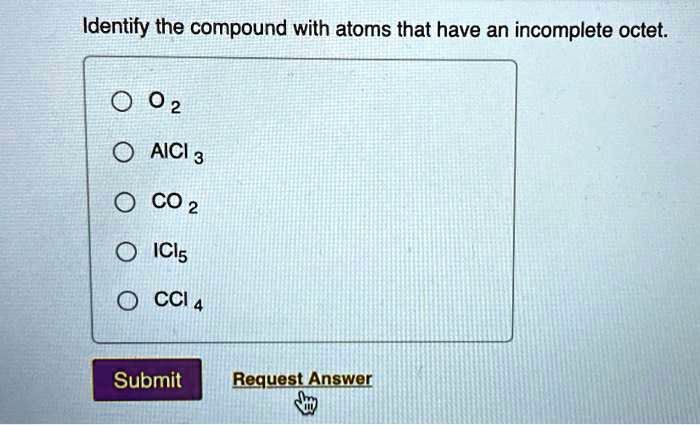
Identify The Compound With Atoms That Have An Incomplete Octet
Hey there, science curious friend! Ever feel like some atoms are a little, well, needy? Like they’re always looking for a friend, or maybe just a snack? Today, we’re diving into a super interesting corner of chemistry: atoms with an incomplete octet. Sounds fancy, right? But trust me, it’s way less intimidating than it sounds, and actually pretty cool once you get the hang of it!
So, what’s the big deal about an “octet”? Basically, atoms are a bit like us – they strive for stability. And for most atoms, especially the ones you see in everyday life, stability means having a full outer shell of electrons. Think of it like a perfectly organized closet with all the hangers filled. The magic number for this full shell is usually eight electrons. This is our famous octet rule. It’s like the ultimate goal, the chemical equivalent of finding your perfect pair of cozy socks.
When an atom has eight electrons in its outermost energy level, it’s generally happy and content. It’s not going to go out of its way to react with anything else. It’s in that sweet spot, that chemical equivalent of a Sunday afternoon nap. Ah, blissful peace!
But, and this is where things get fun, not all atoms play by the same rules. Some atoms, bless their little electron shells, just can't quite get to that magical number of eight. They’re the ones who are still rummaging through the sock drawer, looking for that missing mate. These are our atoms with incomplete octets. They’re the ones who are a bit more eager to mingle, to bond, to, shall we say, share some electrons to get closer to their goal.
Think about it this way: if you’re at a party and everyone’s got a perfect circle of friends (eight friends, in our atom analogy), and you’ve only got three, you’re probably going to be looking around for more people to talk to, right? You’re more likely to initiate a conversation, to try and join a group. That’s exactly what these atoms are doing in the chemical world!
So, How Do We Spot These Eager Beavers?
Identifying a compound with atoms that have an incomplete octet isn't about pulling out a magnifying glass and inspecting individual atoms (though that would be a pretty cool science experiment!). It’s usually about looking at the types of elements involved in the compound. Certain elements are notorious for not quite reaching that octet, especially when they’re forming bonds.
The usual suspects are elements found in the first few periods of the periodic table, particularly those in groups 1, 2, and 13. These guys are naturally a bit electron-deficient. They don’t have as many valence electrons to begin with, making it harder for them to accumulate a full eight in their outer shell through simple bonding.
Let’s break it down a bit. Remember valence electrons? Those are the electrons in the outermost shell, the ones that get all the action in chemical bonding. When an atom forms bonds, it’s usually these valence electrons that are involved in sharing or transferring. The goal, as we’ve established, is a cozy eight.

The Usual Suspects: Metals in the Early Groups
Metals in Group 1 (like lithium, Li) and Group 2 (like beryllium, Be, and magnesium, Mg) are prime examples. These elements tend to lose their few valence electrons to form positive ions. But sometimes, when they form covalent bonds (where electrons are shared), they might end up with fewer than eight electrons around them.
For instance, take beryllium. It’s in Group 2, so it has two valence electrons. When it forms compounds like Beryllium Chloride (BeCl₂), it can end up surrounded by only four electrons. Yes, four! That’s like being at the party with only one other person when everyone else has a whole squad. Beryllium is practically waving a tiny, invisible flag saying, "Anyone wanna share some electron love?"
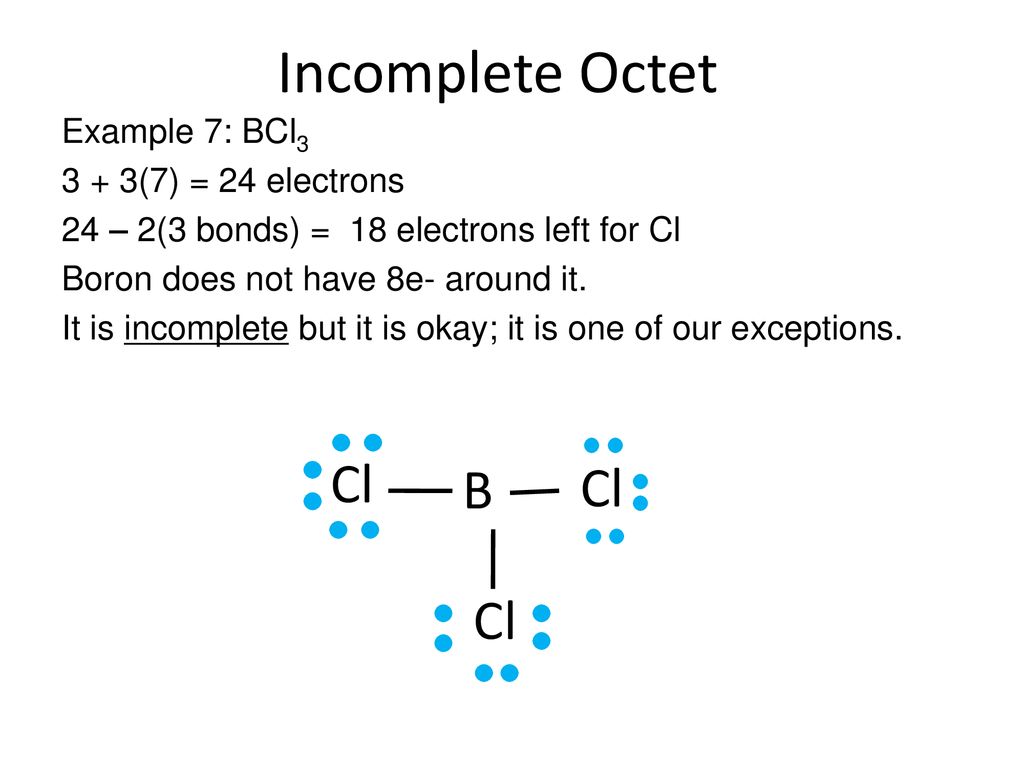
Another one to watch out for is boron (B), found in Group 13. Boron has three valence electrons. In compounds like Boron Trifluoride (BF₃), the boron atom is often surrounded by only six electrons. Six! It’s getting closer, but still not quite there. It’s like having seven out of eight pizza slices – so close, yet so far from that perfect circular satisfaction!
These elements, beryllium and boron especially, are the most common offenders when it comes to incomplete octets in neutral molecules. They’re like the friendly but slightly awkward guests who are always looking for a bit more interaction.
Why Does This Happen? It's All About Energy!
You might be thinking, "But why don't they just try harder to get eight electrons?" Well, it's not just about wanting it; it's about the energetic landscape. Sometimes, forming bonds that result in an incomplete octet is actually the most stable arrangement for that particular atom in that specific compound. It's like finding a comfortable enough position, even if it's not the absolute most ideal. It's good enough for now!

The atom is trying to minimize its overall energy. If forcing an extra bond to get to eight electrons would actually require more energy than it saves, the atom will happily settle for a slightly less crowded outer shell. It’s a cost-benefit analysis, but with electrons!
The Role of Lewis Structures
The best way to visualize this is by drawing Lewis structures. These are like simple diagrams that show the atoms in a molecule and the valence electrons around them. They use dots for lone electrons and lines for shared electron pairs (which represent covalent bonds).
When you draw a Lewis structure and count the electrons around an atom (remembering to count electrons in bonds twice), and you find fewer than eight for elements like Be or B, bingo! You’ve found a compound with an atom that has an incomplete octet. It’s like solving a little chemical puzzle!
For BF₃, for example, you’d draw the boron in the center, bonded to three fluorine atoms. Each bond is a pair of electrons, so that’s 2 electrons * 3 bonds = 6 electrons around boron. The fluorine atoms, being halogens, will typically have their octets filled with lone pairs, but the boron is the star of our incomplete octet show here.
For BeCl₂, you’d have beryllium in the center bonded to two chlorine atoms. Each bond is 2 electrons, so 2 electrons * 2 bonds = 4 electrons around beryllium. Again, the chlorine atoms will likely have their octets satisfied, but beryllium is the one with the electron deficit.
It’s Not Always a Bad Thing!
Now, you might think that having an incomplete octet is like a chemical handicap. But in the world of chemistry, it’s often the very thing that makes these compounds so interesting and reactive! Atoms with incomplete octets are like the energetic youngsters of the molecular world, always ready for the next adventure.
Because they have that “electron gap,” they are excellent electron acceptors. They are positively charged, or at least have a strong tendency to attract electrons. This makes them prime candidates for reacting with things that are electron-rich. They are the hungry ones at the electron buffet!
This characteristic is super important in many chemical reactions, especially in acid-base chemistry. Lewis acids, for example, are species that accept electron pairs. Many Lewis acids contain atoms with incomplete octets. So, these seemingly “incomplete” atoms are actually crucial players in making chemical transformations happen!
Think about it: if every atom was perfectly happy with its eight electrons, the world would be a lot less dynamic! There would be far fewer reactions, far less interesting chemistry happening. These incomplete octet compounds are the spark plugs of many reactions, the catalysts that get things moving.
Common Examples to Keep an Eye Out For
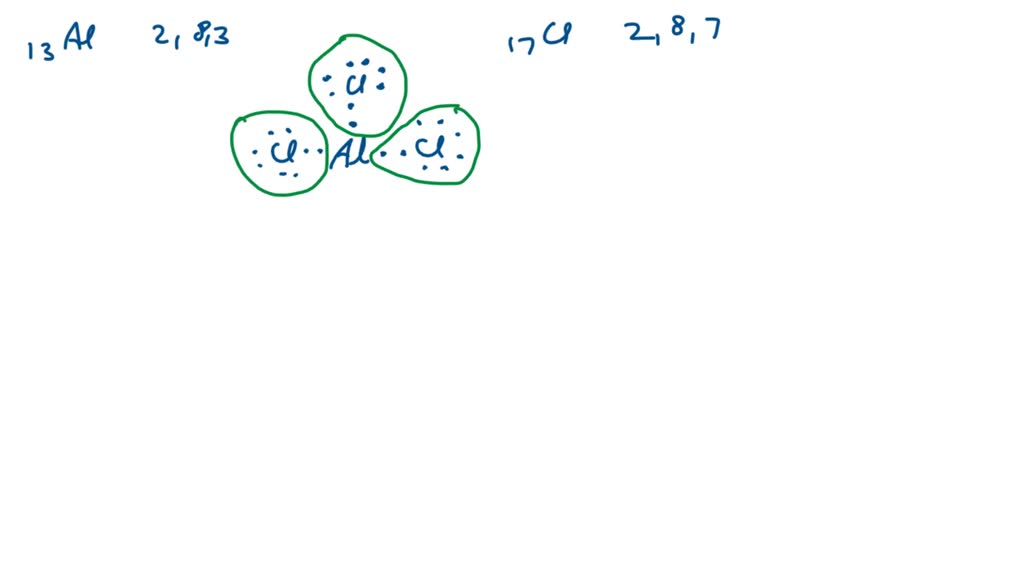
Besides BF₃ and BeCl₂, you’ll often see this phenomenon with other boron and beryllium halides. For instance, Aluminum Chloride (AlCl₃) can also exhibit some characteristics of an incomplete octet, though it's a bit more complex in its behavior and can form dimeric structures. But for a general rule of thumb, focus on boron and beryllium bonded to electronegative atoms like halogens.
The key takeaway is to remember that the octet rule is a guideline, not an absolute law. Nature loves to keep us on our toes, and chemistry is no exception! Some atoms are perfectly content and stable with fewer than eight valence electrons. They might not have a full complement, but they’ve found their own version of stability and reactivity.
It’s a bit like when you’re making a jigsaw puzzle. Most of the pieces fit together perfectly, but sometimes you have a few odd shapes that, while not fitting the “ideal” slot, are still essential for the overall picture. These incomplete octet atoms are those essential, fascinating pieces!
The Fun Part: What Does This Mean for You?
Understanding incomplete octets helps us predict how molecules will behave. It tells us why certain compounds are so reactive, why they can act as catalysts, and how they participate in forming new substances. It’s like having a secret decoder ring for chemical interactions!
So, the next time you’re looking at a chemical formula, try to identify the elements. If you see boron or beryllium, especially in compounds with other non-metals, do a quick mental check. Could that central atom be rocking an incomplete octet? It's a fun little detective game!
And here’s the uplifting part: these atoms, the ones that don't quite fit the standard octet mold, aren't "less than" or "deficient." They are simply different. They have a unique role to play, a special kind of reactivity that makes the chemical world so vibrant and diverse. They teach us that stability comes in many forms, and that sometimes, being a little bit different is exactly what makes you wonderfully, powerfully useful!
So, let’s celebrate these atoms with their incomplete octets! They’re not missing anything; they’re just perfectly designed to be the go-getters, the change-makers, the ones who keep the chemical world buzzing with excitement. Keep exploring, keep questioning, and never stop being amazed by the incredible, often surprising, elegance of chemistry. It’s a universe of tiny particles, each with its own story and its own unique way of shining!
