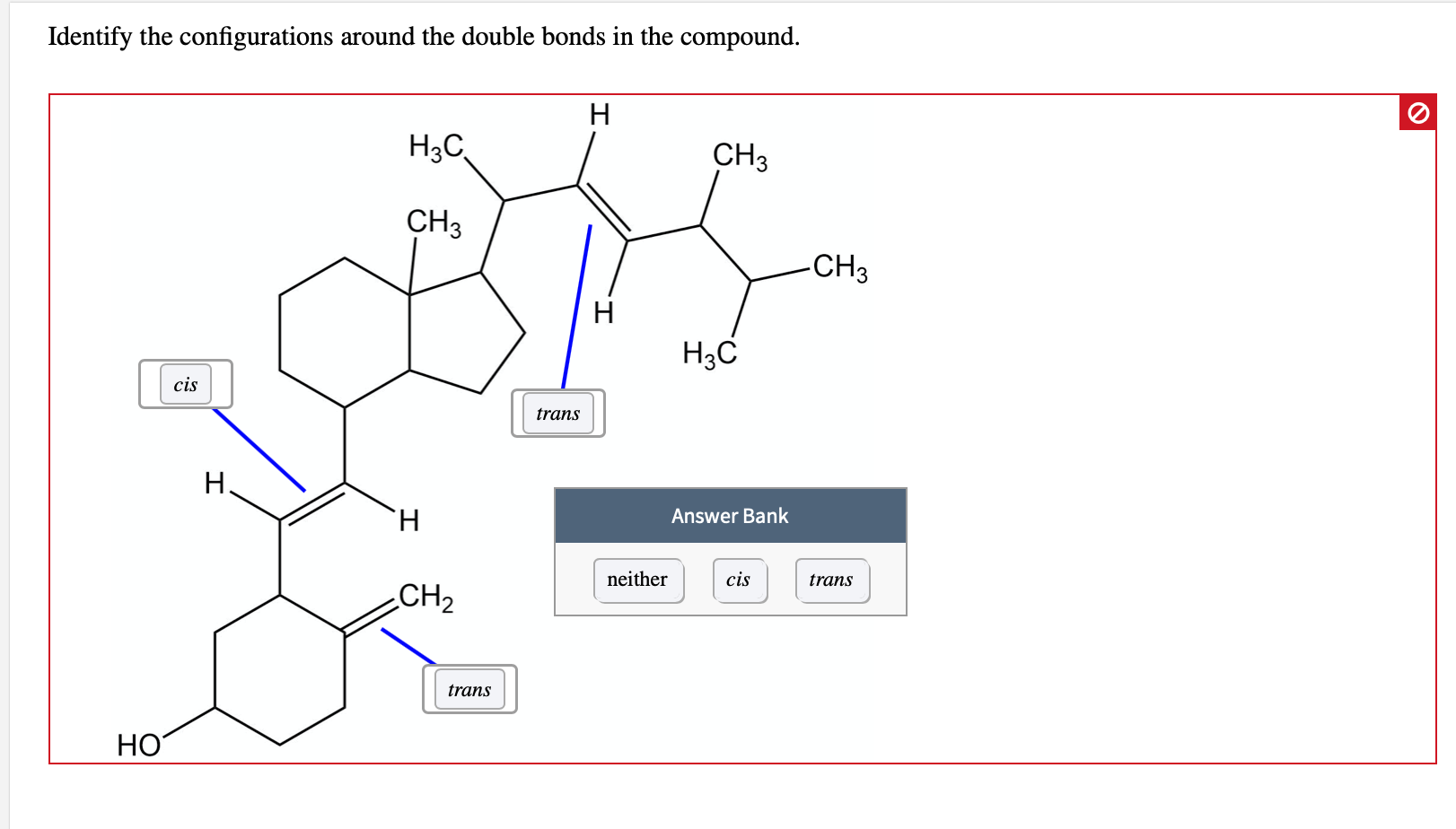
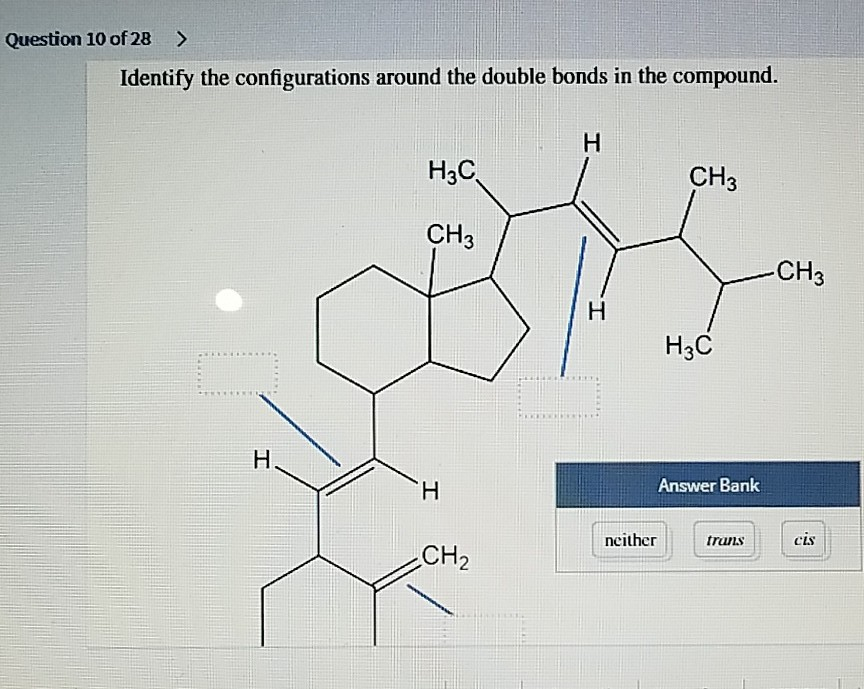
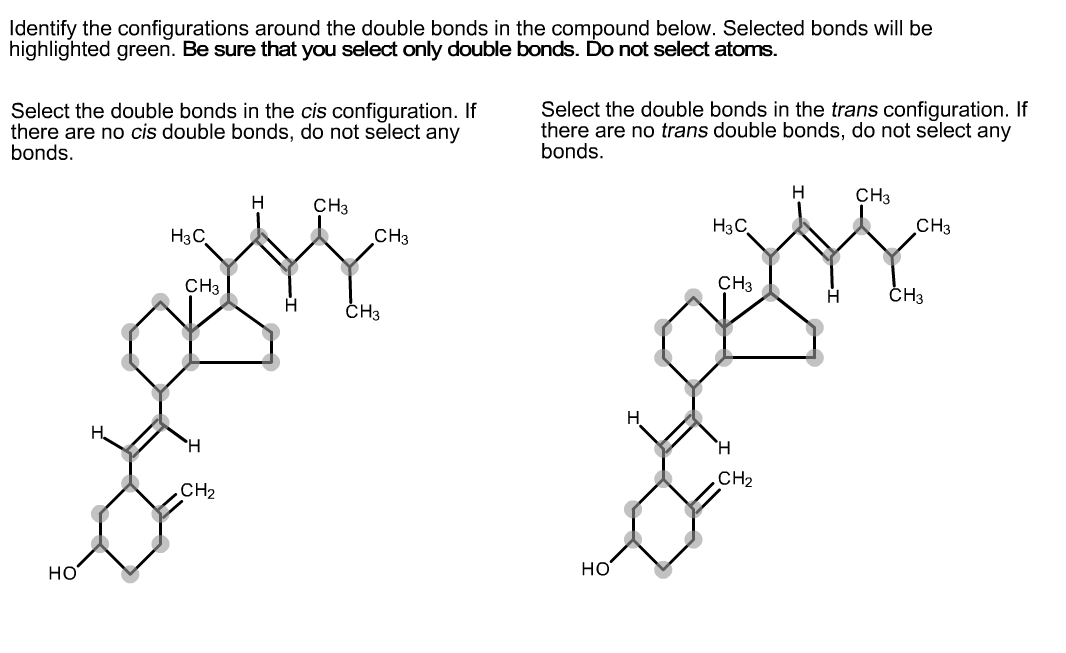
Identify The Configurations Around The Double Bonds
Alright, gather 'round, fellow caffeine aficionados and those just trying to survive Monday. We're about to dive into something that sounds super fancy, like it belongs on a chemistry textbook cover next to a picture of a beaker wearing a tiny graduation cap. But trust me, it's way more interesting than you think. We're talking about the wild and wacky world of double bonds and their configurations. Yeah, I know, sounds like a robot doing yoga. But stick with me, because this is where things get surprisingly… geometric. And a little bit like a choose-your-own-adventure story, but with atoms.
So, imagine you've got two carbon atoms having a serious heart-to-heart. They're so close, they decide to hold hands not once, but twice. That's your double bond. It's like a super-strong handshake that just won't let go. These little duos are everywhere, from the tasty fats in your avocado to the building blocks of life itself. They're the rockstars of the molecule world, always grabbing attention.
But here's where the plot thickens, like a perfectly brewed latte. When these double bonds get their two handshakes going, the atoms they're attached to can't just spin around willy-nilly anymore. They're kind of stuck in place, like kids who've been told to freeze during a game of musical statues. This rigidity is what leads to different configurations. Think of it like having two people holding hands: you can stand facing each other, or you can stand back-to-back. Same hands, same bond, totally different vibe.
Let’s introduce our two main characters: cis and trans. Don't let the fancy names fool you; they're actually pretty straightforward. Imagine our double bond is a fence. The atoms attached to the fence posts are like little garden gnomes. Now, if you have two gnomes on the same side of the fence, that's a cis configuration. They’re chilling together, sharing gossip over the fence. It’s like your best friend standing next to you, not across the street.
On the flip side, if those two gnomes are on opposite sides of the fence, well, that’s a trans configuration. They're practically shouting at each other from a distance. Think of it as your arch-nemesis across the street, giving you the stink-eye. It’s the same fence, the same gnomes, but their spatial arrangement changes everything. It's like the difference between a friendly wave and a territorial standoff.
Now, why should you care about whether your gnomes are on the same side or opposite sides of the fence? Because it matters! These subtle differences can drastically alter how a molecule behaves. For instance, our beloved fats can be cis or trans. Trans fats, those sneaky devils you find lurking in processed foods, are the ones with the atoms on opposite sides. They're like the rigid, inflexible relatives at family reunions – they just don't bend well. Our bodies have a harder time processing them, which is why they’ve been demonized more than a rogue squirrel in a bird feeder. They can mess with your cholesterol levels, turning your blood vessels into a traffic jam on a Friday afternoon.
Cis fats, on the other hand, are generally the good guys. They’ve got the atoms on the same side, making the molecule bend in a more natural, welcoming way. Think of them as the flexible, easy-going friends who are always up for anything. Your body is much happier dealing with these. They’re the reason why olive oil is so good for you, and why that avocado toast isn't secretly plotting your demise.
It’s not just about fats, though. This whole cis-trans thing pops up in all sorts of cool places. Take, for example, a common chemical called an alkene. It's basically a molecule with a double bond. If you have an alkene where the two carbons in the double bond each have two different things attached, then you can have a cis and a trans version. It's like having a party where two people have the same name, but they’re completely different individuals. One might be super outgoing, the other incredibly shy, even though they share a name (or in our case, atoms attached to the same carbon).
Imagine you have two choices for each side of the double bond. Let's say you can have a cherry or a grape. If both carbons have a cherry on the same side of the double bond, it's cis. If one carbon has a cherry on the top and the other has a cherry on the bottom, it's trans. And yes, you can get confusing combinations. It’s like trying to explain your fantasy football picks to someone who doesn’t care about sports – it can get complicated quickly.
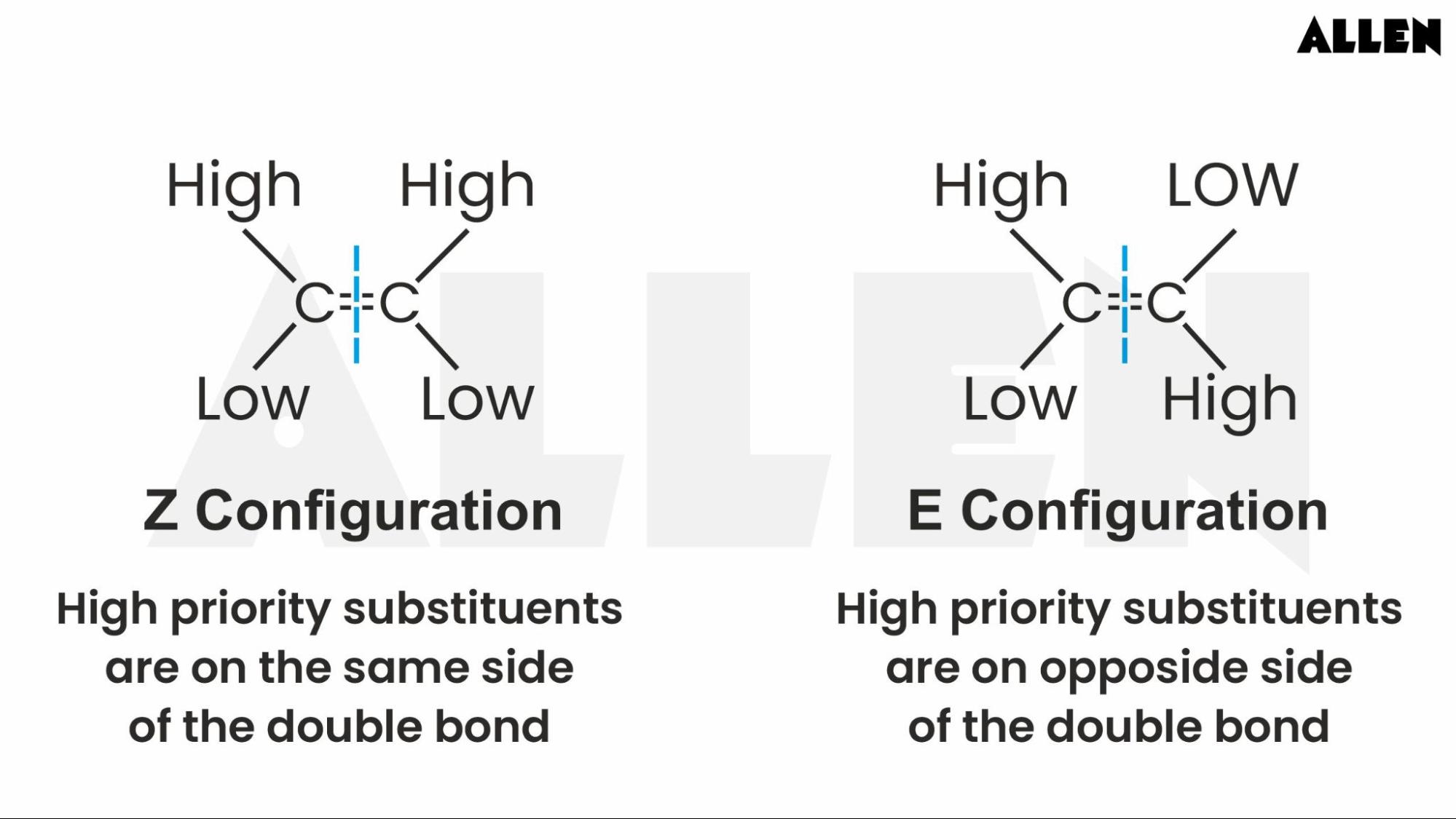
Sometimes, the atoms attached are so different in their 'size' or 'priority' that chemists have their own fancy way of naming these configurations. It’s called E/Z nomenclature. Don't panic! It's just a more precise way of saying cis or trans when things get a bit crowded or confusing. Think of it as the official way to label your gnomes. They assign priorities to the groups attached to each carbon of the double bond, like giving them VIP passes based on their perceived importance. If the high-priority groups are on the same side, it's a 'Z' configuration (from the German word zusammen, meaning together). If they're on opposite sides, it's an 'E' configuration (from the German word entgegen, meaning opposite).
So, even though it sounds like advanced calculus for robots, the cis-trans (or E/Z) configurations are all about spatial arrangement. It’s about whether things are huddled together or spread out. And this simple difference can have profound effects on everything from your health to the properties of materials. It’s like a tiny tweak in a recipe that can turn a bland dish into a culinary masterpiece, or in some cases, a culinary disaster that you’d rather forget.
Next time you're looking at a food label or just pondering the universe over a cup of coffee, remember the humble double bond. It’s not just a strong connection; it’s a geometric decision-maker. It's the difference between a molecule that’s flowing and one that’s rigid. It’s the reason why some things are good for you and others… well, not so much. So, go forth, my friends, and impress your friends with your newfound knowledge of atomic gymnastics. You’re practically a molecular contortionist now. Just try not to bend over backwards explaining it to them. Unless, of course, you're in the cis configuration, then you might just have to.
