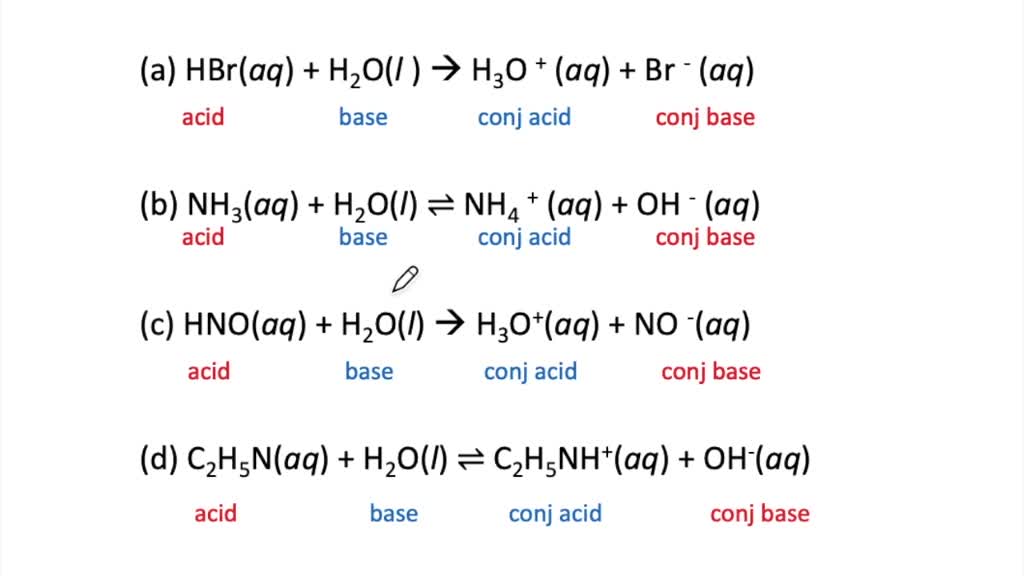
Identify The Bronsted Lowry Acid In This Reaction

Alright, let's dive into the wonderful world of chemistry. It’s a place where things get a bit… proton-y. You might think chemistry is all about explosions and bubbling beakers. Well, sometimes it is! But today, we're focusing on a slightly less flashy, but equally important, aspect. We’re playing a fun little game of "Spot the Acid!"
Imagine a chemical party. Everyone's mingling, bumping into each other, and sometimes, someone’s just giving something away. That's kind of what happens with acids and bases. They're like the ultimate sharers. And our particular sharer today is the Bronsted-Lowry Acid.
So, what is this mysterious Bronsted-Lowry Acid? Think of it as a generous donor. It’s the one in the reaction who’s just itching to get rid of a proton. Yes, a single, tiny, positively charged particle. It's practically begging to be handed over.
Now, this might sound a bit like a weirdly specific hobby. "Oh, I'm just really into giving away protons today." But in the grand scheme of chemical reactions, it's a pretty big deal. This proton-sharing is what makes things happen. It’s the spark that ignites the whole show.
Let's look at our hypothetical reaction. Imagine you have a bunch of molecules hanging out. One of them is just bursting with a proton. It’s practically vibrating with the urge to share. This is our star player, the Bronsted-Lowry Acid.
It's not shy about it, either. It sees another molecule that’s looking a bit… empty. Or maybe it just likes the look of that molecule. Whatever the reason, the Bronsted-Lowry Acid steps up and says, "Here, have a proton!"
So, how do we spot our proton-pusher? You’ve got to look for the molecule that is losing a proton. It’s the one that transforms into something else because it’s given its precious particle away. Think of it as shedding a layer.
This is where things can get a little tricky. Sometimes, a molecule might look like it's doing something else. It might be taking a proton, or it might be doing a little dance. But we’re specifically hunting for the one that’s saying goodbye to a proton.
The other participant in this proton-transfer is the Bronsted-Lowry Base. It’s the one doing the accepting. It’s like the eager recipient of our generous donor’s gift. They form a complementary pair, a chemical dynamic duo.
Our Bronsted-Lowry Acid is the giver, the proton-provider. The Bronsted-Lowry Base is the receiver, the proton-taker. They are the Yin and Yang of this particular chemical reaction.
So, when you see a chemical equation, your mission, should you choose to accept it, is to find the molecule that’s demonstrably shedding a proton. It’s the one that’s left with one less hydrogen atom (because a hydrogen atom is just a proton and an electron, and in acid-base chemistry, we focus on that proton!).
Sometimes, the Bronsted-Lowry Acid is really obvious. It’s like a neon sign pointing to it. Other times, you have to squint a bit. It requires a keen eye for detail and a healthy dose of chemical intuition.

Let's imagine a very simple scenario. You have water. Water is a bit of a chameleon. It can act as an acid or a base. That’s part of its charm, I suppose.
But if water encounters something even more eager to give away a proton, then water itself becomes the acceptor. And the other thing? That’s our Bronsted-Lowry Acid. It’s the proton-giver in that case.
Conversely, if water encounters something that’s just dying to grab a proton, water will step up and offer one. In that scenario, water is the Bronsted-Lowry Acid! See? It’s all about context. It’s like being the life of the party at one gathering and a quiet observer at another.
So, to identify the Bronsted-Lowry Acid, you’re looking for the species that donates a proton. It’s the molecule that’s actively losing an H+ ion. This is its defining characteristic in this particular chemical dance.
Don't get bogged down in other possibilities. We're not interested in what the base is doing right now, or what other transformations are happening. We're laser-focused on the proton-giver. It's the hero of our story, in a very small, proton-sized way.
Think about it like this: if you’re at a bake sale, the Bronsted-Lowry Acid is the person handing out cookies. They are the ones giving something away. The person taking the cookie? That’s your base. We’re looking for the cookie-giver.
This proton transfer is fundamental. It's the engine that drives many chemical reactions. Without acids acting as proton donors, a whole lot of chemistry wouldn't happen. It’s like the opening act that sets the stage for the rest of the performance.
So, next time you’re faced with a chemical reaction and asked to identify the Bronsted-Lowry Acid, just remember: look for the molecule that's feeling generous with its protons. It’s the one that’s making a sacrifice, giving away a tiny piece of itself to facilitate the reaction.
It’s a simple concept, really. Yet, it’s the foundation for so much. Understanding this proton donation is key to understanding how acids and bases interact. It’s the first step in unraveling a chemical puzzle.
And honestly, who doesn't love a good puzzle? Especially one where the main clue is a disappearing proton. It’s like a magic trick, but with science.

So, keep your eyes peeled for that proton-donor. That’s your Bronsted-Lowry Acid. It’s the generous soul of the reaction, making the world of chemistry a more exciting place, one proton at a time.
It's an unpopular opinion, perhaps, but I think the Bronsted-Lowry Acid deserves more recognition. It's the quiet achiever, the unsung hero of the proton exchange.
It’s not always the flashiest molecule. It might not be the one doing the most dramatic things. But without its willingness to part with a proton, the reaction wouldn’t get off the ground. It’s the domino that starts the chain reaction.
So, when you see a reaction, and you're asked to point out the Bronsted-Lowry Acid, just ask yourself: "Who is letting go of a proton?" The answer is your acid. Simple as that. You're basically a proton detective now.
Remember, it's about the donation. It's about the giving. It's the act of releasing that positively charged friend, the proton. That's the hallmark of a Bronsted-Lowry Acid.
And that, my friends, is how you spot the proton-pusher. Go forth and identify those acids! It's a noble quest. A quest for protons and understanding.
