Identify The Correct Values For A 3p Sublevel.
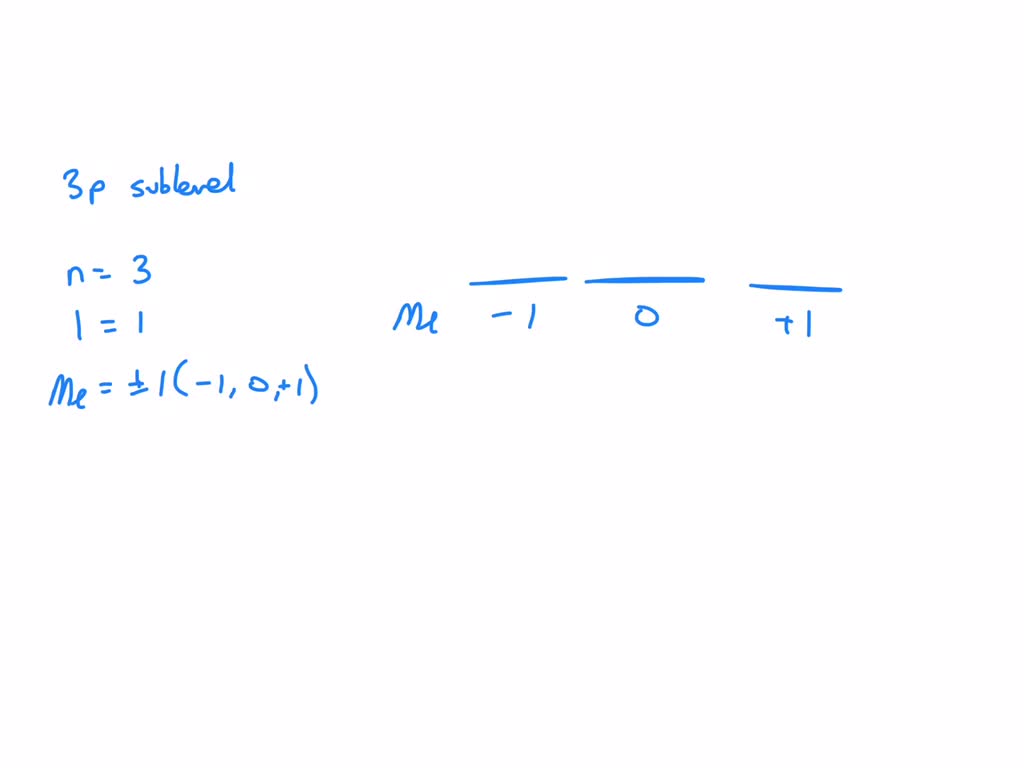
Ever wondered what makes the universe tick at its tiniest, most fundamental level? It's a bit like solving a cosmic puzzle, and one of the most intriguing pieces involves the way electrons arrange themselves within atoms. Forget complicated jargon; we're diving into the exciting world of electron shells and sublevels, specifically the elusive 3p sublevel! Think of it as discovering the perfect seating arrangement for energetic party guests (electrons) in a special room (an atom). Understanding this is not just for super-smart scientists; it unlocks a deeper appreciation for everything around us, from the sparkle of a diamond to the way your phone works.
So, why is this particular sublevel, the 3p sublevel, so interesting? Well, it’s a crucial stepping stone in understanding how atoms bond and interact. Each atom has a unique electron configuration, like a fingerprint, and the 3p sublevel plays a significant role in defining that fingerprint. When we can correctly identify the values associated with the 3p sublevel, we’re essentially decoding the atom’s personality. This knowledge helps us predict how elements will behave, why certain chemical reactions occur, and how new materials are formed. It’s like having a secret code to unlock the mysteries of chemistry!
The Magic Numbers: Quantum Numbers!
To figure out the "correct values" for the 3p sublevel, we need to introduce its trusty sidekicks: the quantum numbers. These aren't just random numbers; they are the universal address system for electrons within an atom. Each electron in an atom has a unique set of four quantum numbers that tells us where it's likely to be found and what its energy and spin are like. They are:
- The principal quantum number (n): This is like the main building number. It tells us the energy level or shell the electron is in. Higher numbers mean higher energy levels and being further from the nucleus.
- The angular momentum quantum number (l): This is like the apartment number within the building. It describes the shape of the electron's orbital within a sublevel.
- The magnetic quantum number (ml): This is like the specific room number within the apartment. It specifies the orientation of the orbital in three-dimensional space.
- The spin quantum number (ms): This is like whether you're facing left or right in the room. It describes the intrinsic angular momentum of the electron, often visualized as spinning either clockwise or counterclockwise.
These four numbers work together to paint a complete picture of an electron's state. It’s a bit like giving someone a full address: street, house number, apartment, and which side of the room they're sitting on!
Unpacking the 3p Sublevel
Now, let's zero in on our star player: the 3p sublevel. When we talk about the "3p" designation, we're already getting clues about some of its quantum numbers.
The '3' in 3p directly tells us the principal quantum number (n). So, for any electron in the 3p sublevel, n = 3. This means these electrons are in the third energy shell, further out from the nucleus than electrons in the first or second shells.
The 'p' in 3p is a code for the angular momentum quantum number (l). Different letters correspond to different values of 'l':

- s sublevels have l = 0
- p sublevels have l = 1
- d sublevels have l = 2
- f sublevels have l = 3
Since we're looking at a 'p' sublevel, we know that for electrons in the 3p sublevel, l = 1. This value of l = 1 tells us that the orbitals in this sublevel have a specific shape – they are dumbbell-shaped, with two lobes on opposite sides of the nucleus.
The Orientations: Finding the ml Values
With n = 3 and l = 1, we can now determine the possible values for the magnetic quantum number (ml). The rule here is that 'ml' can take on any integer value from -l to +l, including zero. Since our 'l' is 1, the possible values for 'ml' are:

- ml = -1
- ml = 0
- ml = +1
These three values of 'ml' represent the three different p orbitals within the 3p sublevel. Each of these orbitals has the same dumbbell shape (because l=1) but is oriented differently in space. We often refer to these as the 3px, 3py, and 3pz orbitals, indicating their orientation along the x, y, and z axes, respectively. Think of them as three identical party rooms, but each is positioned differently in the apartment!
The Spin: The ms Value
Finally, we have the spin quantum number (ms). This number can only be one of two values: +1/2 or -1/2. This represents the two possible spin orientations of an electron. So, for any electron in the 3p sublevel, ms can be either +1/2 or -1/2.

According to the Pauli Exclusion Principle, no two electrons in an atom can have the exact same set of all four quantum numbers. This means that each of the three 3p orbitals (defined by their ml values) can hold a maximum of two electrons, one with ms = +1/2 and the other with ms = -1/2. Therefore, the entire 3p sublevel can accommodate a total of 3 orbitals * 2 electrons/orbital = 6 electrons.
Putting It All Together
So, to summarize, when we identify the correct values for a 3p sublevel, we are referring to the set of quantum numbers that describe the electrons residing there. For any electron in the 3p sublevel, the quantum numbers are:
- n = 3 (the principal energy level)
- l = 1 (indicating a p sublevel with dumbbell-shaped orbitals)
- ml = -1, 0, or +1 (representing the three possible spatial orientations of the p orbitals)
- ms = +1/2 or -1/2 (representing the two possible spin states of the electron)
Understanding these values allows us to accurately draw electron configurations, predict chemical reactivity, and appreciate the fundamental building blocks of matter. It's a small piece of the cosmic puzzle, but a truly essential one that helps us understand the fascinating world of atoms!
