Identify The Electrophile In The Bromination Of Benzene
Alright, let's talk about a little something that might sound a bit intimidating at first. We're diving into the exciting world of chemistry, specifically a reaction called the bromination of benzene. Now, before you imagine beakers bubbling and mad scientists cackling, let's just take a deep breath. It's not as scary as it sounds. Think of it like a cosmic dating game for molecules.
Our main player in this drama is a super chill, six-carbon ring called benzene. It's like the popular kid in school, always looking for a dance partner. Benzene is pretty stable, sort of like someone who’s happy with their current life. It’s got its electrons all cozy and paired up. But sometimes, even the most content molecules get a little curious. They see other, more energetic molecules buzzing around and think, "Hmm, what's all the fuss about?"
Enter our other character: bromine. Now, bromine is usually a bit of a drama queen. It comes in pairs, like a bickering couple, as Br₂. This molecule, Br₂, is a bit like two people who can't quite agree on anything. One bromine atom is always trying to hog the electrons, and the other feels a bit neglected. This constant tug-of-war makes one of the bromine atoms just a little bit positive.
And that, my friends, is where our superhero, or perhaps more accurately, our villain, comes into play. We're looking for the electrophile. Now, the word "electrophile" sounds fancy, right? It's like something out of a sci-fi movie. But really, it just means "electron-lover." Think of an electrophile as that one friend who's always looking for the next big thing, the next exciting adventure. They're attracted to places that have lots of electrons, like a moth to a flame, or me to a free slice of pizza.
So, in our benzene bromination scenario, who is this electron-lover? Who is the molecule that's going to make benzene say, "Ooh, tell me more!"? It’s not the whole Br₂ molecule, not as it is. That's like a whole relationship. We need someone a bit more focused, a bit more… intense. We need a molecule that’s positively charged, or at least has a strong positive vibe going on.
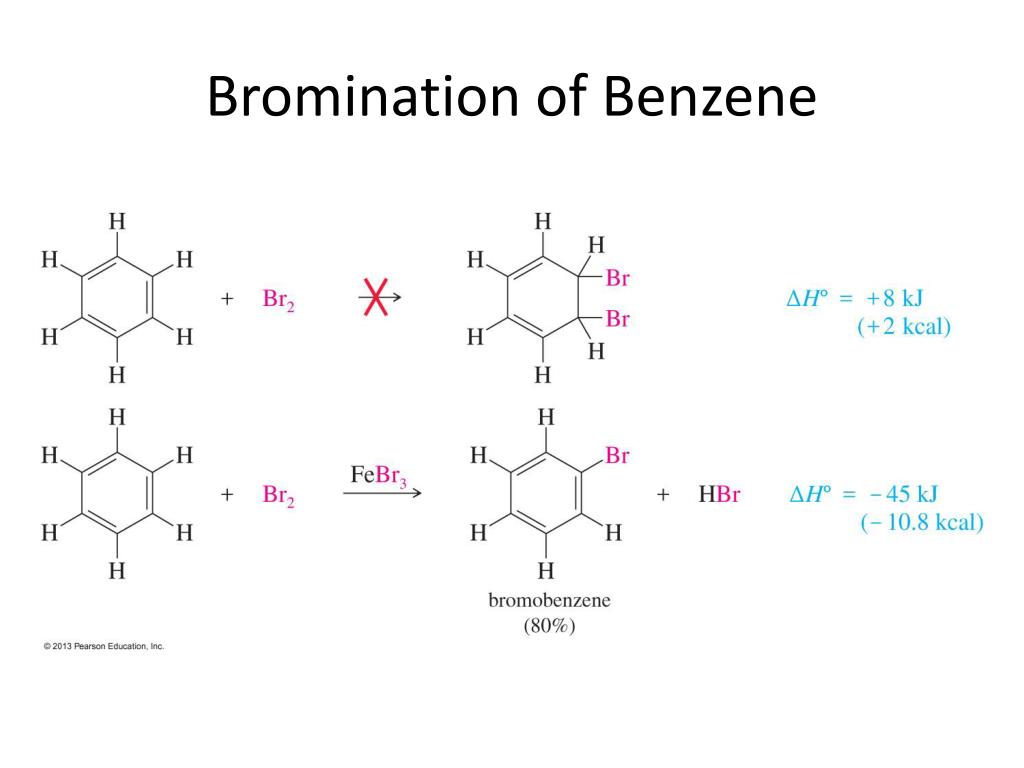
Here’s where it gets interesting, and perhaps a little controversial. You might think it’s the whole Br₂ molecule. And I get it. It’s right there, ready to mingle. But honestly, and this is my unpopular opinion, the full Br₂ molecule is a bit too… diffuse. It’s like inviting a whole band to a small party. It’s a bit much.
What we really want is someone with a singular, powerful attraction. We want a focused individual, someone who knows exactly what they’re looking for. And in this case, the real star of the show, the one that benzene can’t resist, is actually a fragment of that bromine molecule. It's like when you go to a party, and you don't talk to the whole couple, you talk to the one person who catches your eye.
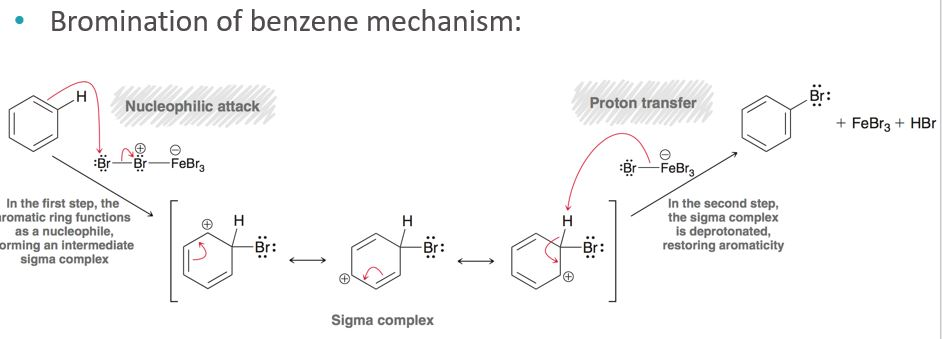
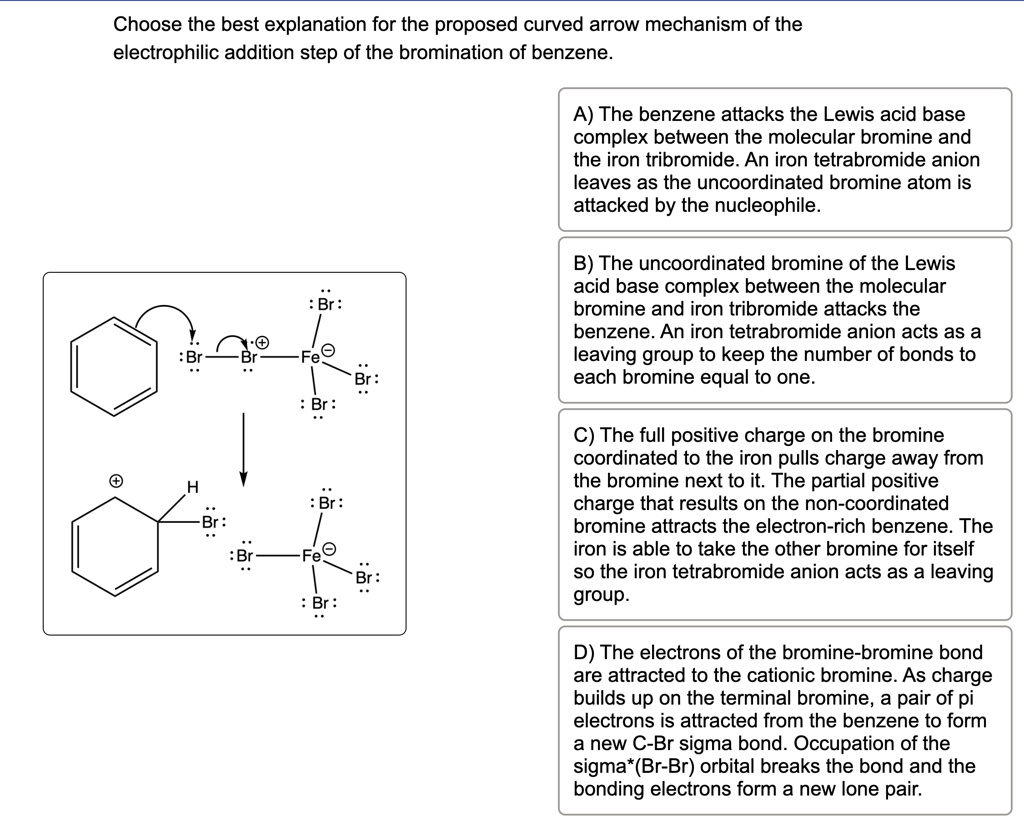
The true electrophile is a single, positively charged bromine atom. Think of it as a free agent, ready to make a move.
This little guy, this positively charged bromine atom, is like the charming stranger at the ball. It's got this positive energy that just screams "electron-deficient!" Benzene, with all its electron-rich glory, is like, "Hello there, handsome!" This positively charged bromine atom is the one that directly interacts with benzene. It’s the one that initiates the whole exciting tango.

The other part of the Br₂ molecule? Well, it's kind of just hanging around, maybe a little jealous, maybe just waiting for its turn. But it’s not the main attraction. It’s not the one that’s electrophilic. It’s the lone ranger, the solo act, the positively charged bromine ion, that’s the key player here.
So next time you hear about the bromination of benzene, don't get bogged down by the whole Br₂. Remember the little guy with the big attitude, the one with the positive charge. That’s your true electrophile. It’s the spark that ignites the reaction. It’s the molecule that makes benzene forget its chill and jump into action. And isn’t that, in its own chemical way, just a little bit romantic?
