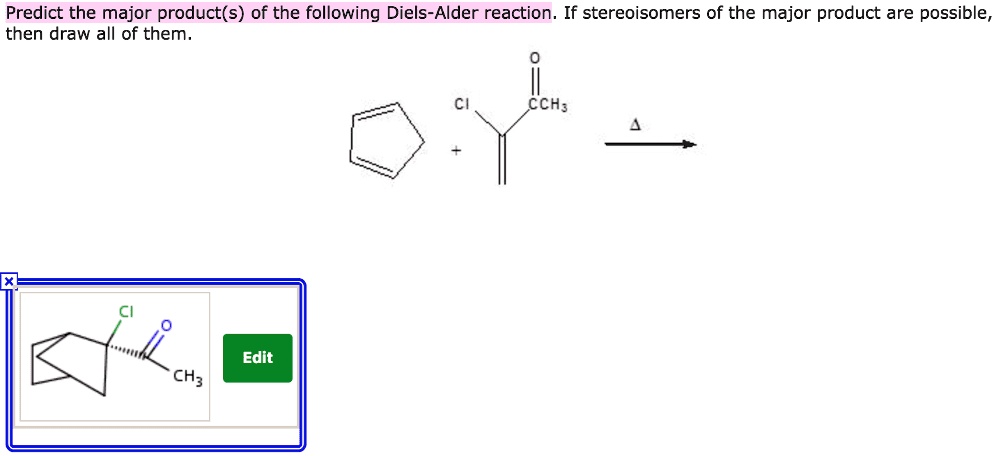
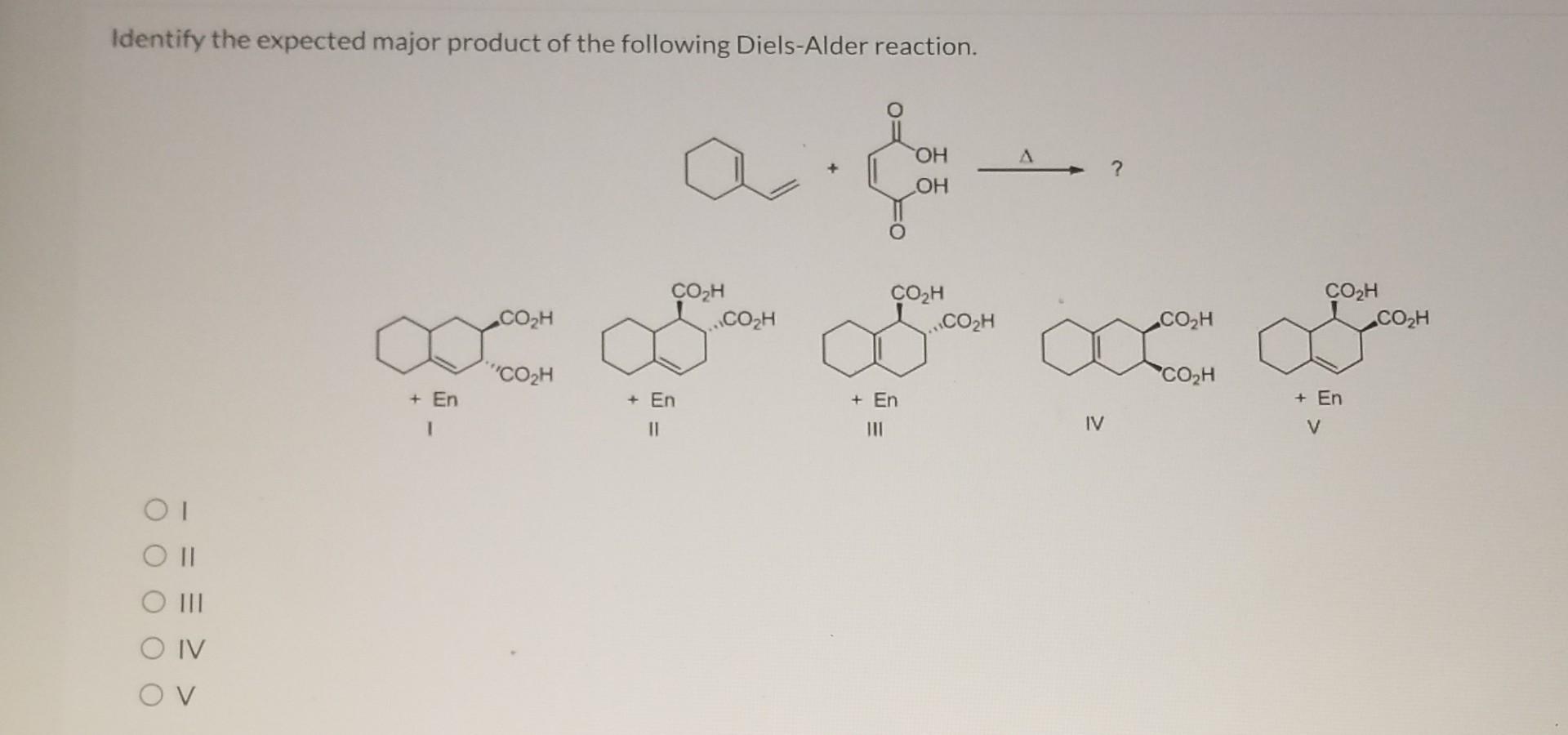
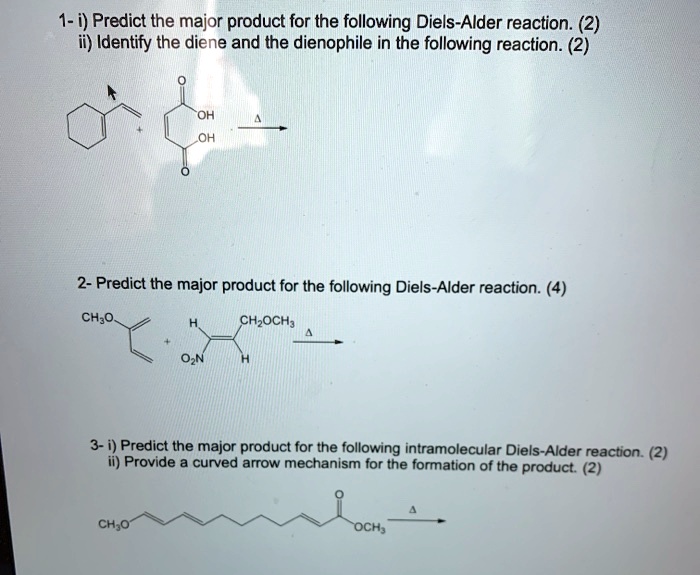
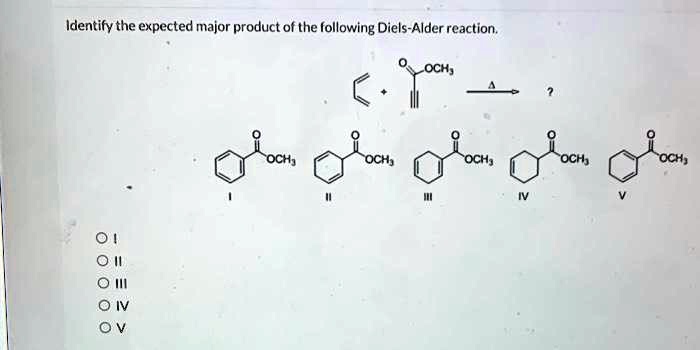
Identify The Expected Major Product Of The Following Diels-alder Reaction.

Alright, settle in, grab your latte, and let’s talk about something that might sound super nerdy but is actually way cooler than you think. We're diving into the wild world of chemical reactions, specifically the Diels-Alder reaction. Now, don't let the fancy name scare you. Think of it as a molecular party, a get-together where some very specific guests are invited to form something new and exciting. It’s like the ultimate blind date, but for atoms!
So, what’s the big deal? Well, the Diels-Alder reaction is how we chemists, and nature, for that matter, build these awesome ring-shaped molecules. They’re like the LEGO bricks of the organic world. And today, we've got a specific scenario. Imagine we've got two molecules, ready to mingle. On one side, we have our diene. Think of it as the enthusiastic, outgoing one, with its arms (or, you know, double bonds) wide open, just itching to connect. On the other side, we have our dienophile. This one’s a bit more focused, maybe a tad shy, but equally keen to form a bond. It’s got its own double bond, just waiting for the right partner.
Now, the magic happens when these two meet. It’s a beautiful dance of electrons, a coordinated effort where they basically hug it out and form a brand new six-membered ring. It’s so efficient, so clean, it’s like watching a perfectly choreographed ballet, but with atoms. And the best part? It usually happens with minimal fuss. No messy byproducts, just pure, unadulterated ring formation. It’s the chemical equivalent of ordering a pizza and it arriving perfectly cooked, exactly how you wanted it, with no anchovies!
So, let's get down to the nitty-gritty of our particular party. We've got our diene, which looks a bit like this: two double bonds separated by a single bond. It’s usually something like butadiene or cyclopentadiene. Picture it as a molecule that’s got plenty of flexibility to get cozy with its partner. It’s like that friend who’s always up for anything, ready to bend and shape to make things work.
And then we have our dienophile. This one’s got a double bond, often hooked up to something that makes it extra eager to react. Think of it as having a little “electron-pulling” superpower. It’s like the discerning guest at the party, looking for someone with the right kind of energy to match its own. Common dienophiles might have a cyano group (-CN) or a carbonyl group (C=O) attached. These groups are like little magnets for electrons, making the dienophile’s double bond super attractive to our diene.
Now, the actual reaction: it's a concerted reaction. This is a fancy way of saying it all happens in one go. No intermediate steps, no lingering hesitations. It’s like a lightning-fast handshake that instantly solidifies into a bond. The electrons from the diene’s double bonds and the dienophile’s double bond rearrange themselves, like a group of friends spontaneously deciding to hold hands and form a circle. This forms the new six-membered ring, and voilà! We have our product.
So, what does this ring look like?
This is where it gets interesting, because the geometry matters. Remember, molecules aren't just blobs; they have shapes! For the Diels-Alder reaction to happen smoothly, our diene needs to be in a specific conformation, called the s-cis conformation. Think of it as the diene getting into a little 'hug' position. If it’s in the s-trans position, it’s all stretched out and can’t get close enough to the dienophile for that intimate electron exchange. It's like trying to have a heart-to-heart conversation with someone across a football field – not ideal.
The dienophile, on the other hand, just needs its double bond. The more electron-deficient that double bond is (thanks to those electron-pulling groups we talked about), the faster and more enthusiastically it reacts. It's like finding someone who really wants to connect, not just someone who's passively available. The faster the reaction, the more fun the molecular party!
Now, let's look at the specific reactants we have. Imagine our diene is something like 1,3-butadiene. It's a classic, a real workhorse of the Diels-Alder world. And our dienophile is something like maleic anhydride. Now, maleic anhydride is a super popular dienophile. Why? Because that anhydride group (two carbonyls attached to an oxygen) is a big, fat electron-withdrawing group, making that double bond incredibly reactive. It's like the most popular dance partner at the molecular prom.
When 1,3-butadiene and maleic anhydride get together, they are like a match made in chemical heaven. The butadiene, in its s-cis form, wraps around the maleic anhydride. Their electrons start to flow, creating new bonds. It’s a beautiful symphony of electron rearrangement. The result? A new six-membered ring is formed, with the anhydride part of the maleic anhydride now attached to it.
And the major product? Drumroll, please…
It’s a molecule called norbornene-5,6-dicarboxylic anhydride. Fancy name, I know! But break it down: 'norbornene' is the basic bicyclic structure that gets formed. It's got a six-membered ring fused with a five-membered ring, creating a sort of bridge. And then you have the two carboxyl groups from the original maleic anhydride, attached to the bridge. They form the dicarboxylic anhydride part.
The thing about Diels-Alder reactions is that they can sometimes lead to different isomers, depending on how the dienophile attaches. There's the endo product and the exo product. Think of it like a hug: endo is when the dienophile tucks itself underneath the diene's bridge, while exo is when it sticks out on the other side. Now, here’s a surprising fact: in most Diels-Alder reactions, especially with electron-withdrawing groups on the dienophile, the endo product is kinetically favored. This means it forms faster. It's like the quickest way to get to the good stuff.
However, with maleic anhydride and butadiene, the exo product is often the thermodynamically favored product. This means it’s the more stable product in the long run. It’s like the quickest route gets you there first, but the slightly longer, more roundabout way leads to a much nicer, more permanent destination. So, while the endo might pop out first, given enough time and heat, the reaction might rearrange to give you the more stable exo product.
For this specific reaction, with typical conditions, you'll predominantly get the exo adduct. It's the more stable guy, the one that sticks around. So, when you see 1,3-butadiene and maleic anhydride having their little molecular rendezvous, the expected major product is that solid, stable exo-norbornene-5,6-dicarboxylic anhydride. It’s a perfectly formed, robust molecule, ready to be used in all sorts of chemical adventures. It's proof that sometimes, the most predictable outcome is also the most beautiful!
