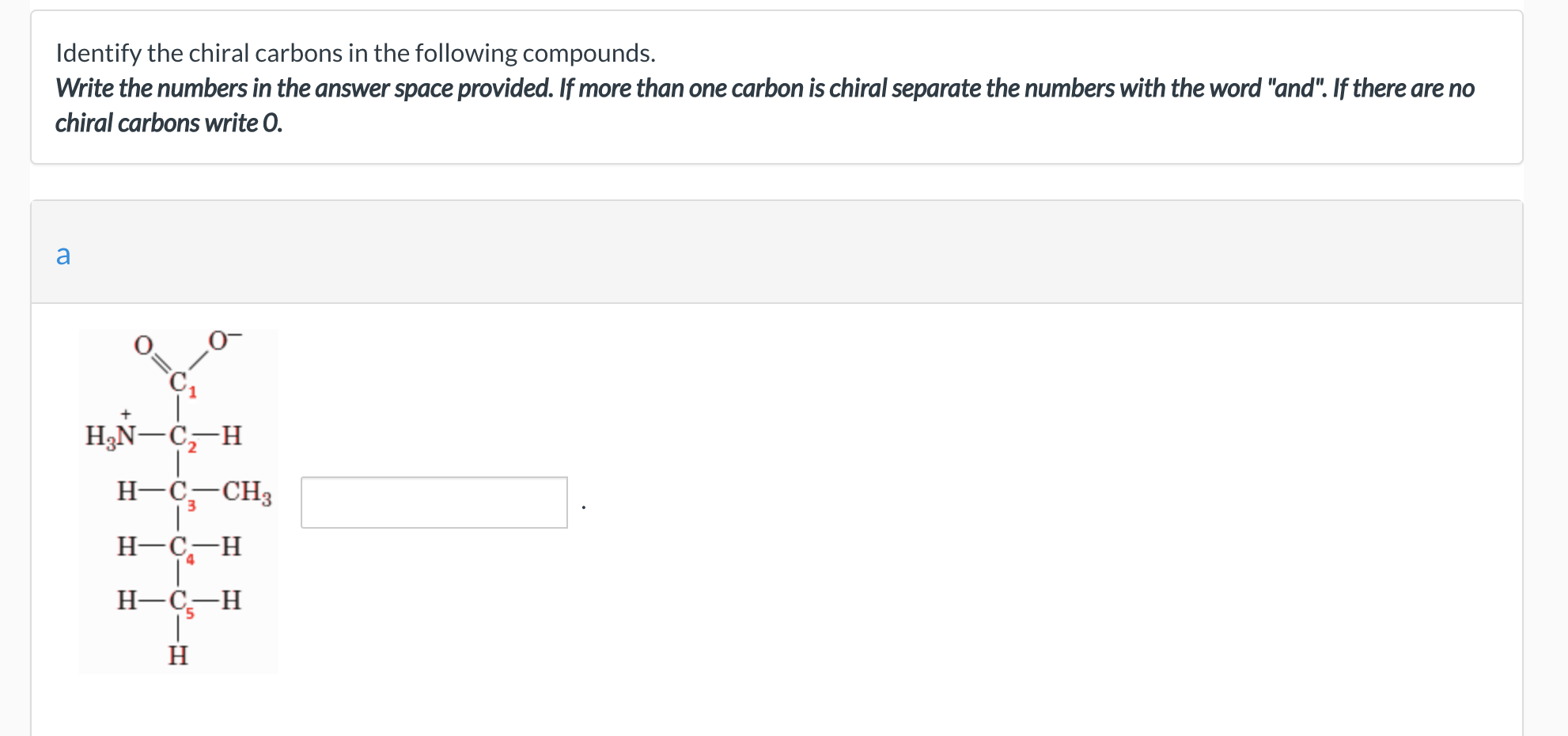
Identify The Chiral Carbons In The Following Compounds
Ever wondered why some medicines are incredibly effective while others have bizarre side effects? Or how our senses of smell and taste can be so incredibly selective? A lot of the magic behind these everyday mysteries boils down to something super cool in chemistry called chirality, and the key players in this phenomenon are chiral carbons. It's like nature's own set of hand-mirror molecules, and learning to spot these special carbons is a fantastic way to unlock deeper understanding of the molecular world around us. Think of it as a detective game for chemists, where you're hunting for a very specific kind of atom that holds the secret to a molecule's 3D personality!
So, what exactly is the big deal with these chiral carbons? Well, the purpose of identifying them is fundamental to understanding how molecules behave, especially in biological systems. Many of the molecules in our bodies – like amino acids and sugars – are chiral. This means they exist in two forms, called enantiomers, which are non-superimposable mirror images of each other, much like your left and right hands. Your body's machinery, which is also made of chiral molecules, can often tell the difference between these two forms.
This is where the benefits really shine. For instance, in the pharmaceutical industry, one enantiomer of a drug might be a powerful medicine, while its mirror image could be inactive or even harmful. The tragic story of thalidomide, where one enantiomer was a sedative and the other caused severe birth defects, is a stark reminder of why this is so crucial. By identifying chiral carbons, chemists can design and synthesize drugs that are pure in their desired form, leading to safer and more effective treatments. Beyond medicine, chirality plays a role in flavors, fragrances, and even the efficiency of pesticides. Being able to pinpoint chiral centers allows scientists to create products that smell just right, taste delicious, or work precisely as intended.
Let's dive into the specifics and learn how to become a chiral carbon detective! A carbon atom is considered chiral if it is bonded to four different atoms or groups of atoms. This is the golden rule! When a carbon meets this criterion, the molecule it belongs to becomes chiral, and that carbon is referred to as a stereocenter or, more specifically, a chiral carbon. It’s this unique arrangement of four distinct attachments that leads to the molecule having a "handedness," existing in its mirror image form.
To get a handle on this, let's look at some examples. Imagine a simple molecule. We’ll start with a basic structure and then explore how adding different groups around a carbon atom can make it chiral. It’s all about what's attached!
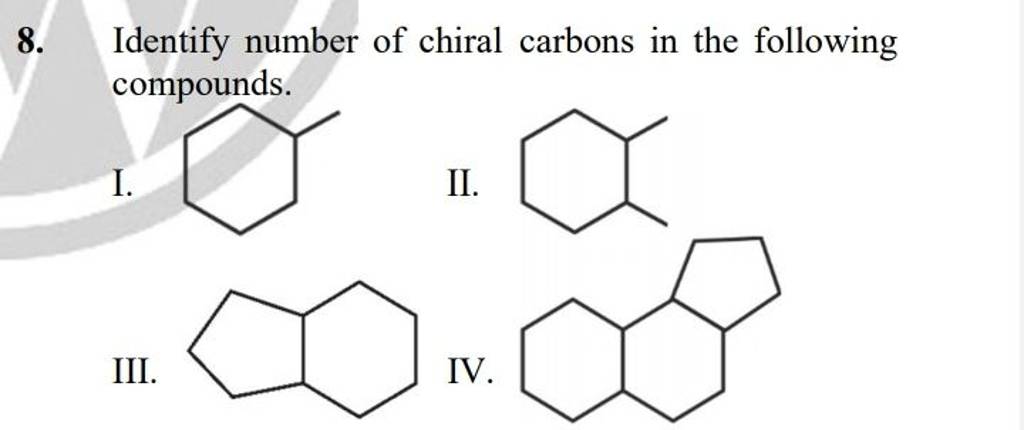
Let's consider a few scenarios to get our detective hats on:
- Scenario 1: The "Easy" Carbon
Picture a carbon atom at the center of a molecule. If this carbon has, for example, two identical hydrogen atoms attached to it, and then two other different groups, it's already disqualified from being chiral. Why? Because it doesn't have four different attachments. Think of it like trying to fit four unique puzzle pieces, but you only have two of one kind – it just doesn't work for creating that specific 3D orientation.
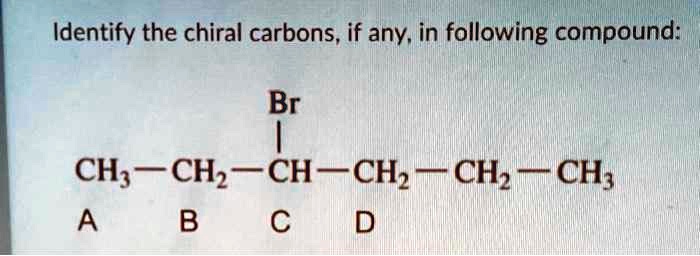
- Scenario 2: The "Almost" Chiral Carbon
Now, let's spice things up. What if a carbon is bonded to three different groups, and then a fourth group that looks similar but isn't quite the same? For instance, one methyl group (CH₃) and one ethyl group (CH₂CH₃). These are different! But if the fourth group was another methyl group, we’d have a problem again. The key is that all four must be distinct.
- Scenario 3: The "Bingo!" Chiral Carbon
This is the one we're looking for! Let's say a carbon atom is bonded to:
- A hydrogen atom (-H)
- A methyl group (-CH₃)
- An ethyl group (-CH₂CH₃)
- And a hydroxyl group (-OH)

The visual aspect is really important here. When you draw these molecules, you can often represent the 3D arrangement using wedges (coming out of the page) and dashes (going into the page). A chiral carbon with four different groups will have a distinct spatial arrangement that is different from its mirror image. It's like a right-handed glove and a left-handed glove – they are both gloves, but you can't wear one on the other hand perfectly!
Identifying these chiral carbons is your first step in understanding the 3D properties of a molecule. It tells you that the molecule has the potential to exist as enantiomers. So, when you're given a chemical structure, the game is to meticulously examine each carbon atom. Look at what each one is directly connected to. If you find a carbon atom connected to four different atoms or groups, you've found a chiral center. This skill is not just an academic exercise; it's a fundamental tool for anyone working with organic molecules, from understanding drug interactions to designing new materials. It’s about seeing the hidden dimensions of molecules and appreciating the subtle yet powerful differences that chirality introduces into the world of chemistry!
