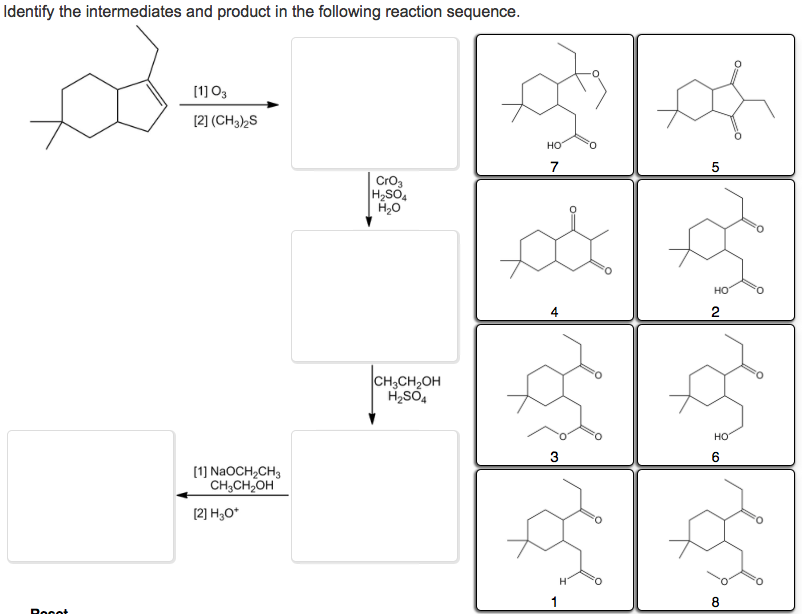
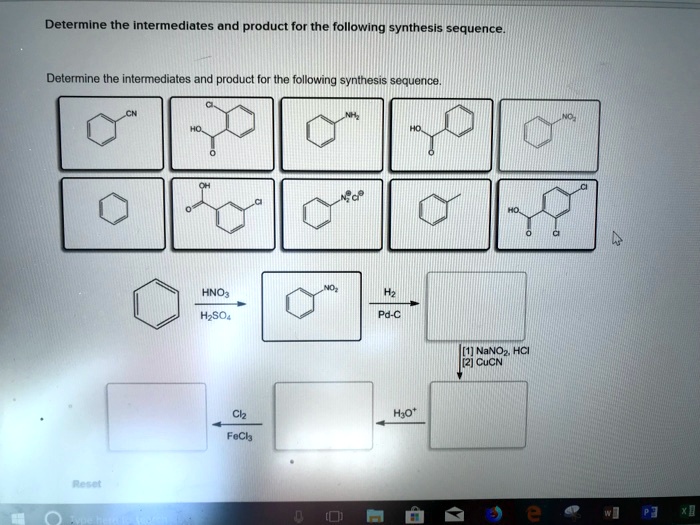
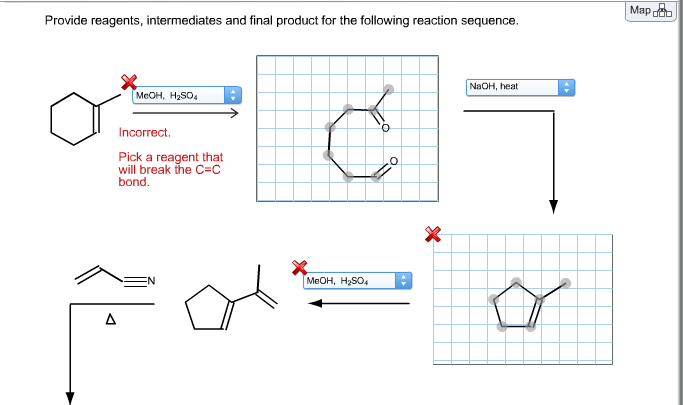
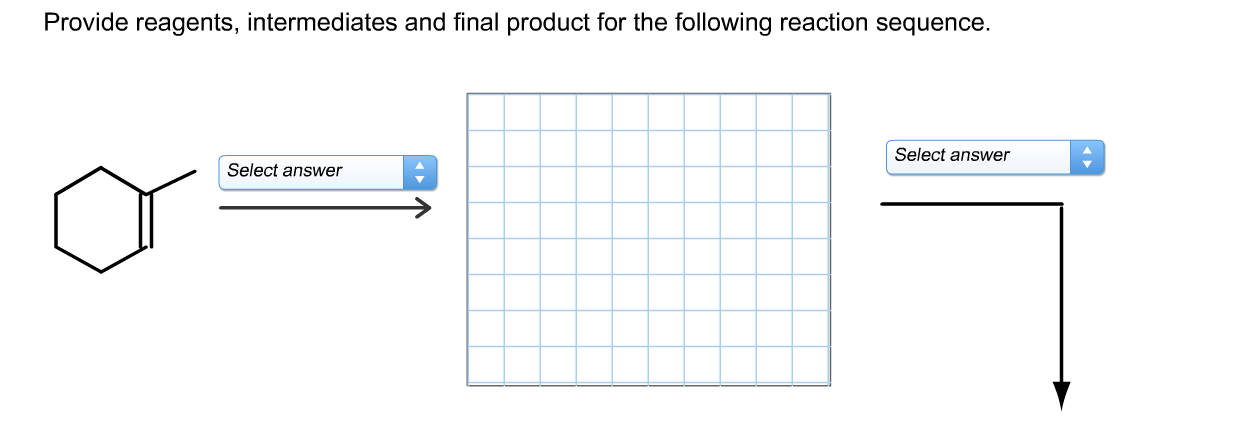
Identify The Intermediates And Product In The Following Reaction Sequence.
Hey there, fellow travelers on this wild and wonderful road of life! Ever feel like you’re just… going through the motions? Like you’re on a conveyor belt, and you’re not entirely sure where it’s taking you? Well, buckle up, buttercups, because today we’re diving into the fascinating world of chemical reactions, but not in the scary, textbook-induced-headache kind of way. Nope, we’re going to approach it like we’re trying to figure out the recipe for Grandma’s legendary chocolate chip cookies, or maybe even deciphering that cryptic instruction manual that came with your IKEA furniture. You know the one – the one that looks like a Rorschach test for engineers.
Think of a chemical reaction like a little journey. You start with some stuff (your reactants), you do some things to them (apply some energy, add a sprinkle of something else), and poof! You end up with something totally new (your product). It's like sending your favorite pair of sweatpants through the wash. You start with comfy, maybe a little stained, sweatpants. You add water, soap, and a spin cycle. And what do you get? Clean, fresh-smelling sweatpants. The sweatpants are still sweatpants, but they’re a better version. Or, you know, sometimes you get that mysterious shrunken sweater that now fits a hamster. That’s a different kind of reaction altogether, usually involving too much heat and a moment of sheer panic.
But sometimes, in this grand chemical adventure, there are little pit stops along the way. These are the intermediates. They’re like the characters in your favorite TV show who aren’t the main star, but they’re super important for the plot to unfold. You might not remember their names a week later, but without them, the hero wouldn't have gotten their coffee, or the villain wouldn't have been tripped by a rogue banana peel. Intermediates are the unsung heroes of the reaction world.
Let’s imagine you’re making a fancy layered cake. Your reactants are your flour, sugar, eggs, and butter. You mix them all up, maybe add some chocolate chips because, well, chocolate chips make everything better. This batter is your first step. But you don’t just eat raw cake batter, do you? (Don't lie, we've all been there). So, you bake it. The batter, as it bakes, transforms. It might go through stages where it’s gooey in the middle and slightly browned on the edges. That partially baked cake? That’s kind of like an intermediate. It’s not the final product, but it’s a necessary step to get there. And then, finally, you pull out that glorious, golden-brown, perfectly baked cake. That, my friends, is your product. The glorious, edible masterpiece.
Sometimes, these intermediates are so fleeting, so shy, that they’re like that one acquaintance you see at parties who always seems to be in the background, nodding along to conversations. You know they're there, contributing to the general vibe, but they don't really do much on their own. In chemistry, these intermediates can be super reactive and disappear almost as soon as they’re formed, zipping off to become the next thing in line.
Let’s talk about a specific kind of chemical journey. Imagine you have a super excited kid, let’s call him Timmy, who wants to build the ultimate Lego castle. His initial toys (the reactants) are a giant box of assorted Lego bricks. Timmy, being the ambitious architect he is, doesn't just slap all the bricks together. He first sorts them by color and size. That sorting process? That's like forming an intermediate. He's organizing the raw materials into something more manageable. Then, he starts building walls, then towers, then maybe a moat with a drawbridge. Each stage of construction, where he’s got a partial wall or a semi-built tower, is another intermediate. He's not finished yet, but he's definitely moved beyond just having a pile of bricks. Finally, after hours of intense concentration, strategic brick placement, and maybe a few existential Lego crises, he unveils his magnificent Lego fortress. That, my friends, is the product. The culmination of all his efforts, the magnificent castle.
The beauty of understanding these intermediates is that it helps us see the process. It's like watching a chef meticulously prepare each component of a gourmet meal. You might only see the final dish, but the chef knows about the perfectly seared scallops, the velvety smooth potato puree, and the delicate herb reduction. Each of those is an intermediate, a crucial step in creating the final, delicious product. Without them, you’d just have raw ingredients and a lot of confusion.

Sometimes, these intermediates are like those little sticky notes you put on your fridge with reminders. You don't live on the sticky note, but it helps you remember to pick up milk or call your mom. In a chemical reaction, an intermediate is a temporary step that’s essential for getting from point A (the reactants) to point B (the product). It’s like the middle child of the chemical world – often overlooked, but absolutely vital for the family’s overall stability and progress.
Let’s get a little more scientific, but in a fun, not-going-to-make-you-want-to-take-a-nap way. Consider the classic example of making water from its basic ingredients. You’ve got your hydrogen gas (H₂) and your oxygen gas (O₂). These are your starting reactants. Now, these two don’t just spontaneously decide to become water. You need to give them a little nudge, like a spark. When that spark happens, you get a highly reactive, super unstable little thing called a hydroxyl radical (•OH). This hydroxyl radical is an intermediate. It’s not hydrogen, it’s not oxygen, and it’s definitely not water. It’s like a temporary celebrity, famous for a split second before it gets right back to work.
This hydroxyl radical then bumps into another hydrogen molecule, and poof! You get another intermediate, this time it’s a hydrogen atom radical (•H) and a water molecule (H₂O) forming. But wait, we’re not quite done. That hydrogen atom radical is also super eager to react. It then grabs an oxygen atom from another O₂ molecule, and bam! You get your final, glorious product: another water molecule (H₂O)! See? You started with H₂ and O₂, had a couple of super-fast intermediates hanging around, and ended up with good ol’ H₂O. It’s like a chemical relay race, with each intermediate passing the baton to the next until the finish line.
Think about it like ordering a pizza. Your reactants are your craving for pizza and your wallet. You go to the pizza place (the reaction conditions). You order the pizza (the intermediate steps). First, they knead the dough, then they add the sauce, then the cheese, then the toppings. Each of those is a little step, an intermediate stage. You wouldn't consider raw dough with sauce on it the final product, would you? It's getting there, but it's not quite pizza. Finally, after it’s baked to golden perfection, you get your delicious, cheesy product. The pizza!
Sometimes, these intermediates are like those awkward teenage years. You’re not a kid anymore, but you’re not quite an adult. You’re in this transitional phase, figuring things out, and maybe a little bit clumsy. Chemical intermediates can be similar. They’re formed from the reactants but haven't quite reached the stable form of the product. They exist for a moment, a chemical blink of an eye, and then they transform into something else.
Let’s imagine you’re making a delicious sandwich. Your reactants are bread, cheese, and ham. You don't just shove them all in your mouth at once, right? You slice the bread, you unwrap the cheese, you lay out the ham. Those are your initial steps, preparing your ingredients. Then, you might put the cheese and ham between the bread slices. That assembled, but not-yet-toasted sandwich? That's an intermediate. It's a definite step towards the final product, but it's not the ultimate sandwich experience. Then, you might toast it, or grill it. That toasted, melty masterpiece? That’s your glorious product.
The key takeaway here is that even though intermediates might be fleeting or seem insignificant on their own, they are the essential stepping stones in a reaction. They are the bridge that connects what you start with to what you end up with. Without them, many of the amazing transformations we see in chemistry, and in everyday life, simply wouldn't happen.
So, the next time you’re trying to figure out a complex process, whether it’s assembling furniture, cooking a meal, or even just planning your day, remember the intermediates. They’re the unsung heroes, the behind-the-scenes magic-makers that get you from where you are to where you want to be. And that, my friends, is a beautiful thing. Cheers to the intermediates!
