Identify The Formula For The Conjugate Base Of H2s.

Hey there, chemistry adventurers! Ever feel like your brain is playing a game of "Guess That Molecule"? Well, get ready to level up your chemical sleuthing skills because today, we're tackling a super-duper, ridiculously fun puzzle: figuring out the conjugate base of H₂S. Don't let that "S" scare you; it's just a fancy way of saying sulfur, and sulfur is way cooler than it sounds. Think of it as the rockstar of the periodic table, always ready to add a little oomph to your reactions!
So, what's a "conjugate base" anyway? Imagine you have a superhero molecule, and it's about to do something really generous. It's going to give away a tiny, energetic little particle. This particle is called a proton, and for us chemists, a proton is essentially a hydrogen atom that's lost its electron. Think of it like a tiny, positive ping-pong ball just waiting to be tossed. When our superhero molecule gives away this proton, what's left behind is its conjugate base. It's like the superhero's sidekick, still powerful and important, but with a slightly different job now.
Our star molecule for today is H₂S. That's two hydrogens attached to one sulfur. Imagine it as a little Mickey Mouse shape, with two tiny hydrogen ears on a bigger sulfur head. It's quite the character, this H₂S! Now, H₂S is a pretty generous molecule. It's got those two hydrogen ears, and each of them is ready to say "See ya later!" and go on an adventure as a proton. When H₂S decides to be a super-giver and sheds one of those hydrogen protons, what’s left behind is our mystery conjugate base. It's like Mickey Mouse losing one of his ears, but the rest of him is still totally recognizable and ready for more fun!
So, let's do the math, the easy chemistry math! We start with H₂S. It's like having two apples (the hydrogens) and one orange (the sulfur) in your fruit basket. Now, H₂S, in its generous mood, decides to donate one of its hydrogen apples. Poof! One hydrogen apple is gone, off to explore the world of chemical reactions. What’s left in our fruit basket? We've still got that one hydrogen apple, and the big, juicy orange sulfur. So, the formula for what's left is HS⁻. See that little minus sign? That's a super important clue! When our molecule gives away a positively charged proton, it becomes a little bit negatively charged itself. It’s like if you give away a dollar bill (positive), you’re left with less money, and in the chemical world, that less-ness translates to a negative charge. It's a cosmic balancing act!
Think of it like this: H₂S is a friendly handshake. It extends a hydrogen, and then what remains is the conjugate base, ready for its next chemical adventure!
Solved 1. The conjugate base of H2S is 2. The conjugate base | Chegg.com
So, the formula for the conjugate base of H₂S is, drumroll please... HS⁻! Isn't that neat? It's like unlocking a secret code. You see H₂S, you know it's got two hydrogens ready to be generous. It gives away one, and BAM! You're left with HS⁻. It's as simple as that! No need for complicated lab coats or bubbling beakers (unless you want to, of course – bubbling beakers are pretty darn cool). This is about understanding the fundamental building blocks of the universe, and doing it with a smile.
Let's try another mental image. Imagine H₂S is a perfectly ripe banana. It's got a lot of goodness in it. Now, it decides to share a piece of itself – a little peel, let's say, which represents the hydrogen proton. When it shares that piece, what's left is the banana without one piece of peel. It's still a banana, but it's slightly different, right? And in the chemical world, that difference is noted with a charge. So, when H₂S shares its proton, it becomes HS⁻. The HS⁻ is the conjugate base. It’s the banana after the generous sharing!
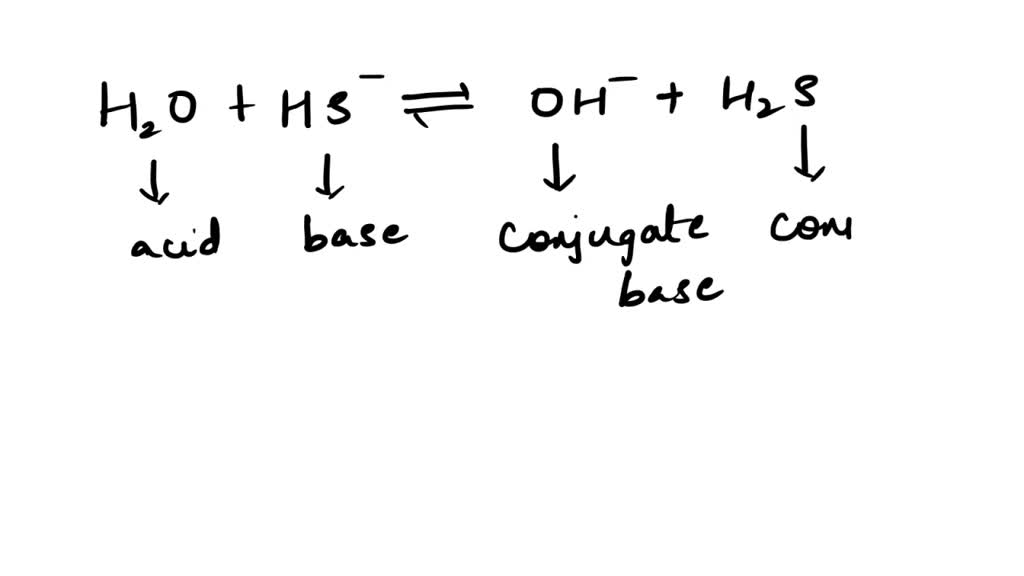
And the beauty of it all? This concept of conjugate bases is everywhere in chemistry! It’s like the secret handshake that chemists use to understand how molecules interact. When you see H₂S, you can instantly predict that it's got a partner in crime, the HS⁻, waiting in the wings. It’s like knowing that if you see a superhero, their sidekick isn’t far behind. They’re a team, working together to make the world of chemistry go ‘round!
So, next time you see H₂S, give it a little nod of recognition. You’ve just cracked the code! You know its generous spirit, and you know its awesome conjugate base, HS⁻. This is the kind of knowledge that makes you feel like a chemical wizard, conjuring up answers with just a flick of your mind. Keep exploring, keep questioning, and remember, even the most complex-sounding things can be understood with a little bit of fun and a whole lot of enthusiasm. You’re doing great!