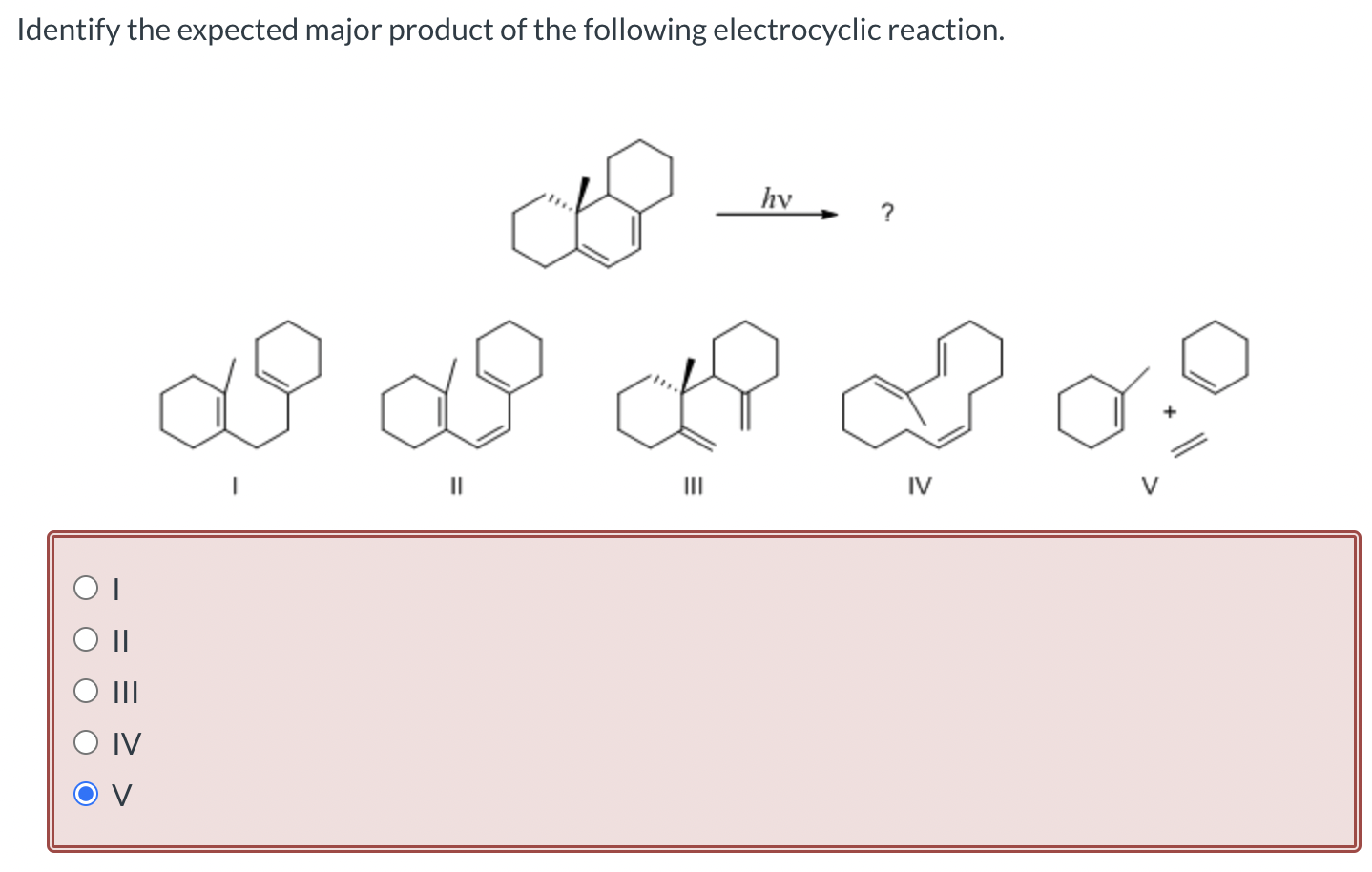
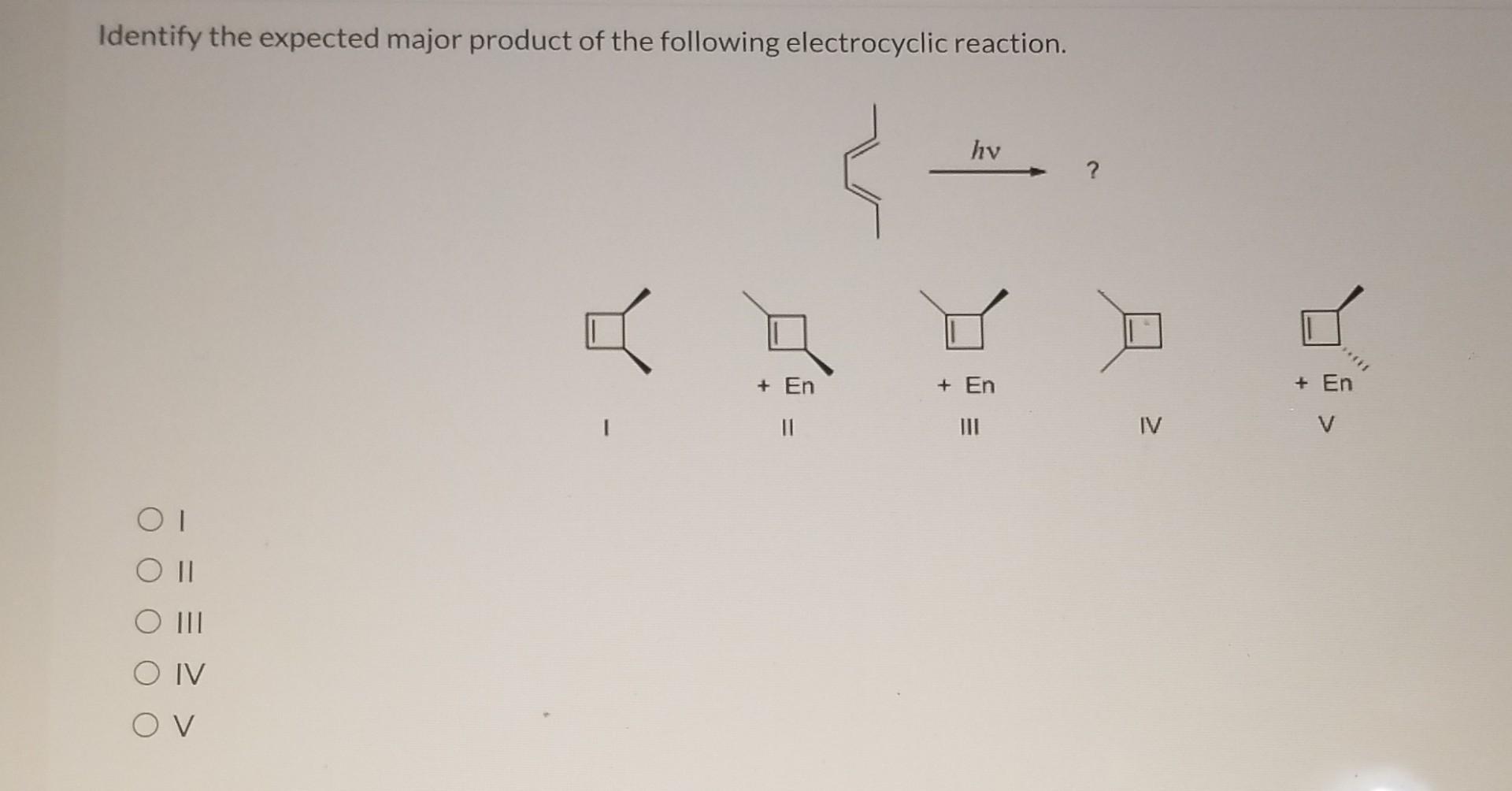
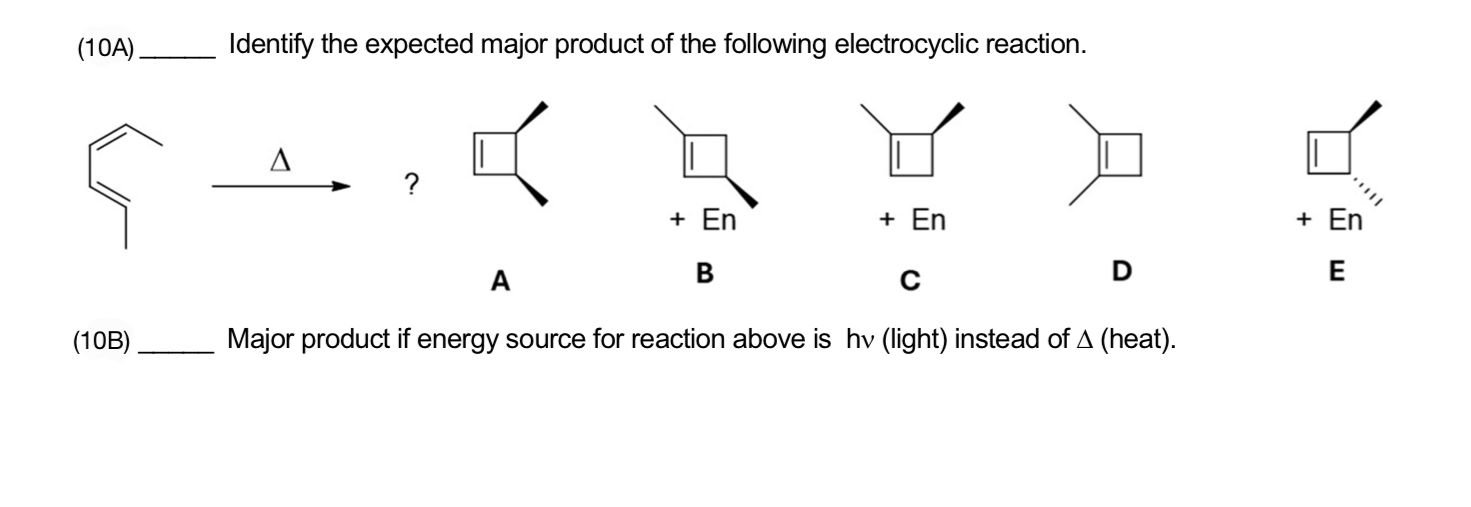
Identify The Expected Major Product Of The Following Electrocyclic Reaction.
Imagine you're a detective, but instead of solving crimes, you're solving molecular mysteries! Today, we're diving into the super cool world of electrocyclic reactions. Think of them as tiny, energetic dances that molecules perform. They're like a secret handshake for atoms, and figuring out who ends up with whom is the fun part.
Our mission, should we choose to accept it, is to peek at a specific chemical puzzle and guess what the final, most likely, major product will be. It's like predicting the winner of a molecular race. We're not just throwing darts; there are some clever rules that help us out.
This isn't your boring old textbook stuff, oh no. This is where chemistry gets exciting! It's like watching a magic trick unfold, but the magician is a bunch of atoms and the trick is rearranging themselves into something new. The predictability of it all, coupled with the surprise of the transformation, is what makes it so captivating.
So, what exactly are we looking at? Well, it's a specific setup. We have a molecule with some double bonds that are ready to tango. When the conditions are just right – usually a little heat or light – these double bonds decide to get together and form a new ring.
It's a bit like a group of friends holding hands. They start out in a line, maybe, and then they decide to form a circle. The way they arrange themselves to make that circle follows some very specific, and frankly, quite elegant, rules.
The stars of our show today are a type of reaction known as pericyclic reactions. Think of "peri" as meaning "around," and "cyclic" as meaning "a circle." So, these are reactions that happen in a circular fashion, with all the atoms involved moving together in a synchronized way.
Electrocyclic reactions are a special flavor of these pericyclic reactions. They involve the breaking and forming of sigma and pi bonds, all in one harmonious step. It's a chemical ballet, if you will. The atoms are the dancers, and the bonds are their flowing movements.
Now, let's get to the juicy part: predicting the product. This is where it gets really fun. We have to channel our inner chemist and think about how these bonds will rearrange themselves. There are principles at play that guide this molecular rearrangement.
The most important principle we use for electrocyclic reactions is called the Woodward-Hoffmann rules. Don't let the fancy name scare you! It's basically a set of guidelines that tell us whether a reaction will happen easily (meaning it's "symmetry-allowed") or if it's going to be a bit stubborn.
These rules look at the electrons involved in the double bonds. They're particularly interested in the symmetry of the atomic orbitals. Think of orbitals as the "neighborhoods" where electrons like to hang out.
For electrocyclic reactions, we have two main ways the atoms can move: conrotatory and disrotatory. Conrotatory is like both hands moving in the same direction to form the circle. Disrotatory is like one hand moving clockwise and the other counterclockwise.
The Woodward-Hoffmann rules tell us which of these movements is favored based on whether the reaction is happening with heat (thermal) or light (photochemical). It's like the molecule has different instructions depending on whether it's feeling warm or getting a tan!
For a typical electrocyclic reaction initiated by heat, the Woodward-Hoffmann rules tell us that a conrotatory movement is favored. This means the double bonds rotate in the same direction to form the new ring.
If, on the other hand, the reaction is triggered by light, then a disrotatory movement is usually preferred. This is where the electrons get excited by the light and decide to rearrange in a slightly different, but equally elegant, way.
So, let's imagine we have a molecule that's ripe for an electrocyclic reaction. We see those double bonds, just itching to connect. We also know if it's going to be a heat-powered dance or a light-show performance.
If it's heat-driven, we're predicting a conrotatory ring closure. This means the ends of our molecule will swing around in the same direction to meet and form that beautiful new ring structure.
The product we'll get is a cyclic molecule. The exact shape and where the remaining double bonds end up are determined by this conrotatory motion. It's like a well-choreographed dance that always leads to the same graceful finale.
The "major product" is simply the one that forms the most, the most likely outcome of this molecular choreography. It's the star dancer that everyone is watching.
Why is this so entertaining? Because it’s predictable, yet every molecule has its own unique twist. It's like having a puzzle where you know the general shape of the final picture, but the tiny details of how the pieces fit together are always a little bit surprising and beautiful.
It’s a glimpse into the fundamental forces that govern how matter is put together. These reactions are happening all around us, in nature and in the lab, creating all sorts of amazing compounds.
Think about it: a simple molecule, a little bit of energy, and poof! A new ring is formed. It’s a powerful demonstration of how subtle changes can lead to significant transformations.
The elegance of the Woodward-Hoffmann rules is what truly makes this special. They provide a theoretical framework to understand these seemingly complex rearrangements. It’s like having a secret decoder ring for molecular behavior.
This is the kind of chemistry that sparks curiosity. It makes you wonder what other molecular dances are happening, and how we can influence them to create new materials, medicines, or even flavors!
So, the next time you encounter a molecule with the potential for an electrocyclic reaction, don't just see a string of atoms. See a stage set for an incredible transformation. See a dance waiting to happen.
And when you're asked to predict the major product? You're not just guessing. You're using the elegant rules of chemistry to foresee the outcome of a molecular performance. You're becoming a molecular detective, and that's pretty darn cool.
It's about understanding the language of molecules. It's about appreciating the underlying order and beauty in the seemingly chaotic world of chemical reactions.
The journey from a linear molecule to a cyclic one, guided by the symmetry of electrons and the energy input, is a testament to the fascinating principles that govern our universe at the smallest scales.
So, keep your eyes peeled for those double bonds, consider the energy source, and let the Woodward-Hoffmann rules be your guide. The major product is waiting to be discovered, and the process of finding it is an adventure in itself.
It's a little bit of science magic, explained. And that, my friends, is what makes this kind of chemistry so absolutely entertaining and special.
