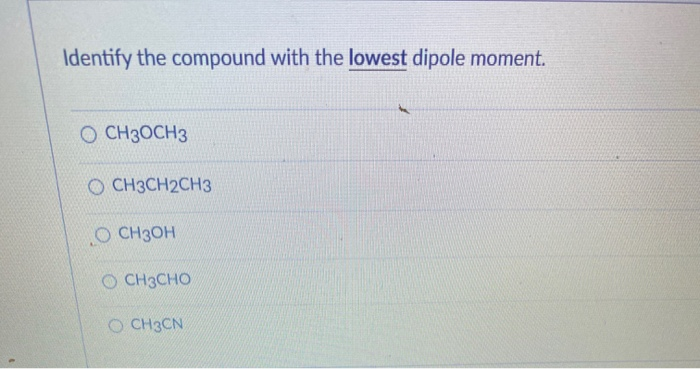
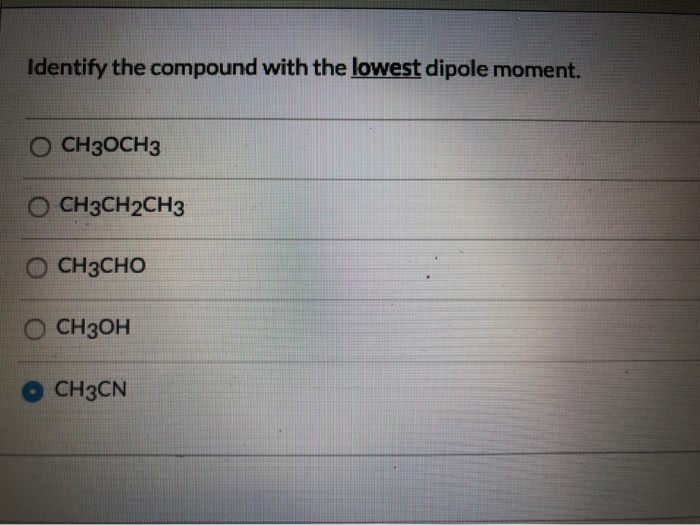
Identify The Compound With The Lowest Dipole Moment
Ever wondered why some liquids feel "slippery" while others feel "sticky"? Or perhaps you've noticed how oil and water just refuse to mix? These everyday observations, and a whole universe of chemical wonders, can often be traced back to a fascinating molecular property called the dipole moment. It might sound like something straight out of a sci-fi movie, but understanding dipole moments is actually incredibly useful and can even be a fun little puzzle for your brain!
The Tiny Dance of Electrons
Imagine a molecule as a tiny, busy dance floor with electrons. In some molecules, the electrons are shared equally between the atoms, like a perfectly balanced dance where everyone has an equal say. These are what we call nonpolar molecules. But in other molecules, one atom is a bit of a hog – it pulls the shared electrons closer to itself. This creates a slight imbalance, with one end of the molecule having a bit more negative charge and the other end a bit more positive charge. Think of it like a dance where one partner keeps pulling the other slightly closer, creating a little bit of a lean. This uneven distribution of charge is what gives a molecule a dipole moment.
The dipole moment is essentially a measure of this charge separation. A larger dipole moment means a bigger tug-of-war for electrons, resulting in a more polar molecule. A smaller dipole moment means the charges are more evenly distributed, making the molecule less polar. And when that dipole moment is practically zero, or so tiny it's almost undetectable, we have a truly special molecule: one with the lowest dipole moment.
Why Does This Matter? The Practical Magic of Polarity
So, why should we care about this electron tug-of-war? Well, the dipole moment is a fundamental property that dictates how molecules interact with each other. This, in turn, affects a huge range of things we encounter daily:
- Solubility: "Like dissolves like" is a famous saying in chemistry, and it's directly related to dipole moments. Polar molecules tend to dissolve in other polar solvents (like water), and nonpolar molecules dissolve in nonpolar solvents (like oil). This is why you can't easily wash off grease with just water!
- Boiling and Melting Points: Molecules with larger dipole moments have stronger attractions to each other. These stronger attractions require more energy to overcome, leading to higher boiling and melting points.
- Intermolecular Forces: The dipole moment is the basis for several types of intermolecular forces, the "glue" that holds molecules together in liquids and solids. These forces influence everything from the viscosity of a liquid to the texture of a solid.
- Biological Processes: Even our bodies rely on polarity! Many biological molecules are polar, allowing them to dissolve in water and participate in the complex chemical reactions that keep us alive.
The Quest for the Lowest Dipole Moment: A Molecular Scavenger Hunt
Finding molecules with the lowest dipole moment is like a fun detective game. We're looking for those molecules where the electrons are shared as equally as possible. This often happens in a few key scenarios:
Firstly, molecules made up of only one type of atom, like diatomic elements such as O2 (oxygen gas) or N2 (nitrogen gas), inherently have no dipole moment. Since both atoms are identical, they pull on the electrons with exactly the same strength, leading to perfect sharing. There's no hogging, no leaning – just pure equality!
Secondly, even with different atoms, symmetry plays a crucial role. Imagine a molecule where polar bonds exist, but the molecule is arranged in such a way that the individual bond dipoles cancel each other out. A classic example is carbon dioxide (CO2). While the bonds between carbon and oxygen are polar (oxygen pulls electrons harder than carbon), the molecule is linear. The pull of the oxygen on the left is exactly counteracted by the pull of the oxygen on the right, resulting in a net dipole moment of zero.

Another fantastic example is methane (CH4). Carbon is slightly more electronegative than hydrogen, meaning the C-H bonds are slightly polar. However, methane has a symmetrical tetrahedral structure. The individual bond dipoles point outwards in different directions, but because of the perfect symmetry, they all cancel out, leaving methane as a highly nonpolar molecule with a very, very low dipole moment.
So, the next time you're looking at water and oil not mixing, or marveling at how easily sugar dissolves in your tea, you can think about the invisible dance of electrons and the fascinating concept of the dipole moment. It's a tiny property with a giant impact on the world around us!
