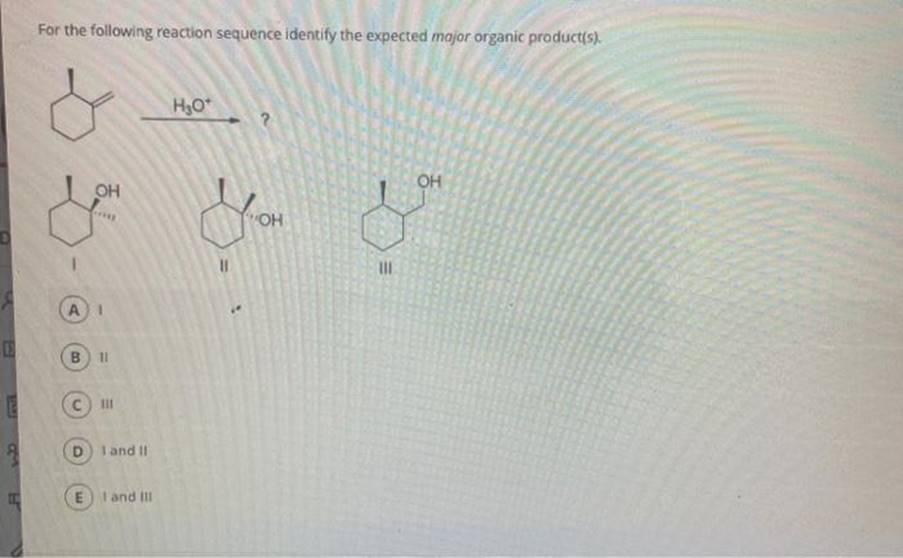
Identify The Correct Product Of The Reaction Sequence Below:
So, you know those moments when you're trying to bake a cake, and you've got all the ingredients laid out – flour, sugar, eggs, the whole shebang – but then you realize you forgot the baking soda? Disaster, right? Or maybe you're building some IKEA furniture, and you've got a whole pile of oddly shaped wooden bits and a bag of screws, and you're staring at the instructions like they're written in ancient hieroglyphics. You squint, you turn it upside down, you might even do a little jig of frustration. That feeling, that exact feeling of trying to put pieces together to get a final, functional thing, is basically what chemists have to deal with every single day. Except, you know, with molecules instead of flour and particleboard.
And speaking of putting molecules together, we're going to dive into a little chemical puzzle today. It’s like a detective story, but instead of a smoking gun, we're looking for the final product of a reaction sequence. You know, where you start with one thing, do some magic with it, then do some more magic with the result, and so on, until BAM! You’ve got your prize. It’s all about understanding how these tiny building blocks behave and what happens when you nudge them in certain directions. Fun, right?
Our little adventure today involves a specific set of chemical reactions. Think of it as a recipe, but instead of telling you to "add a pinch of salt," it tells you to "treat with reagent X under condition Y." The goal? To figure out what delicious (or, you know, just formed) molecule pops out at the end of all the fuss. It's a common task for any aspiring chemist, and honestly, it’s pretty satisfying when you nail it. Like finally getting that IKEA drawer to slide smoothly. Ah, the little victories!
So, let's get down to business. We're going to look at a sequence of reactions and try to pinpoint the correct product. This isn't just about guessing; it's about using our knowledge of organic chemistry principles to predict what will happen. Think of it as a mental workout for your brain. And hey, who doesn't love a good mental workout? Especially when it involves cool chemical transformations.
The Starting Point: A Familiar Face
Our journey begins with a molecule that's probably quite familiar to you, even if you don't realize it. We're talking about benzene. Yep, that classic six-membered ring with alternating double bonds. It's a pretty stable molecule, a bit of a rockstar in the organic chemistry world. It’s the foundation for a lot of important compounds, from pharmaceuticals to plastics. So, it's a good place to start. It's like the sturdy base of your cake – essential for everything that follows.
Benzene looks like this (imagine a hexagon with a circle in the middle, or alternating double bonds – chemists love their shorthand, you know). Its aromaticity, that special kind of stability, makes it resistant to some reactions but very susceptible to others. It’s not going to just randomly fall apart; you need to give it a good reason to react, and a specific kind of reason at that.
Now, what do we do to our beloved benzene? The first step in our sequence involves a nitration reaction. Ever heard of it? It's essentially adding a nitro group (-NO2) to a molecule. And for benzene, the classic way to do this is by using a mixture of concentrated nitric acid (HNO3) and concentrated sulfuric acid (H2SO4). Sulfuric acid acts as a catalyst here, which is super important. It helps generate the electrophile that's going to attack the benzene ring.
The electrophile in this case is the nitronium ion (NO2+). It's positively charged, meaning it's electron-deficient and looking for some electron-rich territory to latch onto. Benzene, with its delocalized pi electrons, is just the place. So, the NO2+ will happily attack the benzene ring, and after a few steps (which involve losing a proton to regain that aromatic stability, because benzene really likes being aromatic), we end up with nitrobenzene.
So, our first product is nitrobenzene. See? Not too scary, right? We’ve successfully put a nitro group onto our benzene ring. It’s like successfully adding the flour to your cake batter. One step down, a few more to go. And don't worry if the mechanism sounds a bit technical; the important thing is to grasp the overall transformation: benzene becomes nitrobenzene.
Step Two: More Than Meets The Eye
Now, what do we do with our shiny new nitrobenzene? The next step is a reduction reaction. Reduction, in organic chemistry, can mean a few things, but most commonly it involves adding hydrogen or removing oxygen. In this scenario, we’re going to reduce the nitro group (-NO2) all the way down to an amino group (-NH2). This is a pretty significant transformation because it changes the functionality of the molecule drastically.
There are several ways to achieve this reduction. Common reagents include things like catalytic hydrogenation (using hydrogen gas and a metal catalyst like palladium or platinum) or using metals like tin (Sn) or iron (Fe) in acidic conditions. For our purposes, let's imagine we're using something like tin and hydrochloric acid (Sn/HCl). This combination is a classic for reducing nitro groups to amines.
The reaction involves a series of electron and proton transfers, where the oxygen atoms of the nitro group are gradually replaced by hydrogen atoms, and eventually, the nitrogen atom is bonded to two hydrogens. The outcome? We transform nitrobenzene into aniline. Aniline is a really important compound itself, serving as a precursor for dyes, plastics, and pharmaceuticals. It’s like, after you’ve got your cake batter, you’ve now got it perfectly mixed, ready for the oven. The structure is fundamentally different and opens up new possibilities.
So, after step two, we have aniline. We started with benzene, nitrated it to nitrobenzene, and then reduced that nitro group to an amino group, giving us aniline. Feeling like a chemist yet? It’s like following a recipe, step by step, and seeing your ingredients transform into something new and exciting. Keep those hands clean, and your understanding sharp!
The Plot Thickens: Introducing A New Player
Alright, we’ve got aniline. It’s got that –NH2 group, which is pretty reactive. Now, things get a little more interesting. The next reaction involves treating aniline with sodium nitrite (NaNO2) in the presence of a strong acid, typically hydrochloric acid (HCl), and at a low temperature (usually 0-5°C). This set of conditions is crucial, so pay attention!
This reaction is called diazotization. What happens is that the primary amino group (-NH2) of aniline reacts with nitrous acid (HNO2), which is formed in situ from sodium nitrite and HCl. The low temperature is essential because the intermediate formed is quite unstable and can decompose at higher temperatures. This is a good reminder that sometimes, the conditions are just as important as the reagents themselves. It's like making sure your oven is at the exact right temperature for that delicate pastry.
The product of this diazotization reaction is a diazonium salt. Specifically, when aniline reacts with NaNO2/HCl at low temperatures, it forms benzenediazonium chloride. This molecule has a –N≡N+ group attached to the benzene ring, with a chloride ion as the counterion. The structure looks like a benzene ring with a –N≡N+Cl- group. This diazonium group is a fantastic leaving group, which is key to what happens next.
So, after step three, we have benzenediazonium chloride. This is a really versatile intermediate, and chemists have come up with all sorts of clever ways to use it. It's like you've just mastered a complex culinary technique – now you can create all sorts of amazing dishes. It’s a stepping stone to many other fascinating molecules.
Step Four: The Grand Finale (Almost!)
Now, what do we do with this unstable but oh-so-useful benzenediazonium chloride? This is where we get to choose our adventure a bit, as diazonium salts can react in many ways. However, in our specific reaction sequence, the question asks for the correct product. This implies a particular pathway is intended. Let's assume the next step involves reacting the benzenediazonium chloride with water (H2O), and this time, we're going to heat it up. Remember that low temperature in the previous step? This is the opposite!
When benzenediazonium chloride is heated in water, the diazonium group (–N≡N+) is an excellent leaving group. It’s so good, in fact, that it leaves as a molecule of nitrogen gas (N2). Yes, that inert gas that makes up most of our atmosphere. And as the diazonium group departs, a water molecule comes in to fill the void. Specifically, the oxygen from the water molecule will attack the carbon atom that was previously bonded to the diazonium group.
This leads to the formation of a phenol. Phenols are compounds where a hydroxyl group (-OH) is directly attached to an aromatic ring. So, our benzenediazonium chloride, upon heating in water, will transform into phenol. The benzene ring will now have a –OH group attached to it, and we'll have N2 gas and HCl as byproducts.
So, our sequence has taken us from benzene, through nitrobenzene and aniline, to benzenediazonium chloride, and finally, to phenol. This is a classic route for synthesizing phenol, and it demonstrates several key organic reactions: electrophilic aromatic substitution (nitration), reduction of a nitro group, diazotization of a primary amine, and nucleophilic aromatic substitution where the diazonium group acts as the leaving group.
Putting It All Together: The Final Answer
Let’s recap our journey. We started with benzene. We added a nitro group to make nitrobenzene. We reduced the nitro group to an amino group to make aniline. We then converted aniline into a diazonium salt, benzenediazonium chloride, under specific cold, acidic conditions. And finally, by heating that diazonium salt in water, we replaced the diazonium group with a hydroxyl group.
Therefore, the correct product of this reaction sequence is phenol.
It's like building a Lego castle. You start with a few base bricks (benzene), then you add more specific pieces (nitro group), then you modify those pieces (reduce to amino), then you add a special connector (diazonium salt), and finally, you attach the flag or the roof (hydroxyl group). Each step builds upon the last, and understanding each transformation is key to predicting the final outcome.
Why is this important? Well, being able to predict reaction products is fundamental to organic chemistry. It's how new drugs are designed, how new materials are synthesized, and how we understand the complex processes happening in nature. It’s the core skill that allows chemists to be molecular architects.
Consider the alternatives. If we had stopped after the first step, we’d have nitrobenzene. If we’d stopped after the second, we’d have aniline. If we had treated the diazonium salt differently, say, with a copper(I) halide, we might have gotten a halogenated benzene (like chlorobenzene or bromobenzene). But given the specific prompt of heating the diazonium salt in water, phenol is the clear and correct destination.
It's a beautiful demonstration of how functional groups can be manipulated and transformed sequentially to achieve a desired molecule. And while it might seem like a lot of steps, each one follows well-established chemical principles. It's about understanding the reactivity of each intermediate and the conditions required for each transformation.
So, next time you're faced with a reaction sequence, think of it as a chemical puzzle. Break it down step by step, identify the functional groups, consider the reagents and conditions, and predict the most likely outcome. It’s a skill that improves with practice, and the satisfaction of cracking the code is, well, pretty awesome. And that, my friends, is how we arrive at phenol!
