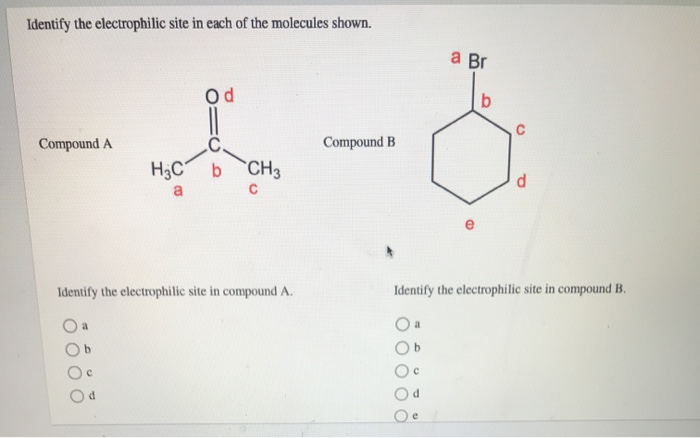
Identify The Electrophilic Site In The Molecule Shown
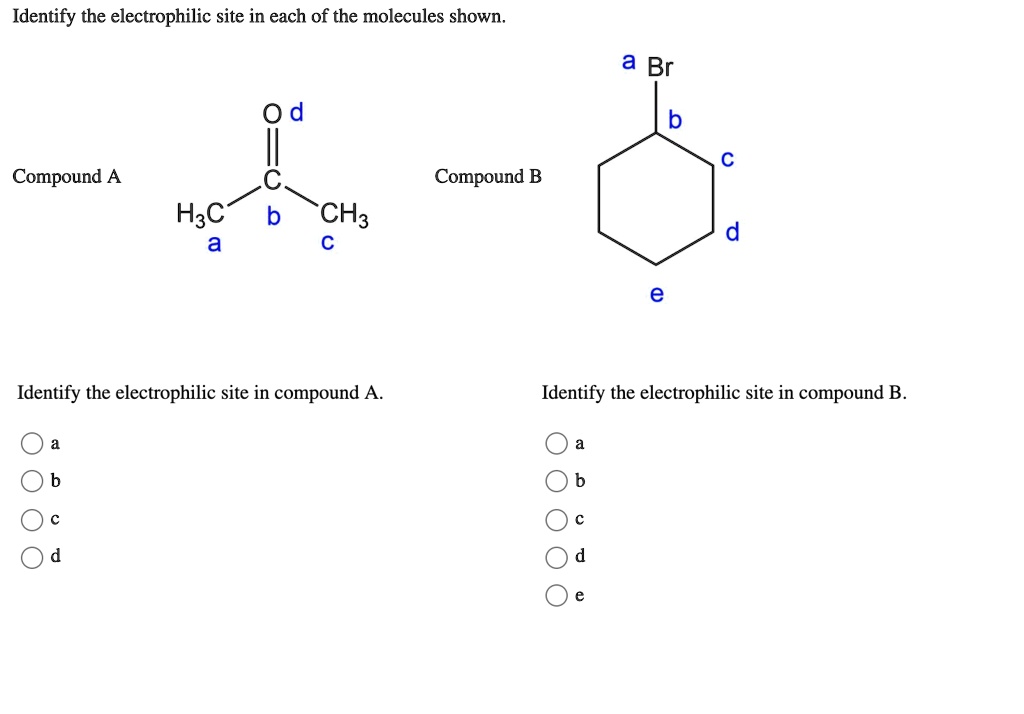
Alright, so imagine your molecule is a bustling little neighborhood, right? And in this neighborhood, we've got some residents who are a bit like the popular kids at school – they've got something others are really, really keen on. In chemistry speak, those "popular kids" are our electrophiles.
Think of it this way: an electrophile is basically a molecule (or a part of one) that's dying for some attention. It's a bit like that friend who's always checking their phone, desperately waiting for a text. They're hungry for something, and that "something" is usually a negatively charged particle, or a place with a whole lot of electrons. They're the ones with a bit of a "hole" in their electron wallet, and they're looking to fill it up, pronto.
It’s kind of like when you’re at a buffet, and there’s that one dish everyone’s raving about – the one with the extra crispy bits or the secret sauce. Everyone’s circling it, eager to snag a piece. That dish is our electrophile. It's the most tempting target, the one that’s most likely to get "attacked" by something else.
So, our mission, should we choose to accept it (and we definitely should, because it's kind of fun!), is to identify this "popular kid," this electron-starved character, within a given molecule. It's like playing detective, but instead of a magnifying glass, we've got our brains and a good understanding of how atoms like to play nice (or not so nice) with each other.
Let's break down what makes a molecule a prime candidate for being an electrophile. It's not usually a random thing. There are some tell-tale signs, like particular atoms or groups of atoms that tend to be a bit electron-poor. Think of it like looking for someone wearing a sign that says "Will Trade Happiness for Electrons."
One of the biggest giveaways is when an atom is bonded to something way more electronegative. Electronegativity, in case you’ve forgotten your high school chemistry or just conveniently blocked it out (totally understandable!), is basically an atom’s "clinginess" for electrons. Some atoms are like super-magnets for electrons, while others are more like those flimsy fridge magnets that barely hold on.
When a less electronegative atom is chained to a super-clingy one, it’s like that atom’s electrons are being yanked away. Imagine you’ve got a really strong friend, and you’re holding hands. If they suddenly yank their arm, you’re going to be pulled along, right? That’s kind of what’s happening with the electrons. They get pulled towards the more electronegative atom, leaving the other atom with a bit of a deficit. This electron-deficient atom is our budding electrophile.
A classic example of this is something like a halogen (like chlorine or bromine) bonded to a carbon. Oxygen is also a notorious electron-hogger. So, if you see a carbon atom attached to an oxygen atom, especially in certain arrangements, that carbon might be feeling a little light in the electron department. It’s like a kid who’s had their candy snatched by a bigger sibling – they’re looking for someone to share their toys with.
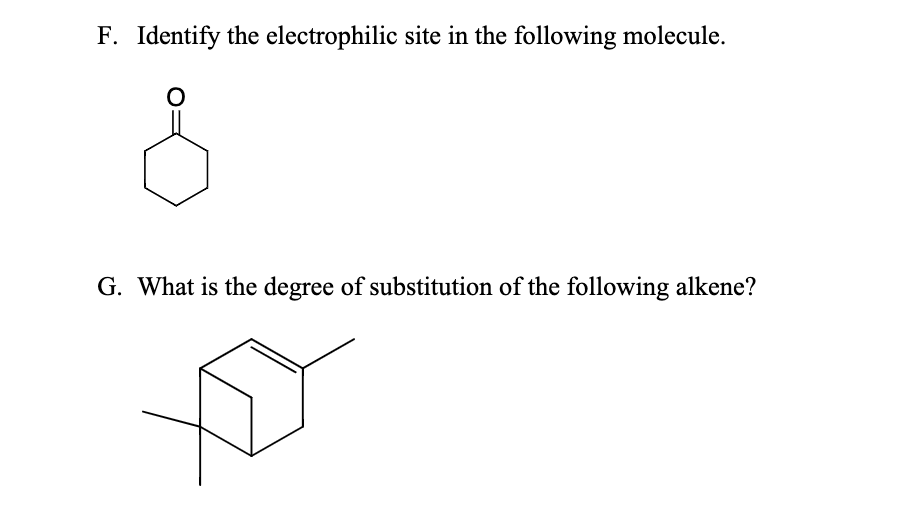
Another common scenario is when an atom has a positive formal charge. Now, formal charge can sound a bit intimidating, but think of it like a tally mark on an atom’s report card. If an atom ends up with more "belongs to me" electrons than it "should have" electrons, it gets a little positive +. This positive charge makes it incredibly attractive to anything with extra electrons. It's like putting out a "Free Hugs" sign, but with a charge.
You might see this in something like a carbocation. A carbocation is basically a carbon atom that’s lost an electron and now has a positive charge. It’s like the molecule has a little "missing" sticker on it, practically begging to be filled. These guys are super reactive and are always on the lookout for a electron-rich party guest to settle down with.
Sometimes, it's not an explicit positive charge, but a partial positive charge. This happens because of that electronegativity thing we talked about. The electrons are pulled away, leaving a region of the molecule that's a bit electron-poor. Think of it like a really popular restaurant. The main entrance is always packed, right? That packed area is like a partial positive charge – it's where all the action is happening, where the electron-rich folks are going to flock.
We often see this in molecules with double or triple bonds. Now, double and triple bonds are like the VIP lounges of the molecular world. They’ve got extra electrons floating around, making them quite attractive. But, sometimes, one side of that double or triple bond can become more electron-poor than the other. This can happen if there are electron-withdrawing groups attached to one of the atoms in the double bond. It’s like one side of the VIP lounge has better snacks than the other, so everyone crowds over there.
A classic example is a carbonyl group, which is a carbon double-bonded to an oxygen (C=O). Oxygen is super electronegative, so it pulls the electrons in the double bond towards itself. This leaves the carbon atom with a partial positive charge, making it our electrophilic site. It's like the oxygen is the stingy one, hogging all the good stuff, and the carbon is left feeling a bit needy. The carbon in that C=O is the prime suspect for being an electrophile.

Another scenario involves atoms that are "electron-deficient" because they’re trying to be good. Yes, you heard that right. Sometimes, an atom can be electron-deficient because it’s part of a group that's aiming for stability. For example, Lewis acids are often considered electrophiles. A Lewis acid is a substance that can accept an electron pair. Think of it like a very generous host who’s always ready to provide a comfortable spot for a guest to relax. They're not necessarily "missing" electrons in a negative way, but they have an "empty orbital" that's just waiting to be filled.
Consider something like a metal cation, like a positively charged sodium ion (Na+). It's basically a little ball of pure positive charge, practically screaming for electrons. It's like someone holding out an empty cup, hoping you'll fill it with something. These are very strong electrophiles because that positive charge is so intense.
We also see electrophiles in molecules that have undergone certain reactions. For instance, if a molecule is attacked by a strong oxidizing agent, it can lead to the formation of an electrophilic site. Oxidation often involves the loss of electrons, leaving a more electron-poor region. It’s like your car getting a bit rusty – the metal loses some of its "shine" and becomes more susceptible to further decay.
How do we pinpoint the exact spot?
So, how do we actually identify this electrophilic site in a specific molecule that might be shown to us? It's a bit like having a checklist, but instead of items, we're looking for characteristics. Here’s the game plan:
1. Look for those electron-hungry atoms:
Scan the molecule for atoms that are known to be very electronegative, like oxygen, nitrogen, chlorine, bromine, and fluorine. If these atoms are bonded to less electronegative atoms (like carbon or hydrogen), they’re probably pulling electrons towards themselves. The atom they’re bonded to is our potential electrophile.
2. Spot those positive charges (or hints of them):
Keep an eye out for any explicit positive charges. If you see a '+' symbol on an atom, that’s a dead giveaway. Also, be on the lookout for partial positive charges, indicated by the Greek letter delta with a '+' sign (δ+). These usually arise from the electronegativity differences we discussed. Think of it as a red flag saying, "Electron party here, all welcome!"
3. Investigate the double and triple bonds:
Double and triple bonds are electron-rich centers. However, check if there are any groups attached to the atoms in the bond that are pulling electron density away. If one atom in the double or triple bond is significantly more electron-poor than the other, that’s your electrophilic hotspot. It’s like the donut shop with the best jelly filling – everyone's going for that specific donut.

4. Consider the overall structure and functional groups:
Certain functional groups are notorious for having electrophilic sites. We already mentioned the carbonyl group (C=O). Other examples include epoxides (three-membered rings with an oxygen), acid halides, and alkyl halides. Recognizing these common patterns is like knowing that a pizza place is likely to have pizza – it's a good bet.
5. Think about what's missing:
Sometimes, an atom might have an empty orbital, making it an electrophile. This is common with some metal ions or compounds like boron trifluoride (BF3), where the boron atom doesn't have a full octet of electrons. It’s like having an empty seat at the dinner table; it's just waiting for someone to sit down.
It’s really about developing an intuition for where electrons are likely to be scarce. It’s not always about a direct positive charge; sometimes, it's a subtler imbalance. Think of it like looking at a crowded room. You can often tell where the most sought-after person is by looking at where the most people are trying to get close. That’s our electrophile – the molecule that’s drawing all the attention from electron-rich neighbors.
So, the next time you’re presented with a molecule and asked to find the electrophilic site, don’t panic. Just channel your inner detective, look for those electron-hungry atoms, the hints of positive charges, and the bustling double bonds. It's all part of the grand, slightly quirky dance of molecules, where everyone's looking for their perfect electron partner!
