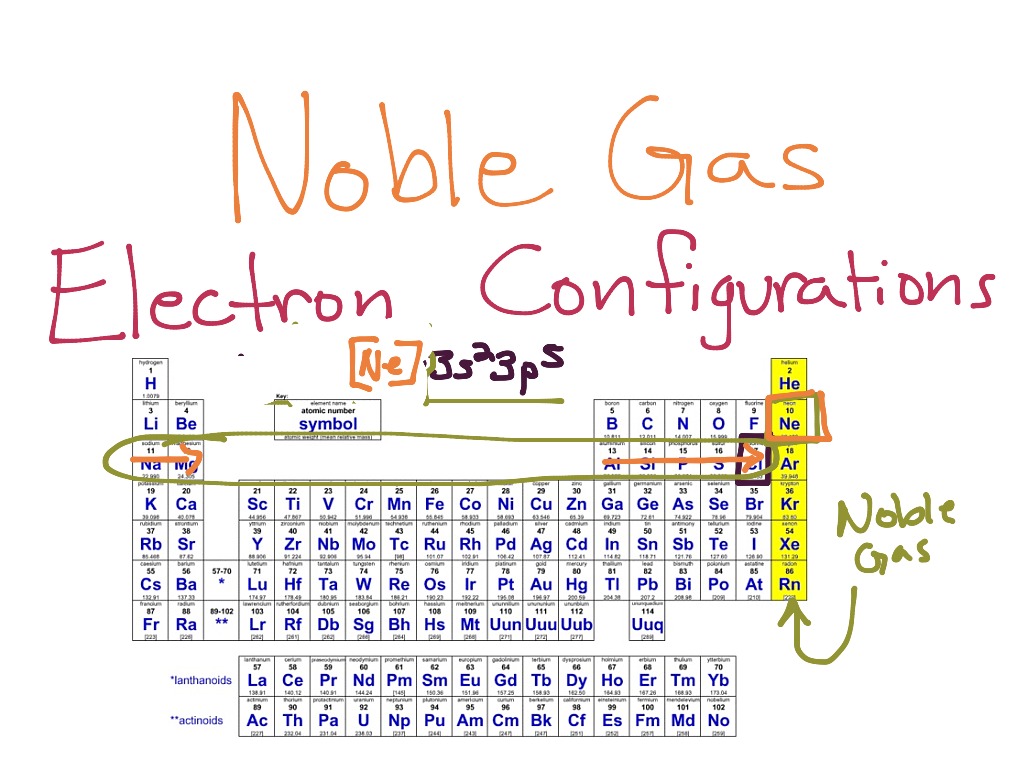
Identify The Generic Outer Electron Configuration For The Noble Gases
Let's talk about the cool kids of the periodic table. You know, the ones who always seem to have it all together. I'm talking about the Noble Gases. They're like the effortlessly popular group in high school, but for atoms. And they all share this one little secret. A secret about their outermost electrons. It's their superpower, really. And honestly, it's kind of hilarious when you think about it.
So, what’s the deal with these awesome atoms? Why are they so chill? Why don't they ever seem to need anyone else? It all comes down to their outer electron shells. Think of electron shells as tiny apartments around the atom's nucleus. Each apartment has a certain number of rooms, and electrons are like the tenants. They like to fill up these rooms, especially the ones on the outer edge, which we call the valence shell. These are the electrons that do all the interacting, the ones that make atoms want to team up and form molecules. But not the Noble Gases. Oh no.
The Noble Gases have a really, really full outer shell. Like, stuffed full. They’ve got all the rooms occupied. They’ve got all the tenants in their seats. They’re all comfy and cozy. And because they’re so complete, they just don’t see the need to do much. They’re perfectly content. They’re the atom equivalent of someone who’s just finished a huge, delicious meal and is now just sitting back, relaxing, and watching the world go by. No need for snacks, no need for company, just pure, unadulterated contentment.
This magical, complete outer shell has a specific, kind of fancy-sounding name. It’s called a full valence shell. And for these particular elements, this full shell is their ultimate badge of honor. It’s what makes them so unreactive. They’ve already achieved their goals. They’ve reached peak atomic happiness. And honestly, who can blame them? If I had achieved such a state of perfect electron arrangement, I’d probably be pretty smug about it too.
So, what does this full outer shell actually look like, in generic terms? Well, it's pretty consistent. The very outermost energy level, that top-floor apartment, is completely filled. Now, the number of electrons needed to fill this outermost shell depends on which energy level we're talking about. But for the Noble Gases, that highest level is always topped out. Imagine each shell being like a set of nesting dolls. The Noble Gases have filled up the outermost doll, and there’s no more space to put anything else inside.

For most of the Noble Gases, this means they have a total of eight electrons in their outer shell. Think of it as a full house. Eight is the magic number for stability, a truly enviable position. It’s like hitting the jackpot in the electron world. This configuration, with eight electrons on the outside, is the key to their legendary unresponsiveness. They’ve got their eight, and they’re like, “Peace out, everyone else! I’m good.”
There’s one little exception to this “eight is great” rule, of course. Because life, and chemistry, would be too simple otherwise. The smallest Noble Gas, Helium, is a bit of a rebel. It only has two electrons in its outermost shell, and for Helium, that's enough! Its first and only shell can only hold two electrons, and it’s happily filled. So, Helium has a full shell with just two electrons. It’s like the precocious child who masters everything super early and then just cruises. Still unreactive, still in its own little happy bubble, but with a slightly different number.
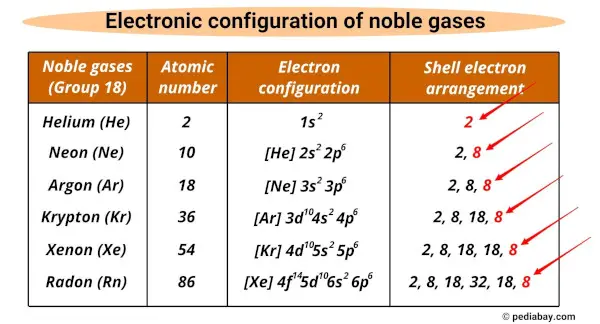
But generally speaking, when we talk about the generic outer electron configuration for the Noble Gases, we're talking about that beautiful, stable, and incredibly desirable setup where the outermost shell has eight electrons. It's like they've all agreed on a secret handshake: "Got my eight? Great. Let's do nothing together." This makes them really useful in situations where you don't want any chemical reactions happening. Think of those fancy light bulbs or the protective atmospheres used in welding. They need something inert, something that won’t muck things up. And guess who they call? The Noble Gases, of course!
It’s just fascinating how a simple arrangement of electrons can make such a huge difference. These elements are so unique because they're so self-sufficient. They don't need to gain, lose, or share electrons to feel complete. They are complete. They’ve reached the atomic equivalent of enlightenment. And while the rest of the periodic table is busy forming bonds, making friends, and generally getting into all sorts of chemical shenanigans, the Noble Gases are just… there. Being noble. Being unreactive. Being, dare I say, a little bit smug. And you know what? I kind of respect it. Their outer electron configuration is their ultimate power move, and it's a pretty sweet deal for them.
So, the next time you see a Noble Gas, give it a nod. It's living its best, most stable, and electron-filled life. And its generic outer electron configuration is the reason why. It's all about that full outer shell, that perfect eight (or two, for Helium). It's the atom's ultimate comfort zone.
This completeness, this lack of desire to interact, is what defines them. They’re the ultimate introverts of the atomic world. They’ve got their perfect setup, and they’re not about to complicate it. And if we're being honest, it's a pretty enviable position. They've cracked the code to atomic chill. The generic outer electron configuration for these stellar elements is simply a reflection of their supreme self-satisfaction. They are the definition of "if it ain't broke, don't fix it," applied to the fundamental building blocks of the universe.
