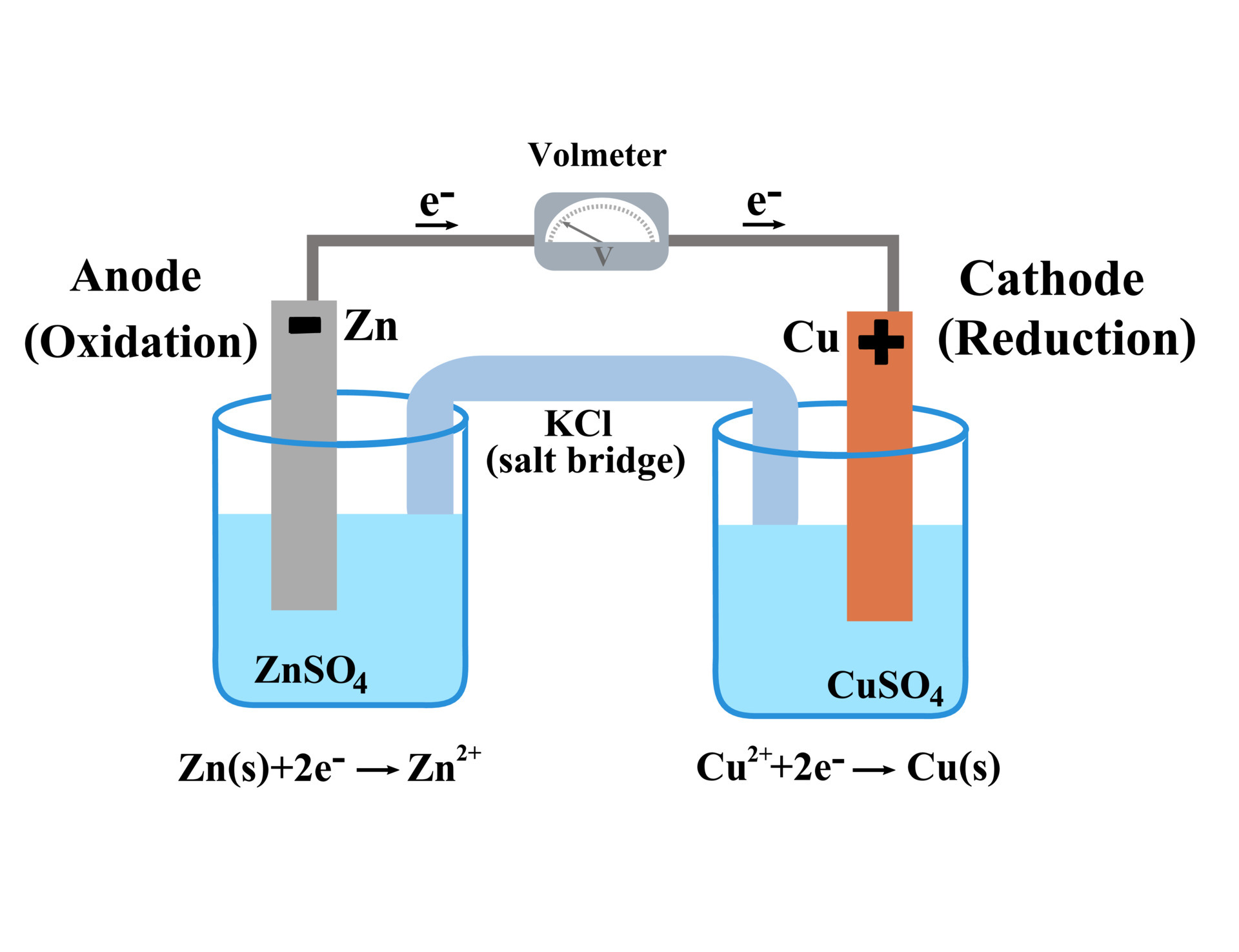
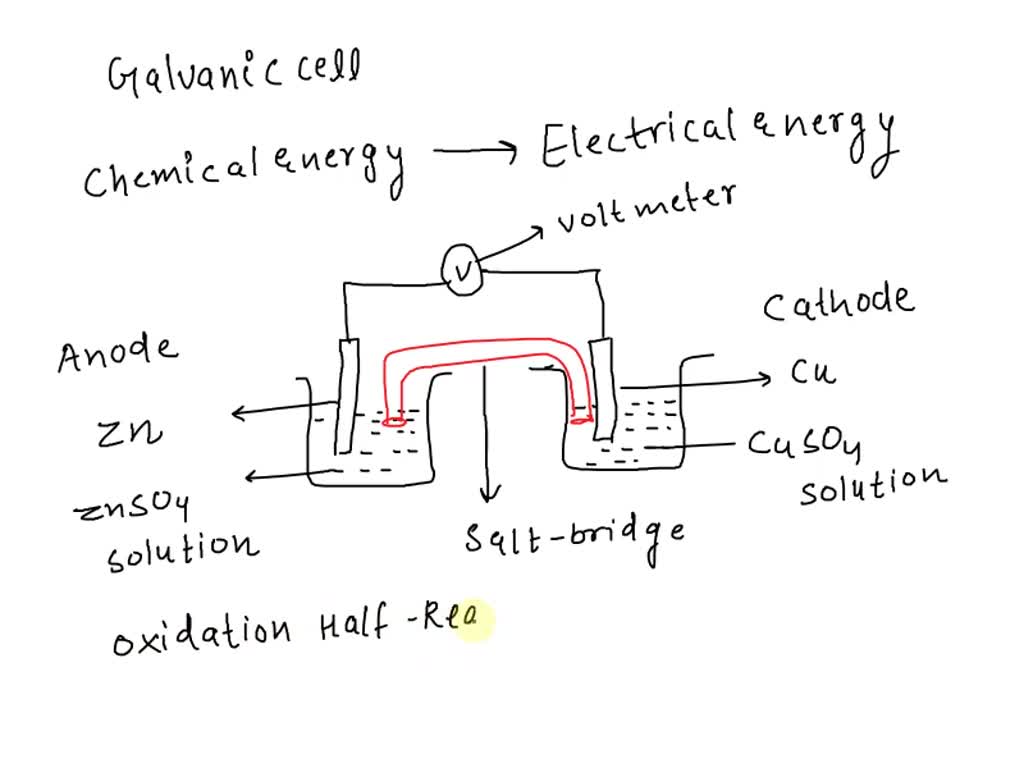
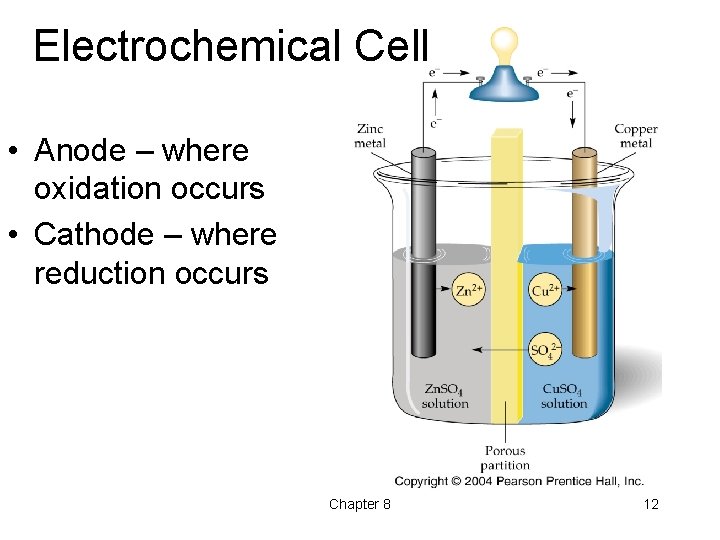
In a typical electrochemical cell, like a galvanic cell (the kind that produces electricity, like your battery), you'll have an anode. And guess what? The anode is where the magic of oxidation happens. It's the place where the electrons are being eagerly relinquished. It’s the electron-dumping ground. Think of it as the "giving station."
Imagine a piece of zinc metal sitting in a solution. Zinc is pretty happy to let go of its electrons. It’s like that guy who always volunteers to take on extra work, even when he’s already swamped. When zinc gets the chance in an electrochemical cell, it’ll happily shed two electrons: Zn → Zn2+ + 2e-. See those little "e-"? Those are the electrons making their grand exit, off to power something or be snatched up by another atom.
So, the anode is your primary suspect for oxidation. If you're looking at a diagram of an electrochemical cell and trying to figure out what’s going on, find the electrode (that's the solid bit where the action happens) that's labeled as the anode. That’s your oxidation hotspot.
Why is it called the anode, anyway? It’s a bit of a mouthful, I grant you. But it’s basically the electrode where oxidation occurs. And here’s a little mnemonic that might help: "An Ox". Anode = Oxidation. See? Easy peasy, lemon squeezy. It's not some mystical incantation; it's just a label for the spot where the electron-shedding is happening.

And just to be clear, this isn't about rust. While rust is also oxidation (iron losing electrons to oxygen), it's a slower, more spread-out affair. In an electrochemical cell, it's a more controlled and directed process. We're talking about a deliberate chemical reaction designed to create or utilize electrical energy. It’s like comparing a toddler’s crayon scribble to a Rembrandt. Both are art, but one is a bit more… intentional.
The Electric Highway: Connecting the Dots
Now, these electrons that are being so generously handed over at the anode don't just vanish into thin air. Oh no. They've got places to go and things to do. They embark on an epic journey through an external circuit – that's the wire connecting your anode to the other side of the cell. This flow of electrons is what we call electric current. It’s the invisible river of energy that powers your gadgets.
So, the anode is where the electrons start their adventure, and they’re heading somewhere else. This journey is crucial. Imagine you’re at a party, and you’ve just eaten a whole bag of chips. You’re feeling a bit… full of energy. You need to do something with it! You go dance, or tell an elaborate story. The electrons at the anode are kind of like you after those chips – they’ve got extra energy (electrons) and they’re off to do something with it.
On the other side of our electrochemical cell, we have the cathode. This is where the electrons get welcomed with open arms. It's the electron-grabbing station. And what happens at the cathode? You guessed it: reduction. Reduction is the opposite of oxidation; it's the gain of electrons. So, while the anode is saying "Here, take these!" the cathode is saying "Yes, please, I'll take 'em!"
The cathode is where another chemical species is eager to receive those electrons. Think of it as the needy recipient. It’s the one that’s a bit electron-deficient and is thrilled to get some extra electrons to become more stable. It’s like that friend who’s always asking to borrow your charger because theirs is always dying. The cathode is that friend.
The relationship between the anode and cathode is like a well-choreographed dance. The electrons are passed from the anode to the cathode, completing the circuit and allowing the chemical reaction to continue. Without the anode’s willingness to oxidize and the cathode’s eagerness to reduce, the whole system would just… sit there. Like a car without an engine, or a comedian without a punchline.
What About Those Ions? The Unsung Heroes
We’ve talked about electrons, but what about the atoms themselves? When an atom loses electrons, it often becomes an ion – it gets a positive charge because it now has more protons than electrons. For example, our zinc atom became Zn2+, a zinc ion. These ions are also busybodies in the electrochemical cell.
To keep the electrical charges balanced within each half-cell, there’s usually an electrolyte. This is a solution containing ions. These ions move around to balance the charges. It’s like a tiny traffic cop making sure everything flows smoothly. For instance, if the anode is losing positive zinc ions into the solution, negative ions in the electrolyte might move towards the anode to keep things neutral. And if the cathode is gaining electrons and forming new neutral atoms or negative ions, positive ions might move towards the cathode.

This movement of ions is just as important as the movement of electrons. It’s the internal plumbing of the electrochemical cell, ensuring that the whole process doesn't grind to a halt due to charge imbalances. Think of it as the supporting cast that makes the lead actors look good. You need those background dancers and stagehands for the show to go on!
Putting It All Together: The "Where" Question Answered
So, to definitively answer the question: "Identify the location of oxidation in an electrochemical cell," you’re looking for the anode. It's the electrode where atoms or molecules are losing electrons. It’s the electron donor, the "giving station," the site of the "Ox" in "An Ox."
When you’re faced with a diagram or a description of an electrochemical cell, ask yourself: “Which component is letting go of its electrons?” That’s your oxidation culprit, and its home is the anode. It's the component that's getting "oxidized." It’s the one that’s becoming more positive or less negative in terms of its charge, because it’s lost those negatively charged electrons.
It’s a fundamental concept, but understanding it opens up a whole world of how batteries work, how we electroplate things (like giving your jewelry a shiny new coating), and even how our bodies generate energy. It’s all about these little electron transfers. So, the next time your phone battery is low, you can nod knowingly, thinking about the anode diligently doing its electron-losing duty, powering your world, one electron at a time. It’s less a mystery, more a well-oiled, electron-driven machine. And you, my friend, are now in on the secret!
