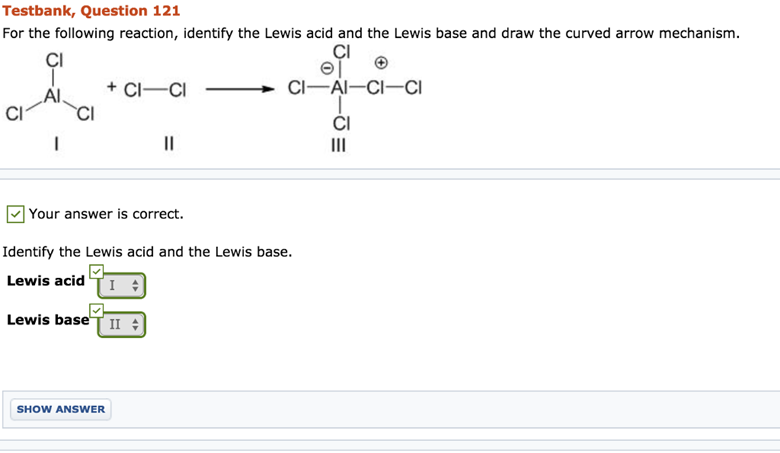
Identify The Lewis Acid In The Following Reaction
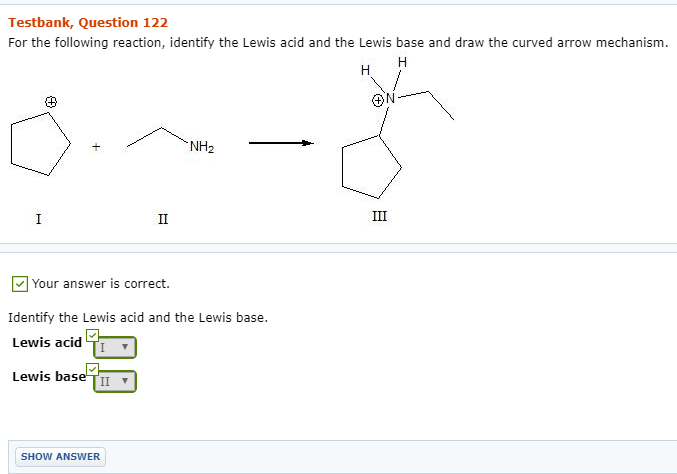
Imagine you're at a lively party, and everyone's trying to make friends. Some people are super popular and have tons of interesting stories to tell – they're the life of the party! Others are a bit shy, maybe looking for someone to share their thoughts with. In the world of tiny, invisible things called molecules, there's a similar kind of social dance happening all the time. Today, we're going to crash one of these molecular gatherings and try to spot our special guest, the Lewis Acid!
Think of molecules as tiny LEGO bricks that love to connect and build new things. Sometimes, to make a new connection, one molecule needs to lend a bit of itself to another. It's like sharing a toy or offering a helping hand. This is where our friend, the Lewis Acid, comes in. They're the molecule that's just itching to accept something from someone else. They're the ones who say, "Hey, you've got something I really need!"
Now, in our particular molecular party, we have a few characters. Let's meet them! We've got Boron trifluoride, a molecule that looks like a tiny, grumpy star with three sparkly arms. Each arm is waving around a very electronegative friend called Fluorine. Now, Fluorine is a bit of a drama queen; it loves to grab electrons, which are like the tiny, energetic snacks of the molecular world. Because the Fluorines are so busy hogging electrons, the central Boron atom is feeling a bit… neglected. It's like it's at the snack table, but everyone else has already grabbed all the good stuff, and poor Boron is left with an empty plate. It's got this big, gaping hole where some electrons should be. It's practically shouting, "Anyone got a spare electron I can borrow?"
On the other side of the room, we have Ammonia. Now, Ammonia is a bit of a different personality. It's shaped like a tiny pyramid with a fluffy cloud on top. That fluffy cloud? That's a pair of electrons that Ammonia is holding onto. It's like Ammonia is carrying a delicious, juicy treat and is looking for someone to share it with. It's friendly, generous, and ready to make a connection.
So, imagine Boron trifluoride is standing there, looking all forlorn with its electron-less center, and Ammonia strolls by, all proud of its electron pair. What do you think happens next? It’s a match made in molecular heaven! Ammonia sees Boron’s desperate need, and Boron practically winks at Ammonia's offered electrons. The electron pair from Ammonia is like the perfect missing piece for Boron’s empty spot. It’s so satisfying, like a jigsaw puzzle finally clicking into place!
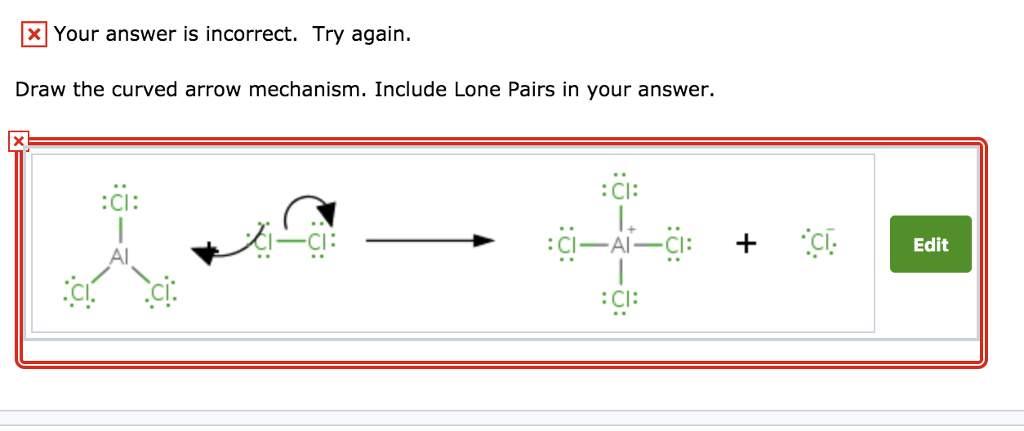
In this little molecular love story, who do you think is the one doing the accepting? Who is the one with the "electron-seeking" gaze? It's our central character, the one with the missing snacks, the one with the hole in its electron shell – that's right, it's Boron trifluoride! It's the one that's receiving the electron pair. It’s the one that’s hungry for electrons. That’s its defining characteristic in this interaction.
Think of it this way: if you're really hungry, and someone offers you a delicious cookie, you're the one accepting the cookie. You're the one who needed it! In the same way, Boron trifluoride is the one accepting the electron pair, making it the Lewis Acid.
(Solved) - For the following reaction, identify the Lewis acid and the
The beauty of this is that it’s not about being "good" or "bad," but about a natural inclination. Some molecules are just born with a strong desire to accept electrons – they're electron magnets! These are our Lewis Acids. They're the takers, but in a way that helps create something new and exciting. They're the ones that initiate the bond by reaching out (or rather, by being open to receive) for those precious electrons. It’s a fundamental part of how molecules interact and form the complex world around us.
So, the next time you see a chemical reaction, remember the party! Look for the molecule that seems a little bit empty, a little bit needy, a molecule that’s just waiting for a friendly electron donation. That’s your Lewis Acid, the star of the show in this electron-sharing dance. And the molecule offering the electrons? That’s a Lewis Base – a generous friend in this molecular world!