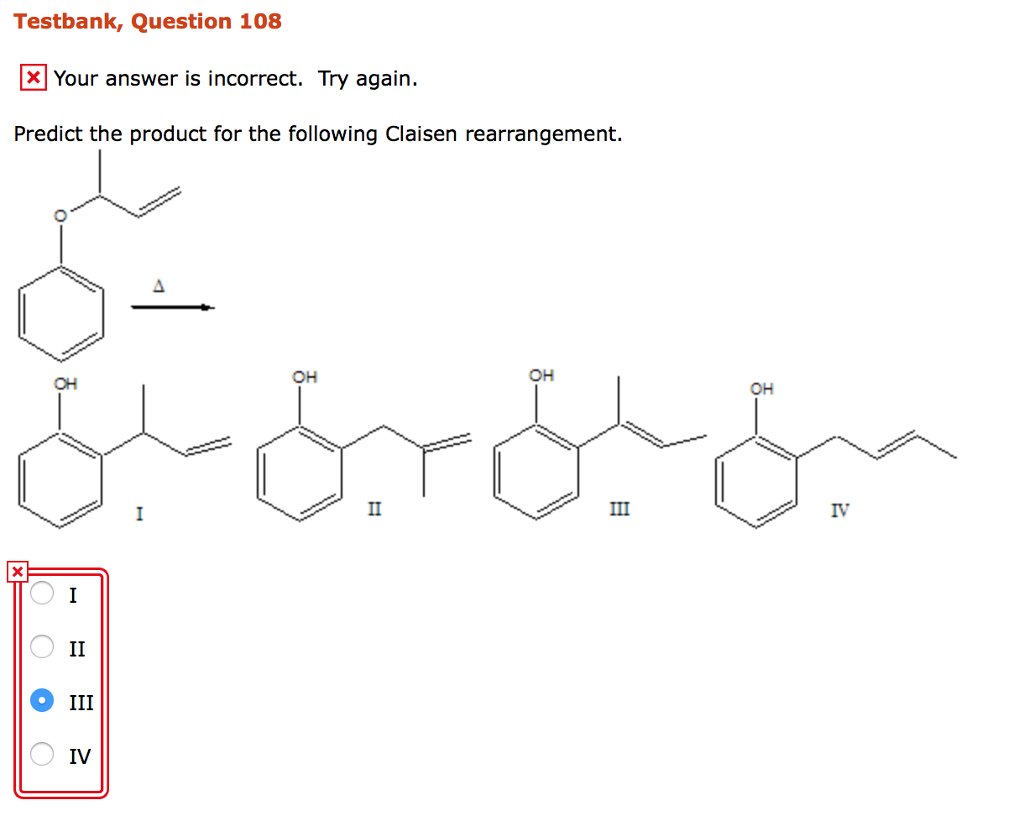
Identify The Expected Product Of The Following Claisen Rearrangement.
Hey there, coffee buddy! So, guess what we're diving into today? Get ready, because we're about to tackle a classic in organic chemistry: the Claisen Rearrangement. Yeah, I know, sounds fancy, right? But honestly, it's kinda like a magic trick, but way more useful and, dare I say, a little bit cool. We're gonna figure out what product pops out after this whole rearrangement thing happens. You ready for this? Grab another sip, let's do this!
So, what exactly is this Claisen Rearrangement? Think of it as a molecular makeover. Imagine a molecule doing a little dance, rearranging its atoms to become something new and, usually, more interesting. It's a type of pericyclic reaction, which basically means it happens in one smooth, coordinated step. No messy intermediates, no weird side reactions (well, mostly!). It’s all very neat and tidy, which, let’s be honest, is pretty satisfying.
The most common type, the one we're probably gonna focus on, involves something called an allyl vinyl ether. Now, that might sound like a mouthful, but let's break it down. We’ve got an allyl group – that's just a three-carbon chain with a double bond at one end. Think of it like a little, bendy stick. And then we’ve got a vinyl ether. That's where we have an oxygen atom attached to a carbon that's part of a double bond. So, basically, our allyl group is hanging off an oxygen, which is then chilling on a double bond. See? Not so scary when you say it like that.
Now, the magic happens when you give this allyl vinyl ether a little nudge. Usually, this nudge comes in the form of heat. Yep, just like making a good stew, sometimes you just need to crank up the temperature. When that heat hits our allyl vinyl ether, the molecule gets excited. It starts to vibrate, it starts to groove, and then… BAM! A new carbon-carbon bond forms, and an old one breaks. It's like a molecular domino effect, but way more controlled.
So, what are we actually doing when we're identifying the expected product? We're basically playing detective. We're looking at the starting molecule, the reactant, and we're predicting where everything is going to end up after the rearrangement. It's all about understanding the pattern, the mechanism of the reaction. Once you get the hang of the pattern, it's like having a cheat sheet for all sorts of similar reactions.
Let's visualize this, shall we? Imagine our allyl vinyl ether. We have our double bond, let's call the carbons in it C1 and C2. Our oxygen is attached to C2. Then, we have our allyl group attached to the oxygen. The allyl group has three carbons: C3, C4, and C5. C3 is attached to the oxygen. C4 is the middle carbon of the allyl group. And C5 is the terminal carbon of the allyl group, with the double bond between C4 and C5 if we were talking about a different starting material, but for the classic Claisen, the allyl group is usually attached via its C3. So, oxygen is attached to C2, and C3 is attached to oxygen. Makes sense? Okay, good. This is where it gets interesting.
When the heat hits, a few things happen simultaneously. A new sigma bond forms between C1 of the original vinyl group and C3 of the allyl group. This is the big one, the new connection that changes everything. At the same time, the carbon-oxygen single bond between C2 and the oxygen breaks. And then, to keep things balanced, the pi bond within the original vinyl group (between C1 and C2) shifts and becomes a new pi bond between C2 and the oxygen atom. Woah! It's a whole musical chairs situation happening inside the molecule!
And what does this molecular dance give us in the end? We get a ketone or an aldehyde. More specifically, if we started with a simple allyl vinyl ether, we'll end up with a gamma, delta-unsaturated carbonyl compound. "Gamma, delta-unsaturated"? What's that? Don't worry, it's just a way of describing where the double bond is relative to the carbonyl group (the C=O part). The carbonyl carbon is our reference point. The carbon next to it is alpha, then beta, then gamma, and then delta. So, in our product, the double bond is sitting between the gamma and delta carbons. Pretty specific, right? It’s like giving directions in the molecular world.
So, to recap the product: the allyl part of the molecule gets incorporated into the main chain, and the oxygen that was originally part of the ether linkage ends up as part of a carbonyl group. The double bond that was initially in the allyl group ends up further down the chain. It’s a really elegant transformation, if you ask me. It takes something that looks a bit unassuming and turns it into a functional group that’s super useful for further reactions.
Let's try a concrete example, shall we? Imagine we have phenyl allyl ether. So, we’ve got our benzene ring (phenyl group), and attached to it is an oxygen, which is then attached to our allyl group. So, the structure is Ph-O-CH₂-CH=CH₂. When we heat this up, what do we expect? The phenyl group is kinda acting like our vinyl group here, with the oxygen attached. So, the oxygen is connected to the benzene ring's carbon. Let's call that carbon C1 of the "vinyl" part. Then we have the oxygen. Then our allyl group: CH₂ (C3) - CH (C4) = CH₂ (C5). Wait, I might have messed up the numbering slightly in my head earlier. Let's be super precise.
For the true Claisen, the starting material is usually an allyl aryl ether or an allyl alkyl ether. Let's stick with allyl aryl ether for now, like our phenyl allyl ether example. The aryl group (like phenyl) has a double bond character within its ring. So, if we consider the carbon of the phenyl ring directly attached to the oxygen as C1, then the adjacent carbons are C2, C3, and so on. The oxygen is attached to C1. The allyl group is attached to the oxygen. Allyl group: Cα-Cβ=Cγ. So, we have Aryl-O-CαH₂-CβH=CγH₂. Heat is applied!
The [3,3]-sigmatropic rearrangement happens. This is the fancy term for the specific way the bonds shift. C1 of the aryl ring forms a bond with Cα of the allyl group. The bond between O and Cα breaks. The bond between C1 and O breaks. And the double bond shifts. This is where it gets a little different for allyl aryl ethers compared to allyl vinyl ethers. The aromaticity of the phenyl ring is disrupted during the initial steps, and then it's restored as part of the rearrangement. It’s like the ring takes a deep breath, does its thing, and then exhales, re-aromatizing itself.
In the case of phenyl allyl ether, the rearrangement leads to a ortho-allylphenol. So, the allyl group ends up attached to the carbon next to the one where the oxygen was originally attached. And the oxygen is now part of a hydroxyl group (-OH), attached to that same carbon where the allyl group used to be in the ether linkage. So, the oxygen ends up on the original C1 position, forming the phenol OH, and the allyl group ends up on the adjacent ortho position. Ta-da! You get a phenol with an allyl group stuck on its side. Pretty neat, huh?
What if the ortho positions are blocked? Oh, that’s a fun twist! If both ortho positions on the phenyl ring are occupied (say, by methyl groups), the allyl group has nowhere to go for that initial ortho-attack. In this case, the rearrangement can still happen, but it takes a slightly different path. The allyl group can migrate to the para position (the position directly opposite the oxygen). This is known as the para-Claisen rearrangement. So, the allyl group attaches to the para carbon of the phenyl ring, and the oxygen remains attached to the original position. The product would be a para-allylphenol. The molecule still tries to find a way to rearrange, even if its first choice is unavailable. It’s like the molecule is determined to complete its transformation!
Let's consider another variation. What about cyclic allyl vinyl ethers? These can get really interesting because the rearrangement is now constrained by the ring structure. For example, a dihydrofuran derivative with an allyl ether attached. The rearrangement can lead to the formation of a new ring or the opening of the existing one, depending on the specific structure. It’s like folding origami – the way you fold it determines the final shape.
Now, you might be asking, "Why do we care about this?" Well, the Claisen rearrangement is a super powerful tool in synthesis. It allows chemists to create complex molecules with specific functional groups and carbon-carbon bonds in controlled ways. It's used to make all sorts of things, from natural products to pharmaceuticals. Think about it, being able to rearrange a molecule like that is like having a secret weapon in your chemistry arsenal. It's all about building blocks and making them do what you want them to do.
To identify the product, you really need to: 1. Identify the allyl group and the vinyl/aryl group. 2. Mentally number the atoms involved in the rearrangement. For a standard Claisen, it's a [3,3]-sigmatropic shift. So, you're looking at a six-atom system where bonds shift. 3. Trace the electron movement. Think of arrows showing where the electrons go. The double bond in the allyl group is key. The double bond in the vinyl/aryl system (or its equivalent) is also important. 4. Predict the new sigma bond formation and the bond breaking. 5. See where the oxygen ends up and what functional group it forms. Let’s do a quick mental walkthrough with a more general allyl vinyl ether: R¹-O-C¹H=C²HR² and then the allyl group attached to the oxygen: -C³H₂-C⁴H=C⁵H₂. Here, R¹ is whatever is attached to the vinyl double bond. The key is the connection between C¹ and C³. When heat is applied, C¹ bonds to C³. The C¹-O bond breaks. The O-C³ bond breaks. And the double bond shifts. So, you end up with a carbonyl group where the oxygen was, and the allyl part has moved. The C¹-C³ bond forms. The double bond that was between C⁴ and C⁵ moves to be between C² and R²'s substituent if it was part of the original vinyl double bond. But in the most basic form, R¹-O-CH=CH₂ + CH₂-CH=CH₂ --> CH₂=CH-CH₂-CH₂-CHO. No, that's not quite right. Let me re-draw that mentally.
Okay, let's stick to the classic: Allyl vinyl ether. Structure: CH₂=CH-O-CH₂-CH=CH₂. This is where things get a bit tricky because you have two double bonds! The Claisen rearrangement specifically targets the allyl group. So, we're looking at the CH₂=CH-O part as the vinyl ether, and the -CH₂-CH=CH₂ as the allyl group. No, that's backward for the classic Claisen. The classic starts with an allyl vinyl ether, meaning the allyl group is attached to the vinyl ether. So, it’s CH₂=CH-O-CH₂-CH=CH₂. Ah, but the rearrangement happens on the allyl part. So the allyl group itself is rearranged.
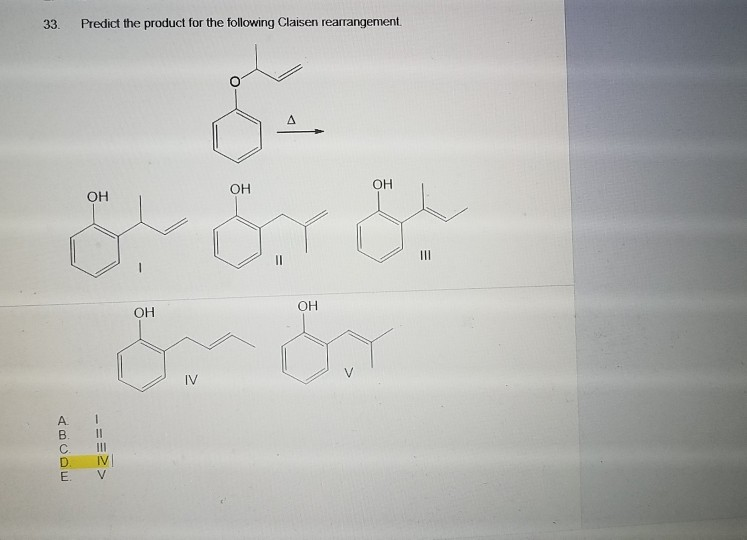
Let's be super clear. The reactant is an allyl vinyl ether. So, we have a vinyl group (C=C-O-) and an allyl group (-C=C-C) attached to the oxygen. For example: CH₂=CH-O-CH₂-CH=CH₂. The rearrangement happens via a [3,3]-sigmatropic shift. The electrons from the pi bond of the allyl group and the sigma bond between the oxygen and the allyl group, along with the pi bond of the vinyl group and the sigma bond between the vinyl carbon and the oxygen, all rearrange.
The starting material is like: A - O - B where A is the vinyl part and B is the allyl part. The rearrangement involves breaking the O-B sigma bond and the A pi bond, and forming a new C-C sigma bond between A and B, and shifting a pi bond. This results in a carbonyl compound.
Let’s draw it out (in our minds, of course!):
The structure:  (This is a generic representation, imagine the starting material is an allyl vinyl ether).
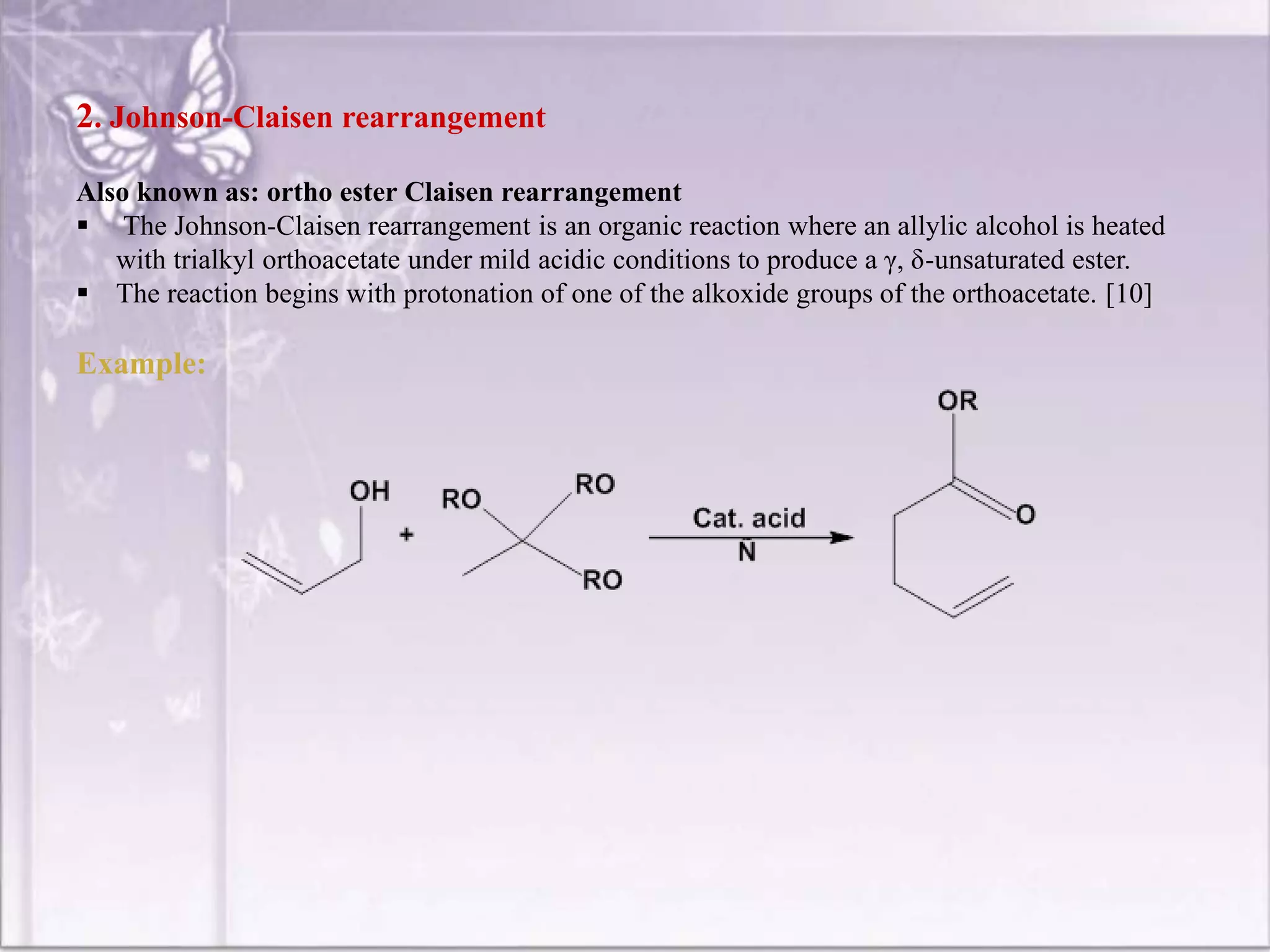
So, in a basic allyl vinyl ether (CH₂=CH-O-CH₂-CH=CH₂), the rearrangement leads to a γ,δ-unsaturated aldehyde. The double bond that was in the allyl group moves, and the original double bond of the vinyl ether becomes part of the carbonyl. The structure of the product will be: CH₂=CH-CH₂-CH₂-CHO. This is 4-pentenal. The allyl group has effectively migrated and the vinyl ether has become an aldehyde. The double bond is now at the gamma-delta position relative to the carbonyl. See how the molecule just shifted gears?
(This is a generic representation, imagine the starting material is an allyl vinyl ether).
So, in a basic allyl vinyl ether (CH₂=CH-O-CH₂-CH=CH₂), the rearrangement leads to a γ,δ-unsaturated aldehyde. The double bond that was in the allyl group moves, and the original double bond of the vinyl ether becomes part of the carbonyl. The structure of the product will be: CH₂=CH-CH₂-CH₂-CHO. This is 4-pentenal. The allyl group has effectively migrated and the vinyl ether has become an aldehyde. The double bond is now at the gamma-delta position relative to the carbonyl. See how the molecule just shifted gears?
So, when you’re faced with a Claisen rearrangement problem, don’t panic! Just look at where the allyl group is attached to the oxygen and the vinyl group. Then, imagine that concerted dance of electrons. Think about the formation of that new carbon-carbon bond and the creation of the carbonyl group. It’s all about following the electrons and the predictable pattern of this amazing reaction. You've got this! Now, pass the sugar, will ya?
