Identify The Hybridization Of The N Atoms In N2h4

Imagine, if you will, a tiny dance party happening at the atomic level. We're not talking about a rave with flashing lights and loud music, but a much more intimate, a really quite charming, gathering of atoms. Today, we're going to peek in on a little molecule called hydrazine, which has the chemical formula N₂H₄. It’s like a molecular handshake between two nitrogen atoms, each holding onto a couple of hydrogen pals.
Now, these nitrogen atoms, they aren't just sitting around looking pretty. They're like busy bees, and they have to arrange themselves in a way that makes everyone in the molecule happy and stable. Think of it like a family getting ready for a group photo. Everyone needs to find their spot so they can all be seen and feel connected.
The way atoms connect is all about their electrons, those zippy little things that orbit the center of the atom. Electrons love to be in pairs, and they're the glue that holds molecules together. But sometimes, the way they naturally arrange themselves isn't quite ideal for forming the strongest, coziest bonds.
This is where a bit of atomic rearranging, or as the scientists call it, hybridization, comes into play. It’s like the nitrogen atom is tidying up its room, moving its electron "toys" around so they can be shared more effectively with its new molecule friends.
In our little N₂H₄ family, the two nitrogen atoms are the stars of the show. They need to be able to bond with each other and with the hydrogen atoms. To do this, each nitrogen atom basically gets its "room" ready for bonding.
Think of it like this: a nitrogen atom usually has its electrons in a few different "orbital apartments." Some are nice and round, like a cozy studio apartment, and others are a bit more shaped, like a dumbbell-shaped bachelor pad. These are called s and p orbitals.
But for making those strong, single bonds needed in hydrazine, the nitrogen atom decides to mix and match. It's like taking a little bit from the s orbital and a little bit from two of the p orbitals. It's a very efficient way to get organized!
When this mixing happens, the nitrogen atom creates three new, identical "rooms" for its electrons. These new rooms are called sp³ hybrid orbitals. They are all shaped similarly and are perfectly positioned to form strong, stable connections with other atoms.
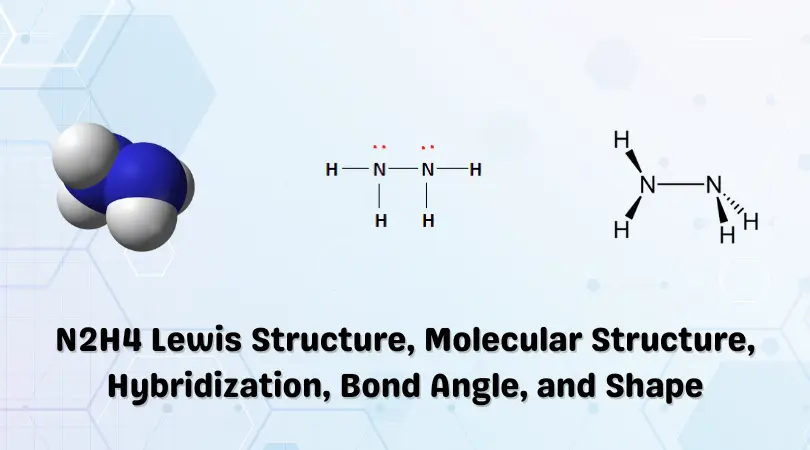
So, each nitrogen atom in N₂H₄ uses these sp³ hybrid orbitals. One of these special orbitals on each nitrogen atom reaches out and forms a strong, single bond with the other nitrogen atom. It’s like a firm handshake between the two nitrogen buddies.
Then, each nitrogen atom has two more of these sp³ orbitals left. And guess what? They use those to reach out and grab onto the hydrogen atoms. Each nitrogen atom forms two single bonds with two hydrogen atoms. So, each nitrogen is holding hands with the other nitrogen and two hydrogens.
It’s a beautifully efficient arrangement! This sp³ hybridization on both nitrogen atoms means that all the bonds are single, strong bonds, and the molecule has a nice, stable shape. It's like everyone in the family is holding hands securely, not letting go!

What’s really neat is that this mixing of orbitals isn't just for show. It directly influences the shape of the molecule. Because the sp³ orbitals are arranged in a specific way, pointing away from each other as much as possible (like trying to avoid bumping elbows in a crowded room), the N₂H₄ molecule ends up with a sort of bent or zig-zag shape.
It’s not a perfectly straight line, but more like a little chain where the nitrogen atoms are in the middle and the hydrogens are sticking out. This shape is crucial for how hydrazine behaves. It’s like the molecule’s posture, affecting how it interacts with other molecules.
And here’s a little heartwarming thought: this neat little atomic arrangement is what makes hydrazine so useful! It’s a building block for all sorts of things. It’s used in rocket fuel, believe it or not. Imagine these tiny, hybridized nitrogen atoms helping to launch rockets into space!
It’s also a key ingredient in making pharmaceuticals and other chemicals that improve our lives. So, the next time you see something manufactured, or even just think about space exploration, remember these little sp³ hybridized nitrogen atoms working together in N₂H₄.
It’s a testament to how even the smallest, most intricate arrangements at the atomic level can have such grand and important consequences. They’re not just atoms; they’re tiny engineers, constantly finding the best way to connect and build the world around us.
So, the answer to our little mystery is that the nitrogen atoms in N₂H₄ are sp³ hybridized. They've tidied up their electron rooms, creating those versatile sp³ orbitals to form strong bonds and give the molecule its characteristic shape. It’s a small detail, but it’s a big part of what makes hydrazine, and the world it helps create, so amazing.
It’s a bit like a well-coordinated dance troupe. Each dancer (atom) has practiced their moves (hybridized orbitals) so they can perform a beautiful and effective routine (molecule). And in the case of hydrazine, that routine is pretty powerful stuff!
Think about the sheer ingenuity. These atoms, without any conscious thought, find the optimal way to arrange themselves for stability and reactivity. It’s a silent symphony of bonding, orchestrated by the fundamental rules of chemistry.

And this sp³ hybridization is a common theme. You'll find it in many other molecules, like methane (CH₄), where a carbon atom is also sp³ hybridized. It's a fundamental way for atoms to get ready for life in a molecule.
So, next time you encounter the molecule N₂H₄, you can smile and think about those two nitrogen atoms, each with their perfectly arranged sp³ orbitals, diligently holding hands and forming this essential little compound.
It’s a reminder that even in the seemingly abstract world of chemistry, there are stories of connection, efficiency, and a surprising amount of elegance. The hybridization of nitrogen in hydrazine is just one tiny, but fascinating, chapter in that ongoing story.
It’s these fundamental, yet often overlooked, details that underpin so much of the technology and materials we rely on every day. The humble N₂H₄ molecule, with its sp³ hybridized nitrogen atoms, is a quiet hero in our modern world.
So, a bit of atomic tidiness leads to a world of possibilities. The nitrogen atoms in hydrazine, through their sp³ hybridization, are ready to bond, ready to react, and ready to contribute to something bigger than themselves. It’s a beautiful, microscopic teamwork.
