Identify The Correct Values For A 2p Sublevel.
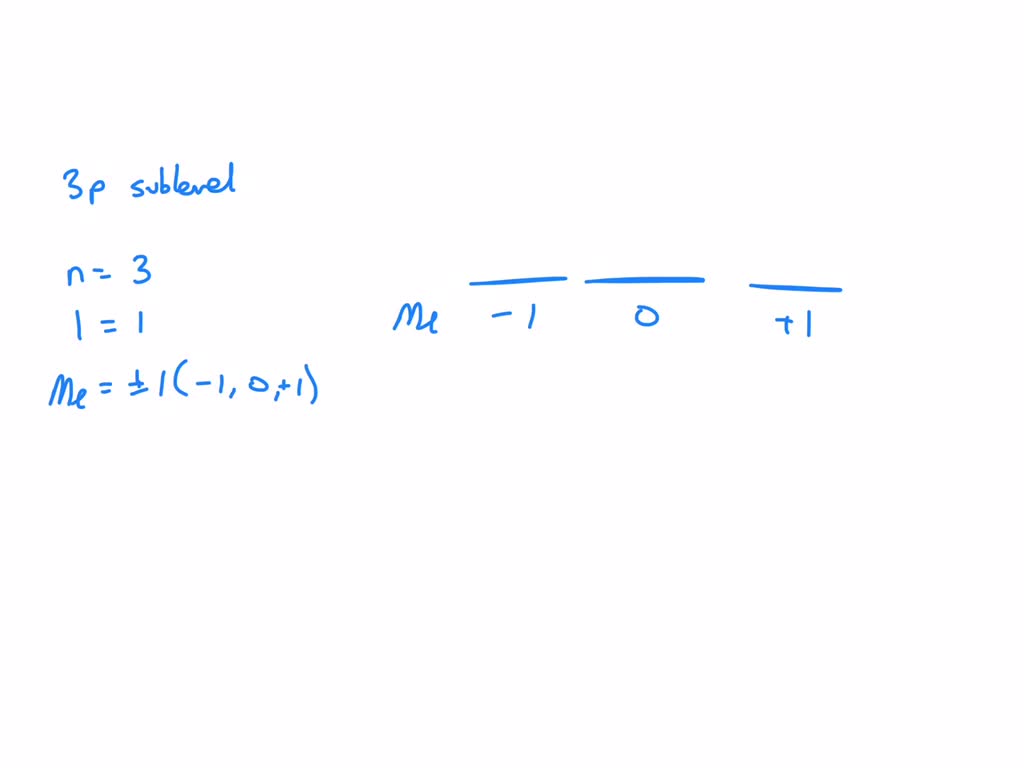
Alright, let's talk about something that might make your eyes glaze over faster than a lukewarm cup of tea. We're diving into the exciting world of... electron shells and their little buddies, the sublevels. Specifically, we're going to shine a spotlight on the rather charming 2p sublevel. Now, I know what you're thinking. "Sublevels? Is this going to be harder than assembling IKEA furniture with only a picture diagram?" Fear not, my friends! We're going to tackle this with the enthusiasm of someone finding an extra fry at the bottom of their fast-food bag.
So, we have our trusty electron, zipping around the nucleus like a tiny, energetic bee. These bees like to hang out in neighborhoods, and these neighborhoods are called shells. Think of them like floors in a really, really big apartment building. The closer the shell is to the nucleus (the landlord, perhaps?), the more desirable the real estate. Shell number one, the 1s shell, is basically the penthouse suite. Super exclusive, only one room, and very, very stable. Boring, if you ask me. Give me a bit of excitement!
Now, as we move up the floors, the apartment building gets a bit more complex. Shell number two, the 2s shell, is like a nice, cozy studio apartment on a slightly less prestigious floor. Still pretty chill. But then, on floor two, we get a whole new wing. This is where things start to get interesting. This wing is the 2p sublevel. Imagine it as a floor with three separate apartments, all a bit different but still on the same level. It’s like a split-level loft with a bonus den, but for electrons.
So, what makes these 2p apartments tick? Well, they're not just randomly placed. They have specific addresses, or in science-speak, they have distinct quantum numbers. These numbers are like the keys to the electrons' little homes. We’ve got the principal quantum number, which tells us which floor we're on. For our 2p sublevel, that's a big ol' 2. Simple enough, right? We're on floor two. No surprises there.
Then we have the angular momentum quantum number. This one is a bit more fancy. It tells us the shape of the apartment. For the s sublevels, the shape is a sphere. Think of a perfectly round balloon. For the p sublevels, the shape is a bit more... dramatic. It's like a dumbbell. You know, those things you see at the gym that look like they're trying to punch each other? Yep, that's the vibe. So, for the 2p sublevel, this number is a 1. It signifies that we’re dealing with these charming dumbbell shapes, not the boring spheres.

Now, here's where it gets really fun, and I’m about to commit a minor academic crime by making it sound fun. We have the magnetic quantum number. This little guy tells us the orientation of those dumbbell apartments in space. Are they standing up? Are they lying down? Are they doing a handstand? There are three of these dumbbell apartments in the 2p sublevel, remember? So, this quantum number can take on three values: -1, 0, and +1. Think of it like this: one apartment is pointing along the 'x' axis, one along the 'y', and one along the 'z'. It's like having three separate bedrooms, each with a different view!
It's not about the destination, it's about the journey... and the orientation of your electron's apartment!
2p Sublevel Values Codes Sale | dev-techtatva.manipal.edu
And finally, the pièce de résistance, the spin quantum number. Every electron, no matter how fancy its apartment, has a little spin to it. It's like it’s constantly doing a tiny, internal pirouette. This spin can be either "up" or "down." So, for any electron in any orbital within our 2p sublevel, this number can be either +1/2 or -1/2. It’s like the electron’s personal mood ring. Up today? Down tomorrow? Who knows!
So, to recap the correct values for a 2p sublevel: The principal quantum number is 2 (we're on the second floor). The angular momentum quantum number is 1 (we've got those fancy dumbbell shapes). The magnetic quantum numbers can be -1, 0, and +1 (three distinct orientations for our dumbbells). And for each individual electron within those orientations, the spin quantum number is either +1/2 or -1/2 (up or down spin).
There you have it! The not-so-secret lives of the 2p sublevel. It’s not as scary as it sounds, is it? It’s just electrons having their own little organized chaos, and we’re just peeking into their world. Now, if you’ll excuse me, I need a cup of that lukewarm tea. All this talk of quantum numbers has made me thirsty for something utterly unremarkable.

