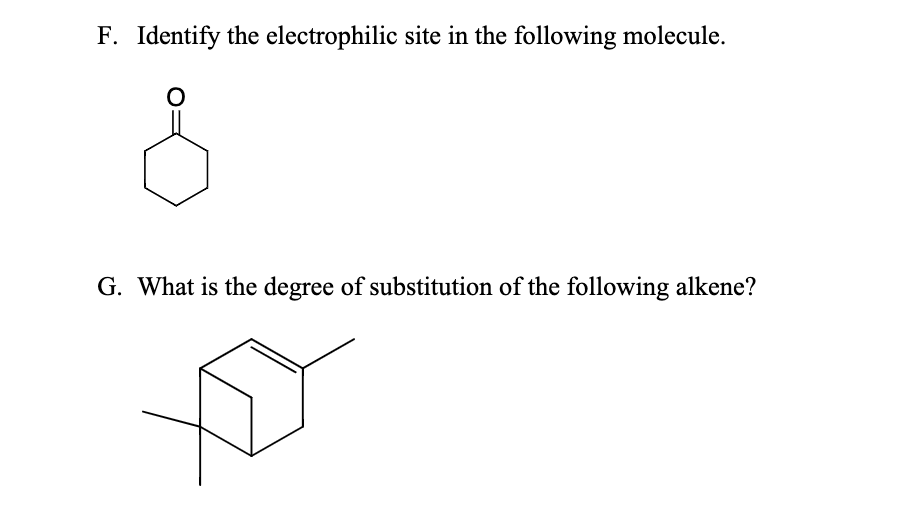
Identify The Electrophilic Site In The Following Molecule
So, picture this: I’m in my first year of organic chemistry, and we’re staring at these complex molecular diagrams. The professor, bless his patient soul, is trying to explain this concept called "electrophilic sites." My brain, at the time, was about as electrified as a potato battery. He kept going on about electron density, positive charges, and how some parts of a molecule are just begging for attention from electron-loving critters. I remember doodling a tiny cartoon of a molecule with its arms outstretched, a big ol' smiling sun (representing electrons) beaming down. My classmates probably thought I was spacing out, but in my head, I was having a serious epiphany. It was like a tiny, chemical romance novel was unfolding on my notebook page.
And that’s kind of what we’re diving into today! We’re going to play detective, but instead of chasing down a missing cookie jar, we’re hunting for electrophilic sites within molecules. Think of it as finding the “hot spots,” the places that are just dying to snag some electrons from their pals. It’s a super fundamental concept in organic chemistry, and honestly, once you get the hang of it, a whole new world of reactions opens up. Seriously, it’s like unlocking a cheat code!
Now, before we get our hands dirty with specific molecules, let’s make sure we’re on the same page about what an electrophile even is. The name itself is a bit of a giveaway, right? “Electro” for electricity, or in this case, electrons, and “phile” from the Greek word meaning “to love” or “to seek.” So, an electrophile is literally an electron-loving species. These guys are typically electron-deficient, meaning they have a bit of a negative charge deficit. They’re like the hungry ones at a buffet, always looking for something to feast on.
Think of it this way: if you're an electrophile, you're probably feeling a bit… lonely. You’ve got more positive charges (protons in the nucleus) than negative charges (electrons) in your immediate vicinity, or you’ve got an empty orbital just begging to be filled. So, you’re scanning the molecular landscape, looking for areas that are rich in electrons, areas that are willing to share. That electron-rich area? That’s the nucleophile. And the part of the nucleophile that’s doing the sharing? That’s where our electrophile wants to park itself. It’s a classic give-and-take, or in this case, a take-and-give!
So, how do we spot these eager electrophiles? It’s all about looking for certain tell-tale signs. The most obvious one is a positive charge. If you see a species with an explicit plus sign, like H₃O⁺ (hydronium ion) or an alkyl carbocation (R₃C⁺), bingo! That’s a prime electrophilic site. The positive charge screams, “I need electrons!” You can almost hear the desperate plea.
But it’s not always that straightforward. Electrophilic sites can also arise from polarity within a molecule. Remember electronegativity? That’s the tendency of an atom to attract electrons in a covalent bond. When you have two atoms with significantly different electronegativities bonded together, the electrons in that bond get pulled closer to the more electronegative atom. This creates a partial negative charge (δ⁻) on the more electronegative atom and a partial positive charge (δ⁺) on the less electronegative atom.
And guess what? That atom with the δ⁺? That’s your electrophilic site! It’s not fully positive, but it’s definitely got a deficiency, making it attractive to electron-rich species. This is super common in molecules containing atoms like oxygen, nitrogen, or halogens bonded to less electronegative atoms like carbon or hydrogen. Think of water (H₂O). Oxygen is way more electronegative than hydrogen, so the oxygen pulls electrons from the hydrogens. The hydrogens in water are slightly positive, making them potential electrophilic sites. Not super strong electrophiles, mind you, but they can still react!
Another big clue is the presence of unfilled valence orbitals. Some atoms or molecules might not have a positive charge or a significant partial positive charge, but they have an empty spot where electrons could go. The classic example is a Lewis acid, like Boron trifluoride (BF₃). Boron has only six valence electrons around it, leaving an empty p orbital. This empty orbital is like an open invitation for electrons. It’s just waiting for someone to come along and fill it. So, BF₃ is a powerful electrophile.
Okay, okay, enough theory. Let’s get to the good stuff. We’re going to look at a molecule and figure out where the electrophilic sites are. Let’s say our molecule is… a simple aldehyde! Something like acetaldehyde, CH₃CHO. Or, to make it a bit more general, R-CHO, where R is some hydrocarbon group.
First things first, let's draw it out or visualize it. We have a carbon atom double-bonded to an oxygen atom, and that carbon is also bonded to a hydrogen atom and an R group. So, it looks something like this:
O
//
R - C - H
Now, let’s analyze this beast. We’ve got carbon, oxygen, hydrogen, and the atoms in our R group. We need to look for those signs we talked about: positive charges, partial positive charges, or empty orbitals.
Let’s start with the atoms themselves. Oxygen is way more electronegative than carbon and hydrogen. This is a crucial piece of information. Because oxygen is pulling electrons towards itself, the electrons in the C=O double bond are going to be pulled towards the oxygen. What does this mean for the atoms involved?
The oxygen atom, because it’s hogging all the electrons, will have a partial negative charge (δ⁻). This makes the oxygen atom a nucleophilic site, not an electrophilic one. It’s too electron-rich to be looking for more. It's the one offering the goodies!
Now, what about the carbon atom in the carbonyl group (the C=O part)? Since the oxygen is pulling electrons away from it, this carbon atom becomes electron-deficient. It develops a partial positive charge (δ⁺). Aha! This is our prime candidate for an electrophilic site! It’s got that δ⁺ that’s just begging for an electron-loving species to come and interact with it.
What about the hydrogen atom bonded to this carbonyl carbon? Hydrogen is less electronegative than carbon. However, in this particular arrangement, the carbonyl carbon is the more electron-deficient center due to the strong pull of the oxygen. So, while the C-H bond might have some polarity, the most significant electrophilic site is definitely the carbonyl carbon.
And the R group? Well, that depends on what R is. If R is something like a methyl group (CH₃), the carbons and hydrogens in the R group are generally not significantly electrophilic. They’re mostly involved in sigma bonds with other carbons and hydrogens, which are pretty evenly shared electrons. However, if R contained something like a halogen or another electronegative atom, then parts of R could become electrophilic. But for a simple aldehyde like acetaldehyde, the star of the show, the electrophilic site is that carbonyl carbon.
So, in R-CHO, the carbon atom of the carbonyl group is the electrophilic site. This is why aldehydes are so reactive towards nucleophiles! Think of adding something like a Grignard reagent (which is nucleophilic) to an aldehyde. The nucleophilic carbon of the Grignard reagent attacks the electrophilic carbonyl carbon, forming a new carbon-carbon bond. It’s a beautiful dance of electron transfer!
Let’s try another one. How about a carboxylic acid derivative, like an ester? Let’s take methyl acetate, CH₃COOCH₃. The structure looks like this:
O
//
CH₃ - C - O - CH₃
Again, we're looking for those electron deficiencies. We have a carbonyl group (C=O) here, just like in the aldehyde. So, we already know that the carbon atom of the carbonyl group is going to be partially positive (δ⁺) because of the oxygen pulling electrons. This makes it an electrophilic site.
What else is going on? We have an oxygen atom bonded to that carbonyl carbon, and that oxygen is also bonded to a methyl group. Let's consider the electronegativity differences. Oxygen is more electronegative than carbon. So, the electrons in the C-O single bond between the carbonyl carbon and the oxygen of the -OCH₃ group are pulled towards that oxygen. This makes the carbonyl carbon even more electron-deficient.
And what about the oxygen atom in the -OCH₃ group? It's bonded to a carbon. Oxygen is more electronegative than carbon, so the electrons in the C-O bond are pulled towards the oxygen. This gives that oxygen atom a partial negative charge (δ⁻), making it nucleophilic.
Now, here's a bit of nuance. In esters, that carbonyl carbon is indeed electrophilic. However, it’s generally considered less reactive towards nucleophilic attack compared to aldehydes or ketones. Why? Because of the electron-donating effect of the oxygen atom from the -OCH₃ group. It pushes some electron density back towards the carbonyl carbon, slightly reducing its electrophilicity. It’s like the carbonyl carbon is getting a little bit of comfort food from its neighbor.
But if we were asked to identify the electrophilic site, it would still be the carbonyl carbon. It’s just that the surrounding atoms and groups can influence how electrophilic it is.
Let's consider something a bit different. What about an alkyl halide? Like, say, bromoethane, CH₃CH₂Br.
Br - CH₂ - CH₃
Here, we have a carbon atom bonded to a bromine atom. Bromine is a halogen, and it’s significantly more electronegative than carbon. So, the electrons in the C-Br bond are pulled towards the bromine. This creates a partial negative charge (δ⁻) on the bromine atom. And, consequently, it creates a partial positive charge (δ⁺) on the carbon atom directly bonded to the bromine.
So, in bromoethane, the carbon atom attached to the bromine is the electrophilic site. This is why alkyl halides are susceptible to nucleophilic substitution reactions. A nucleophile can come in and attack that δ⁺ carbon, kicking out the bromide ion (which is a good leaving group because it can stabilize the negative charge). It's a classic SN2 reaction scenario!
What if we have a molecule with resonance structures? This is where things get really interesting and sometimes a bit tricky. Consider the carbonate ion, CO₃²⁻. It has a formal charge of -2.
If we just look at one resonance structure:
O⁻
|
O = C - O⁻
Here, the carbon atom is double-bonded to one oxygen and single-bonded to two other oxygens, each carrying a negative charge. The carbon has four bonds and no lone pairs, so its formal charge is 0. However, the oxygens are clearly nucleophilic with their negative charges.
But carbon can be electrophilic in other ways. Let’s think about structures that might delocalize negative charge onto carbons. That’s not quite what we’re looking for here.
Let’s revisit the concept of empty orbitals. Consider BF₃ again. Boron has three valence electrons. It forms three covalent bonds with fluorine atoms, which each contribute one electron. That gives boron 3 + 3 = 6 valence electrons around it. It’s also in the second period, and its 2p orbitals are available. There’s an empty 2p orbital on the boron atom. This makes boron trifluoride a potent Lewis acid and thus an electrophile. The boron atom itself is the electrophilic site.
What about aromatic compounds? Take benzene (C₆H₆). At first glance, it looks pretty electron-rich with all those pi electrons. However, benzene is quite stable. But in electrophilic aromatic substitution (EAS) reactions, we do get electrophilic attack on the ring. How does that happen?
Well, the electrophile itself has to be strong enough to react with the relatively stable pi system of benzene. The ring carbons themselves aren’t inherently electrophilic in the way a carbonyl carbon is. Instead, the electrophile attacks the electron cloud of the aromatic ring, and a carbocation intermediate is formed. So, in EAS, we are really looking at the interaction between a strong external electrophile and the pi electron system of the aromatic ring. The carbons within the benzene ring become part of the electrophile's target, but they aren't the primary electrophilic site within the benzene molecule itself unless there's a substituent that makes them so.
Let’s consider a substituted benzene, like nitrobenzene. The nitro group (-NO₂) is electron-withdrawing. It pulls electron density away from the benzene ring through both inductive and resonance effects. This makes the carbons of the benzene ring, particularly those ortho and para to the nitro group, less electron-rich and therefore more susceptible to attack by very strong electrophiles. But the nitro group itself has an electrophilic component. The nitrogen atom in the nitro group is double-bonded to one oxygen and single-bonded to another, which has a negative charge. The nitrogen atom in the nitro group can be considered somewhat electrophilic because it’s bonded to two highly electronegative oxygen atoms and has electron density pulled away from it.
The key takeaway here is to always look for those indicators: positive charges, partial positive charges due to electronegativity differences, and unfilled valence orbitals. These are your bread and butter for spotting electrophilic sites.
It’s like learning a secret language. Once you understand these fundamental principles, you can look at almost any molecule and start to predict where it’s going to react and with what. It’s not about memorizing every single reaction; it’s about understanding the underlying electron flow and what drives these chemical interactions.
So, when you’re presented with a molecule, take a deep breath. Break it down atom by atom. Consider the bonds between them. Ask yourself: “Who’s pulling electrons? Who’s feeling a bit short-changed? Who’s got an empty spot?” Your answers will lead you straight to the electrophilic site. Happy hunting!
