Identify The Enolate Of Ethyl Diethoxyacetate.

So, you're staring at this molecule. It’s ethyl diethoxyacetate. Fancy name, right? Makes it sound like it’s going to a gala. But let’s be honest, most of us just want to know what’s happening under the hood. And when we talk about its enolate, things get a little… interesting.
Imagine this. We’ve got this molecule, ethyl diethoxyacetate, just chilling. It’s got its little ethyl group, its two ethoxy groups, all neat and tidy. But then, someone comes along with a strong base. Think of it as a super-enthusiastic friend who just loves to stir things up. This base is like, "Hey, what's going on over there?" and it pokes at a specific spot on our molecule.
The spot it pokes? It’s the alpha-carbon. That’s the carbon right next to the carbonyl group. You know, the C=O part. It’s like the social butterfly of the molecule, always buzzing with activity. And our strong base? It’s particularly interested in the hydrogen atoms attached to this alpha-carbon. These hydrogens are a bit more… adventurous. They’re a little less attached, a little more ready for a change.
Our base, with all its might, snatches one of these adventurous hydrogens. Poof! It’s gone. And what’s left behind? A charged-up situation. We’ve gone from a neutral molecule to something with a negative charge. This, my friends, is the birth of our enolate!
Now, this enolate of ethyl diethoxyacetate isn’t just a sad, broken molecule. Oh no. It’s a master of disguise. It’s got this cool trick called resonance. Think of it like a gymnast performing a flawless routine. The negative charge can do a little flip-flop. It can hang out on the alpha-carbon, making it nice and nucleophilic. Or, it can slide over to the oxygen of the carbonyl group. That oxygen, with its extra electron baggage, is also looking for something to do.

So, you have two main ways to draw this enolate. One where the negative charge is firmly planted on the carbon. This makes the carbon a bit of a party animal, ready to attack anything electrophilic that comes its way. It’s like a tiny, negatively charged magnet, just looking for something positive to latch onto. The other drawing shows the negative charge happily residing on the oxygen atom. This makes the oxygen a bit more involved, too. It’s like a quiet but powerful force, ready to influence things.
Why is this whole enolate thing so fascinating? Because it unlocks a whole new world of chemical reactions. Our ethyl diethoxyacetate, in its neutral form, is pretty polite. It doesn’t do much exciting. But its enolate? That’s a different story. It’s ready to rumble. It can participate in reactions like alkylations, where it gets to add new carbon chains. It can be involved in condensations, where it teams up with other molecules to build bigger, more complex structures.
It’s kind of like how a shy person at a party can become incredibly outgoing once they’ve had a little encouragement. The base is the encouragement. The enolate is the outgoing version. And the reactions? Those are the fun conversations and dancing that happen after the ice is broken.
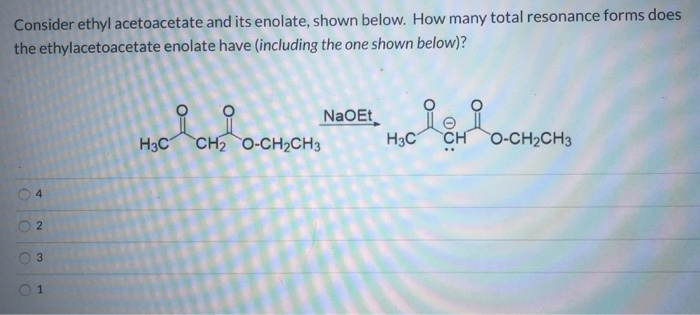
Now, with ethyl diethoxyacetate, things are a tiny bit more complicated than your average ester. Those two ethoxy groups on the adjacent carbon? They’re like little spectators, watching the show. They don’t usually get directly involved in the deprotonation at the alpha-carbon. They’re more like the audience members who are really into the drama, but not actively on stage.
So, when you’re asked to identify the enolate of ethyl diethoxyacetate, you’re essentially looking for that species where the alpha-hydrogen has been removed. You’re looking for the molecule that has this negative charge, ready to exhibit its resonance stabilization, and is now equipped with the power to be a super nucleophile. It’s the molecule that’s ready to break out of its shell and engage with the world of organic synthesis.
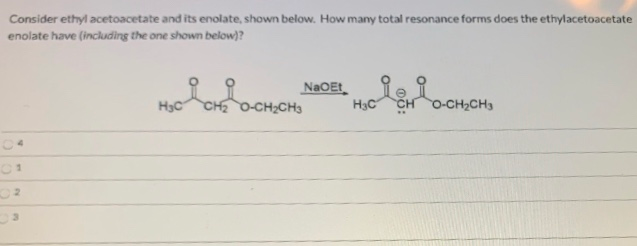
It’s not a particularly flashy molecule on its own, but its enolate form is where the magic happens. It’s the behind-the-scenes hero, the quiet power player. And honestly, sometimes the most interesting characters are the ones who don’t shout the loudest, but have the most impact. The enolate of ethyl diethoxyacetate? Definitely one of those characters.
So, next time you see ethyl diethoxyacetate, remember its hidden talent. Remember the enolate. It’s not just a molecule; it’s a state of potential, a chemical chameleon ready for its moment in the spotlight. And who doesn’t love a good comeback story, even if it’s a negatively charged one?
