Identify The Functional Groups In Cinnamaldehyde
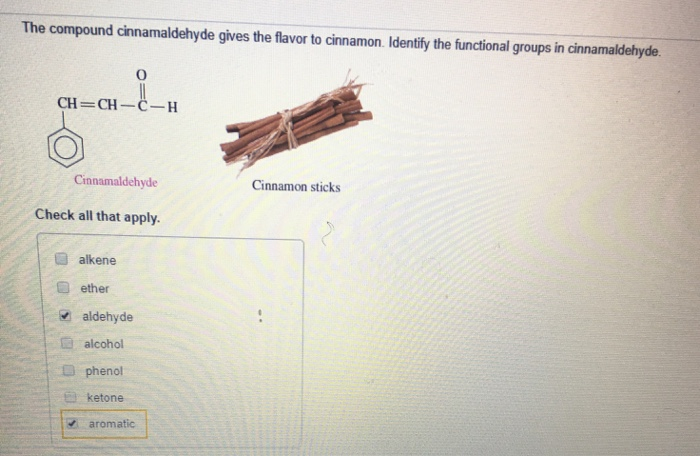
Ever sniffed cinnamon and felt that warm, cozy hug? That’s thanks to a superstar molecule called cinnamaldehyde. It's the main ingredient that gives cinnamon its signature scent and flavor. But what makes this little guy so special? It all comes down to its secret agents, known as functional groups. Think of them as the personality traits of the molecule, making it do all sorts of cool things.
Let’s dive into the delicious world of cinnamaldehyde and uncover its hidden talents. It’s not just about smelling nice, you know! These functional groups are the key to its magic.
First up, we have the aldehyde group. This is like the energetic leader of the group. It’s super reactive and loves to get involved in chemical reactions. You can spot it by its structure: a carbon atom double-bonded to an oxygen atom, and single-bonded to a hydrogen atom. In cinnamaldehyde, this aldehyde group is right at one end of the molecule. It’s responsible for a lot of the punchy flavor we associate with cinnamon. It’s like the sharp, vibrant note in a symphony of smells and tastes. Without this guy, cinnamon would be a lot more… bland. Imagine your favorite cinnamon roll without that zing! That's the aldehyde group working its magic.
Next, let’s talk about the aromatic ring. This is the sophisticated, elegant part of cinnamaldehyde. It’s a six-carbon ring with alternating double and single bonds, making it super stable and giving it a distinct aroma. Think of it as the stylish outfit the molecule wears. This ring is also called a phenyl group when it’s attached to something else, like it is here. It's what gives cinnamaldehyde its slightly woody, spicy undertones. It’s the foundation upon which all the other fun happens. This ring structure is super common in nature and is responsible for the smells of many other plants and flowers too. It’s like a universal scent-maker!
Now, here's where it gets really interesting. These two groups, the aldehyde and the aromatic ring, are connected by a special link. It’s a chain with two carbon atoms, and one of these carbons has a double bond with the other carbon. This is called an alkene group, specifically a carbon-carbon double bond. This double bond is like a little spring, full of energy and ready to react. It's this connection that bridges the gap between the aromatic ring and the aldehyde. It's not just a simple connector; it's an active participant! It allows the molecule to twist and turn in specific ways, affecting how it interacts with our senses and other molecules. This part of the molecule is also responsible for its rigidity, giving it a specific shape that’s important for how it fits into our taste and smell receptors.
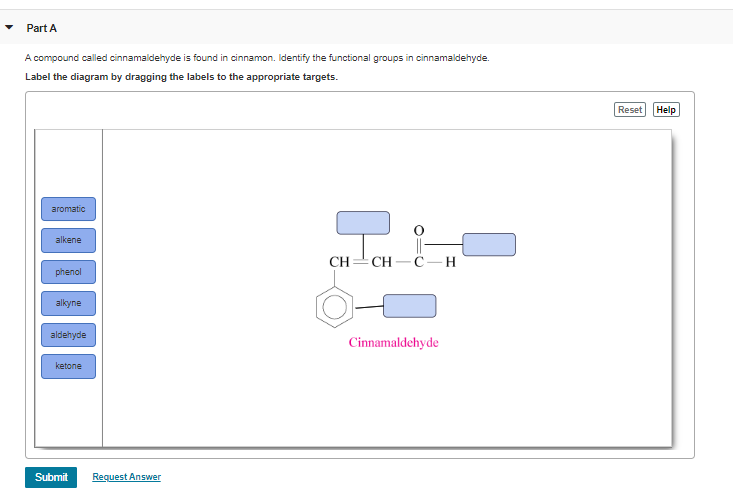
So, let’s put it all together. We have the zesty aldehyde group, the classy aromatic ring, and the springy alkene group all working in harmony. It's like a tiny chemical band playing a delicious tune! The aldehyde brings the zing, the aromatic ring brings the depth, and the alkene group provides the connection and a touch of flexibility. It's this perfect combination that makes cinnamaldehyde so captivating.
Why is this so entertaining? Because these simple structures create something we all know and love! It’s like understanding the secret recipe for happiness in a spice. When you sprinkle cinnamon on your oatmeal, or bake a gingerbread cookie, you're not just adding flavor; you're interacting with these fascinating functional groups. They’re the unsung heroes behind that comforting aroma that fills your kitchen.

What makes cinnamaldehyde so special is its versatility. Beyond just being a flavor enhancer, these functional groups allow it to be used in other ways. For example, the reactive aldehyde group can be modified to create other interesting chemicals. The aromatic ring can also be altered, leading to different scent profiles or properties. It’s a molecule with many potential pathways! It’s like a chameleon, able to adapt and change its role.
Understanding these functional groups is like having a secret decoder ring for the world of smells and flavors.
Imagine the aromatic ring as the core of a wheel, and the alkene and aldehyde groups as the spokes and the rim, all contributing to the wheel's ability to roll smoothly and powerfully. Each part plays a crucial role, and if you take one away, the whole thing changes. Cinnamaldehyde wouldn't be cinnamaldehyde without its aldehyde, its aromatic ring, and that connecting alkene. They are inextricably linked, like best friends who do everything together.
So, the next time you enjoy the warm, spicy notes of cinnamon, take a moment to appreciate the chemistry behind it. You’re experiencing the delightful dance of the aldehyde group, the sophisticated charm of the aromatic ring, and the lively connection of the alkene group. It’s a testament to how simple molecular structures can create such profound sensory experiences. It’s a little bit of science that makes life a whole lot tastier and more fragrant. It’s truly a molecule worth celebrating!
