Identify The Bronsted Lowry Acid In The Following Reaction
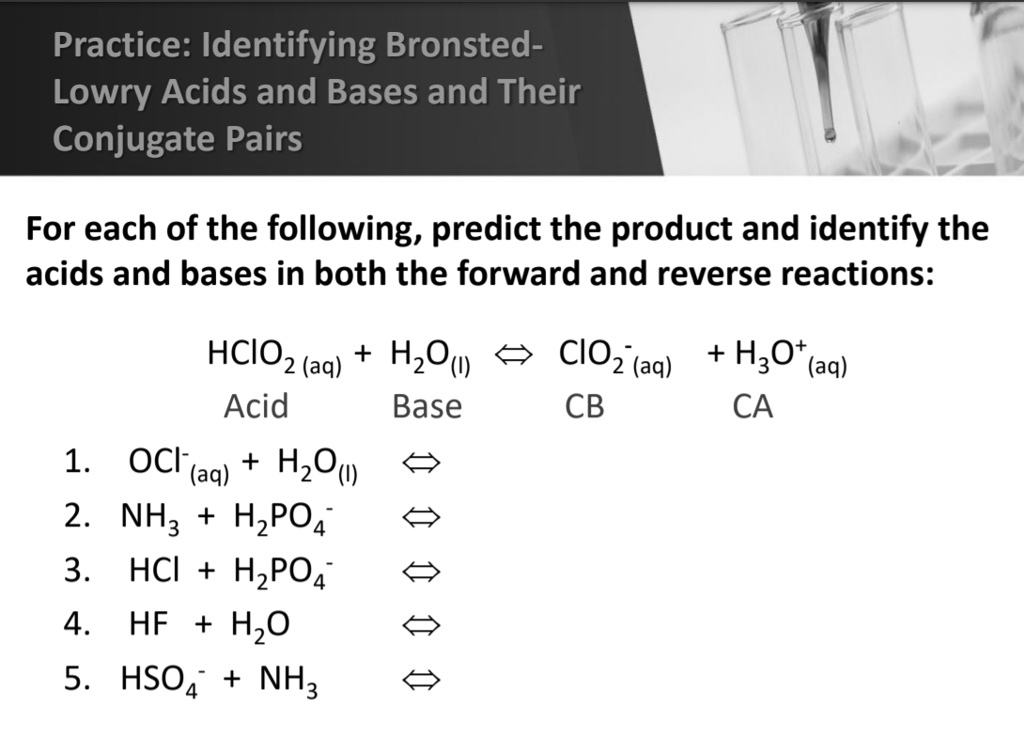
Imagine a bustling party, a really vibrant, energetic get-together where all sorts of characters are mingling. Some are a bit bossy, always trying to hand things out, while others are more eager to snatch things up. It’s a chaotic but ultimately friendly scene, where everyone has a role to play in keeping the party going.
In the grand ballroom of chemistry, we have special guests called acids and bases. They're not your typical partygoers; they have a very specific job. Think of them like the life of the party, always ready to share or accept something important.
Our story today features a rather peculiar duo: a mischievous molecule named H₂O, which we all know as plain old water, and a feisty fellow called NH₃, which you might recognize as ammonia, that strong-smelling cleaner. They've decided to have a little chat, a chemical tête-à-tête, right there at our party.
Now, these two have a unique way of interacting. It’s not about sharing gossip or loaning each other a cup of sugar. Instead, they are all about sharing something much smaller, much more fundamental: a proton.
Think of a proton like a tiny, energetic little guest at the party, carrying a positive charge. It loves to move around, hopping from one molecule to another. And in this particular reaction, there’s a definite dance happening with these protons.
Our friend H₂O, the water molecule, is like a gracious host at this party. It has this proton to spare. It’s feeling generous, perhaps a little bored, and it’s ready to hand it over to someone who’s looking for it.
On the other side of the room, we have NH₃, the ammonia molecule. It’s a bit like someone at the party who’s feeling a little shy, maybe a bit lacking in something. It's got an empty spot, a space just waiting for that tiny, positively charged proton.

So, what happens next is pure chemical magic, a delightful exchange. The H₂O molecule, with its giving spirit, decides to share its proton. It’s like saying, “Here, you look like you could use this!”
And the NH₃ molecule, with its open arms (or rather, its open electron pairs), eagerly accepts this proton. It’s a moment of perfect balance, a harmonious transfer. The party just got a little more interesting.
In the world of chemistry, we have special names for molecules that are so generous with their protons. They are called Brønsted-Lowry acids. These are the donors, the givers, the ones who are always happy to share their protons.
So, when we look at the reaction between H₂O and NH₃, we need to identify who is playing the role of the proton donor. Who is the one readily giving away that little charged particle?
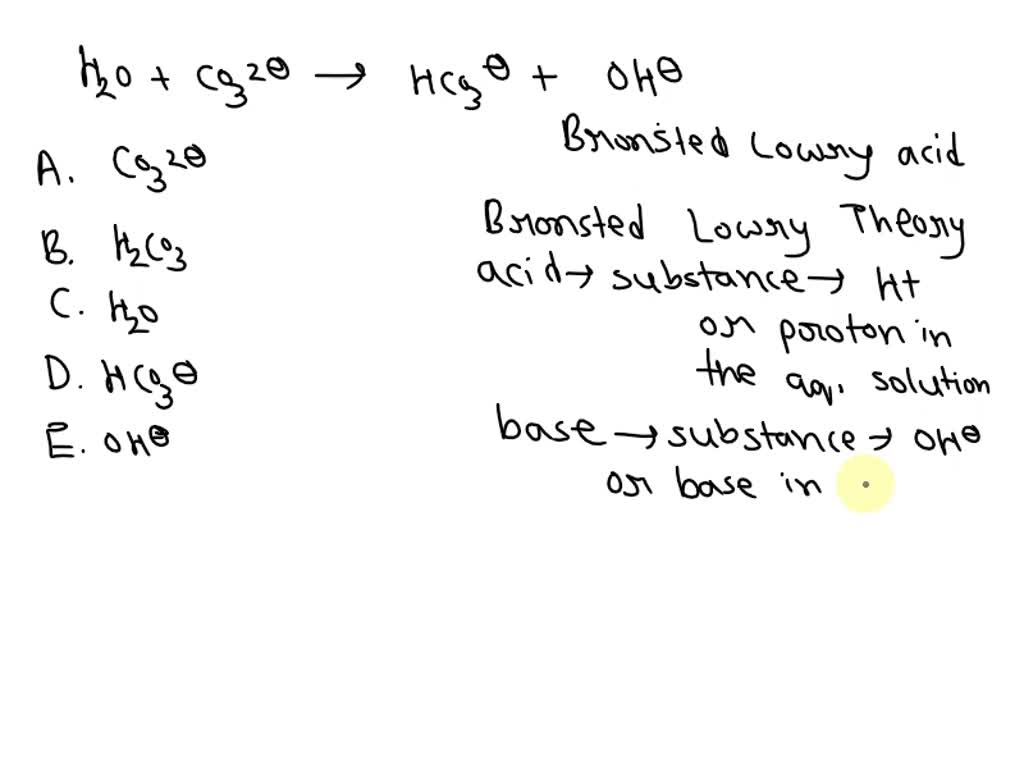
Let’s think about it: H₂O has a proton to offer. It’s got that extra little bit of positive energy it’s willing to part with. It’s the giver in this scenario.
Meanwhile, NH₃ is the one that receives the proton. It's the acceptor, the one that’s happy to take on that extra positive charge. It’s not the donor; it’s the receiver.
Therefore, in this lively little chemical gathering, the molecule that fits the description of a Brønsted-Lowry acid is none other than our familiar friend, H₂O. It’s the one stepping up and saying, “Here, have my proton!”
It’s quite amazing when you think about it. Even something as common as water, which we use for drinking, bathing, and even making our morning coffee, has this incredible ability to act as an acid. It’s a tiny superhero in its own right, always ready to contribute to chemical reactions.
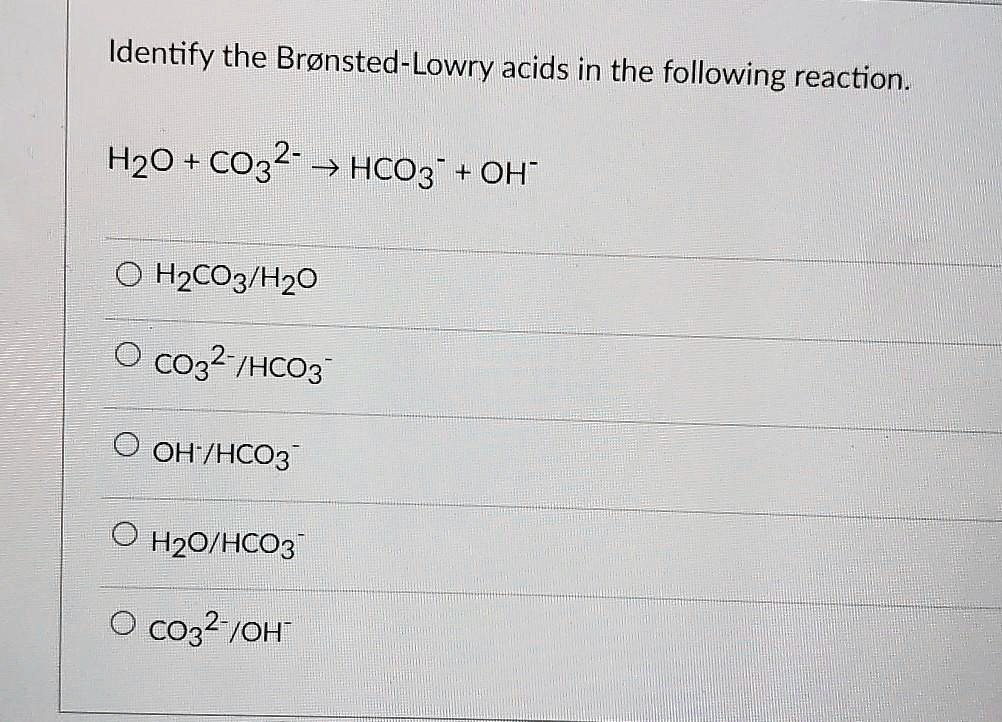
And ammonia, though it has a strong smell and is often associated with cleaning, also plays a crucial role. It’s the yang to water’s yin in this particular exchange, the acceptor that makes the whole interaction possible. Without the acceptor, the donor wouldn’t have anyone to give to, and the party would be a bit one-sided.
This concept, the idea of acids as proton donors and bases as proton acceptors, is a fundamental principle in chemistry. It helps us understand how so many reactions work, from the simplest to the most complex. It’s like understanding the secret handshake of molecules at a party.
So, the next time you see a chemical reaction, especially one involving water, remember the party. Remember the proton hopping around, the generous host, and the eager recipient. It's a miniature drama unfolding, and you now know who the star proton donor, the Brønsted-Lowry acid, is.
It’s a heartwarming thought, isn’t it? That even in the microscopic world of atoms and molecules, there’s this constant exchange, this flow of energy and particles. It's a testament to the interconnectedness of everything, a beautiful dance of giving and taking.
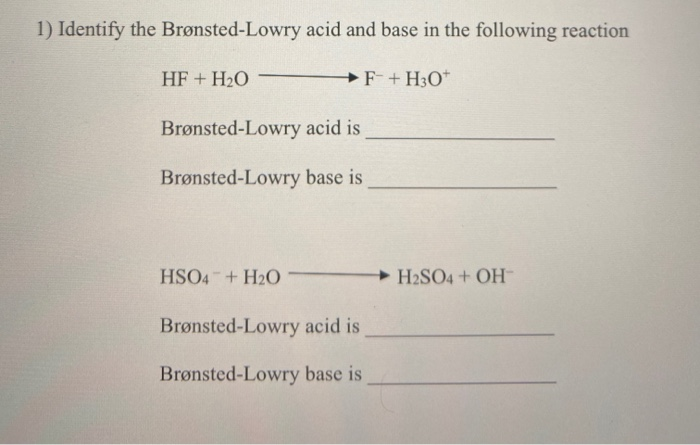
And who knew that a simple reaction between water and ammonia could teach us so much? It’s a reminder that chemistry isn’t just about complicated formulas and beakers; it’s about understanding the fascinating personalities and interactions of the building blocks of our universe.
So, let’s raise a metaphorical glass to H₂O, our friendly neighborhood Brønsted-Lowry acid. It’s always there, ready to share its proton and keep the chemical party going strong. It’s a true hero of the reaction, a generous spirit in the molecular realm.
And in this little molecular dance, the one readily offering up a proton is our star, the Brønsted-Lowry acid. It's the giver, the sharer, the one who makes the magic happen.
It’s a simple idea, but it unlocks a whole universe of understanding. Just by identifying the proton donor, we can figure out so much about how molecules interact. It’s like having a secret decoder ring for the chemical world.
So, if you’re ever presented with a chemical reaction and asked to spot the Brønsted-Lowry acid, just remember our party analogy. Look for the molecule that’s feeling generous, the one with a proton to spare, the one ready to hand it over. You’ll find it, and the world of chemistry will feel a little less intimidating and a lot more fun.
