Identify The Functional Groups In The Following Molecules
+present+in+the+following+molecules..jpg)
Hey there, science nerds and curious cats! Ever peeked at a molecule and thought, "Whoa, what's all this business?" Well, guess what? We're about to dive into the wacky world of functional groups. They're basically the superstars, the secret sauce, the little bits that make molecules do their thing. And trust me, it’s way more fun than it sounds. Think of them like tiny LEGO bricks that snap onto a bigger structure, totally changing its personality.
So, what are we even talking about? Imagine you’ve got a bunch of plain old carbon and hydrogen atoms chilling together. That's like a blank canvas. But then, BAM! A functional group shows up. It's like adding glitter, a funny hat, or maybe even a tiny, grumpy badger. It adds character and, more importantly, function. This is what makes water, well, watery, and why your favorite perfume smells like, well, your favorite perfume!
Let's get down to the nitty-gritty, shall we? We're going to be playing a fun game: "Spot the Functional Group!" It's like a molecular scavenger hunt, and the prize is bragging rights (and a slightly better understanding of the universe). Don't worry, we won't be pulling out textbooks. This is more like a casual chat over coffee, but with more atoms.
The Usual Suspects: Meet the Main Players!
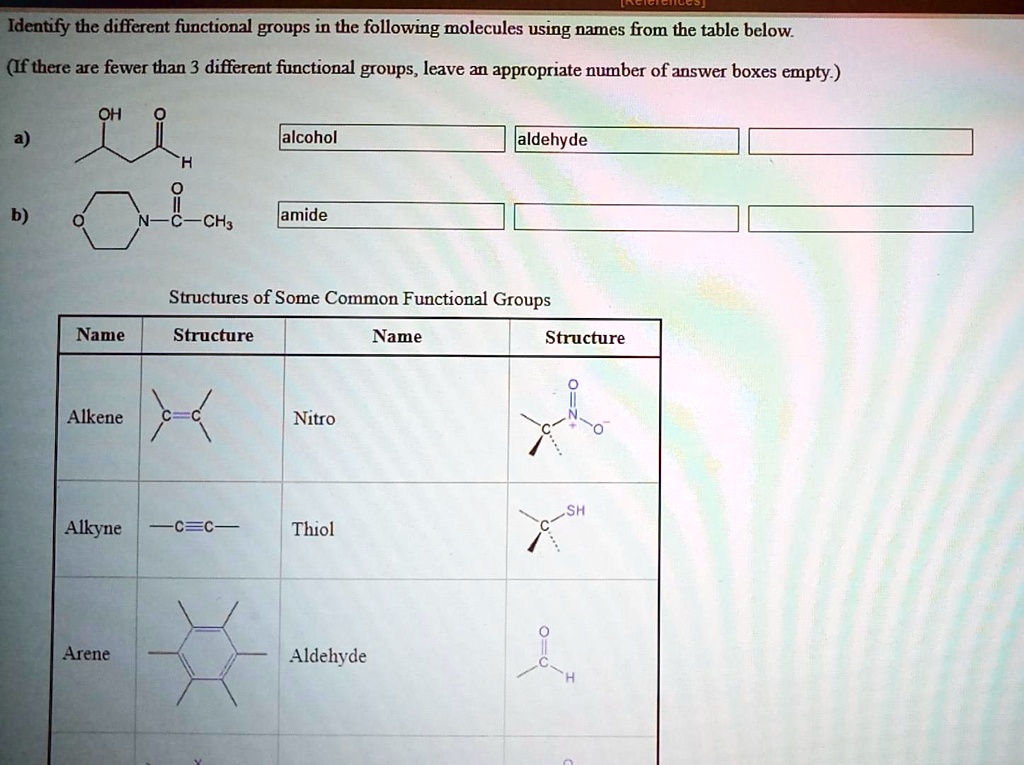
Alright, first up, the simplest of the simple. Meet the hydroxyl group. It’s just an oxygen atom clinging to a hydrogen atom. Like, O-H. So basic, yet SO important. Think of it as the friendly handshake of the molecule world.
Where do you find this little guy? Everywhere! It’s a key ingredient in alcohols, like the ethanol in your celebratory beverage (responsibly, of course!). It's also the reason why water (H-OH) is so darn good at dissolving things. It’s like the molecule’s social butterfly, attracting other molecules like crazy.
Fun fact: The word "alcohol" comes from the Arabic word "al-kuhl," which originally referred to a fine powder used as eye makeup. Who knew your drinking habit had ancient beauty roots? Mind. Blown.
Next, let's talk about the carbonyl group. This one's a bit more dramatic. It’s a carbon atom double-bonded to an oxygen atom. C=O. It’s like a tiny, enthusiastic wink from the molecule.
Now, the carbonyl group has two main personalities, depending on what else is attached. If it's hanging out with two other carbon chains, we call it a ketone. Think of acetone, the stuff that takes off your nail polish. That’s a ketone saying "Hello, I’m here to make things disappear!"
If the carbonyl carbon is also attached to a hydrogen atom, it's an aldehyde. These guys are even more reactive! They're often found in scents, like the vanilla-y goodness of vanillin. So, when you smell something sweet, chances are an aldehyde is waving hello.
Quirky detail: Some aldehydes are incredibly pungent in high concentrations but smell delightful at low concentrations. It’s like a Jekyll and Hyde situation for your nose!

Moving on to the carboxyl group. This is like the carbonyl group’s slightly more sophisticated cousin. It’s a carbonyl group and a hydroxyl group all smooshed together. So, you’ve got C=O and an O-H attached to that same carbon. This little powerhouse is what makes things acids.
Yep, we're talking about the acids you learned about in chemistry class, but in a more organic, friendly way. Acetic acid, the stuff in vinegar? That’s got a carboxyl group. It's the sour tang that makes salad dressing zing.
Funny thought: Imagine molecules having little name tags. The carboxyl group’s tag would say: "Warning: May cause sourness and corrosion. Also, makes things taste interesting!"
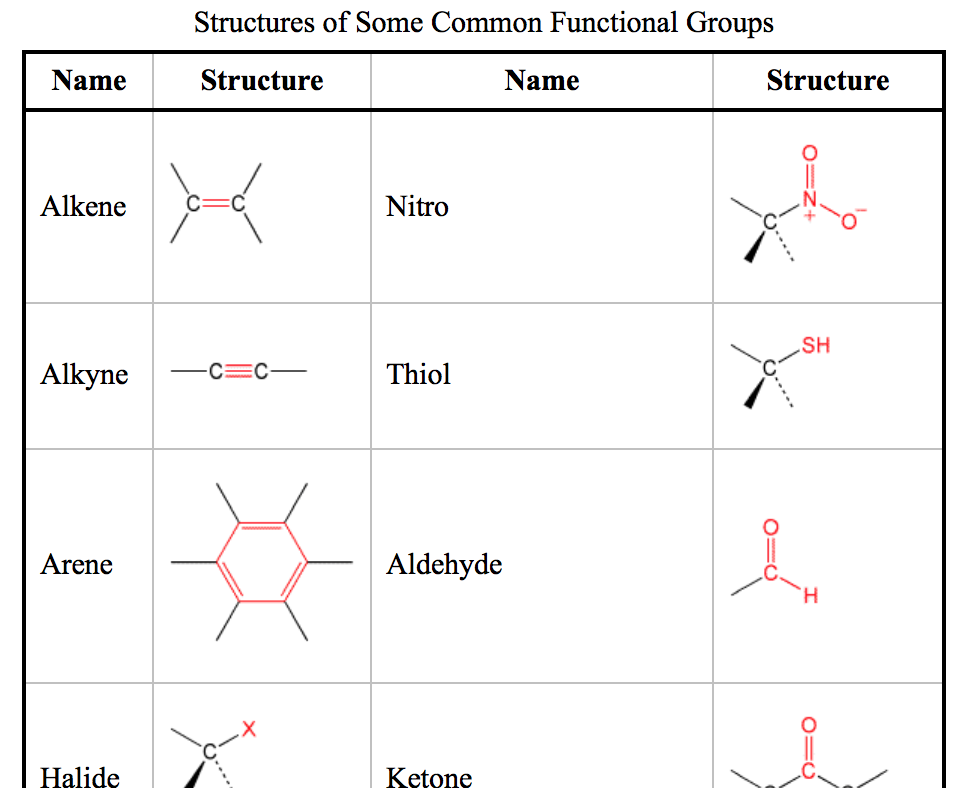
Then there's the amino group. This is where things get a bit more… nitrogen-y. It's a nitrogen atom bonded to two hydrogen atoms. N-H₂. Think of it as the molecule’s friendly wave or its way of saying "Let's form a bond, shall we?"
Amino groups are the building blocks of amino acids, which are the fundamental units of proteins. So, basically, every muscle, enzyme, and a good chunk of you is thanks to these little amino groups doing their thing. Pretty crucial, right?
Quirky fact: Amino groups can act as bases, meaning they can accept a proton. They’re like little molecular sponges, soaking up acidity.
Let’s not forget the ether group. This is where we have an oxygen atom sitting between two carbon atoms. C-O-C. It's like a cozy little bridge.

Ethers are often used as solvents because they're pretty stable. They’re like the reliable friend who’s always there, not causing too much drama. Diethyl ether, for example, was historically used as an anesthetic. Imagine a molecule that just gently lulls you to sleep!
Playful aside: The ether group is like the quiet observer in the molecular party, just watching things happen.
And finally, for our main cast, we have the ester group. This is like a hybrid of a carboxyl group and an ether. It’s a carbon double-bonded to an oxygen, and then another oxygen is attached to that same carbon, which is then attached to another carbon chain. R-COO-R'.
Esters are the rockstars of the scent world. They’re responsible for a ton of the fruity smells you love. Isopentyl acetate? That’s the smell of bananas! Ethyl butyrate? That’s the smell of apples! So, next time you bite into a juicy fruit, give a little nod to the ester groups doing their aromatic magic.
Funny detail: Some esters smell good in small amounts but can be quite… intense in large quantities. It's like a fine perfume versus a whole bottle.
The Molecule Detective Game: Put Your Skills to the Test!
Okay, enough introductions! Time to put on your detective hats. Let’s look at some hypothetical molecules and see if we can unmask their functional groups. Imagine we have a molecule that looks like this:
Molecule A: CH₃-CH₂-OH
What’s that hanging off the end? That’s right, the O-H! It’s the hydroxyl group. This molecule is an alcohol. Simple, elegant, and ready to party (in a controlled scientific manner, of course).
Now, let’s try this one:
Molecule B: CH₃-C(=O)-CH₃
See that C=O in the middle? That’s our carbonyl group. And since it’s surrounded by carbons, this is a ketone. It’s the nail polish remover talking!
How about this one?
Molecule C: CH₃-COOH
Look at the end there: C=O with an O-H attached to the same carbon. That’s our acidic friend, the carboxyl group! This molecule is an acid. It’s ready to add a little zing to your day.
One more for the road!
Molecule D: CH₃-NH₂
That N-H₂ at the end? That’s the amino group. This is a basic building block of life, folks. It’s like the friendly starter for bigger, more complex structures.
Why Does This Even Matter? (Besides Being Super Fun)
You might be thinking, "Okay, I can spot these things. But so what?" Well, my friend, these functional groups are the keys to understanding how molecules behave. They determine everything from solubility and reactivity to smell and taste.
Think about it: The same basic carbon skeleton can have completely different properties depending on which functional groups are attached. It’s like dressing up the same person in a superhero costume, a clown suit, or a scientist's lab coat. They’re still the same person, but their role and interactions change drastically!
Understanding functional groups is also super important for chemists who are designing new drugs, materials, and even food additives. They’re essentially playing with these molecular LEGO bricks to build new things with specific properties. It's a giant, ongoing construction project of the universe, and functional groups are the blueprints and the tools!
So, the next time you see a chemical formula, don't just see a jumble of letters and numbers. See if you can spot the functional groups! They’re the little personalities, the quirky characters that make the molecular world so endlessly fascinating. It’s a fun game, and the more you play, the better you get. Happy molecule hunting!
