Identify The Axial And Equatorial Atoms In The Three-dimensional Structure
Ever looked at a bunch of LEGO bricks and thought, "Man, these things have positions"? Well, guess what? Molecules are kinda like that, but way more… energetic and a whole lot smaller. We're diving into the fascinating, and surprisingly relatable, world of how atoms arrange themselves in 3D space. Think of it as the ultimate game of molecular Tetris, where every piece has a specific place and orientation. And just like you wouldn't put a giant LEGO arch sideways on a flat base (unless you're going for a very specific, wobbly artistic statement), atoms have preferred spots. Today, we're talking about axial and equatorial atoms. Sounds fancy, right? But it's really just about up/down and around.
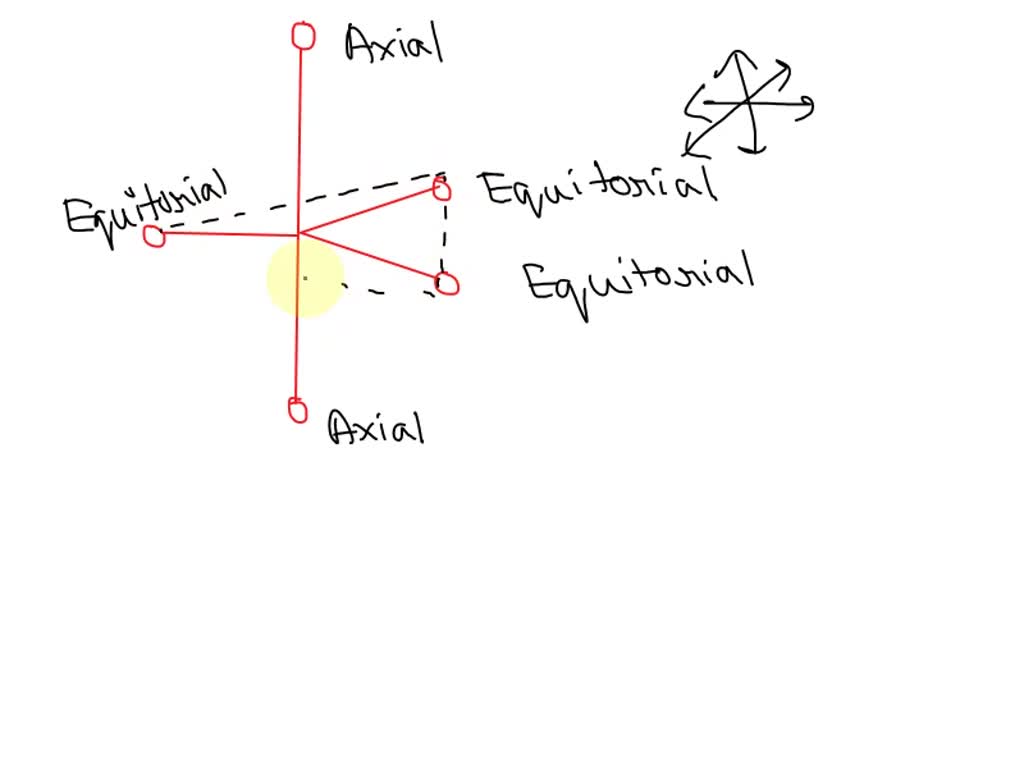
Imagine you're at a ridiculously fancy dinner party. Not just any dinner party, but one where everyone has a designated seat. You've got the host and hostess at the head of the table, right? They're kind of in their own world, looking straight ahead, commanding attention. Then you have everyone else, seated along the sides, facing each other. They're the ones chatting, passing the bread, and generally keeping the party going. In our molecular world, those dinner party VIPs, the ones holding court at the top and bottom, are our axial atoms. They're sticking straight up or straight down, like they're on their own private balcony, surveying the scene.
Now, for the rest of the gang, the social butterflies of the molecular dinner party – they're the equatorial atoms. They're arranged around the middle of the molecule, like guests mingling at a cocktail hour. They're facing outwards, ready for a good conversation (or, you know, a chemical reaction). It’s all about their position relative to the "equator" of the molecule. Think of the Earth: the North Pole and South Pole are axial, while everything around the middle, like your favorite vacation spot, is equatorial. Simple enough, right? We're just giving fancy names to common sense spatial arrangements.
Let's take a common example that everyone can visualize: methane. You know, that stuff that makes some cows sound like they've had one too many beans. Methane (CH4) is basically a carbon atom in the center, with four hydrogen atoms happily attached. In its 3D structure, it forms a beautiful tetrahedron. Now, if we were to imagine ourselves inside that methane molecule, with the carbon atom as the center of our universe, two of those hydrogens would be sticking straight up and down. These are our axial hydrogens. They’re like the two people at the ends of a seesaw, just hanging out there.
The other two hydrogens? They’d be off to the sides, forming a sort of flat plane with the central carbon atom. These are the equatorial hydrogens. They're like the people sitting on the seesaw, ready to push off or come down. They’re the ones in the thick of the action, the real socialites of the methane molecule. So, in methane, we've got two axial and two equatorial hydrogens. It’s a pretty symmetrical arrangement, like a perfectly balanced four-legged stool where two legs go straight up and two go straight out.
But wait, it gets more interesting! Not all molecules are as simple as methane. Take cyclohexane. This is a ring of six carbon atoms. Imagine a hexagonal shape, but it's not flat like a drawing on paper. Oh no, it's all bendy and wiggly, like a rubber band that’s been tied into a loop. It likes to take on shapes called "chair" and "boat" conformations to relieve some internal stress. Think of a comfortable armchair versus a slightly less comfortable rowboat. These shapes are where the axial and equatorial magic really starts to show.
In the chair conformation of cyclohexane, which is the most stable and preferred shape (because, let's be honest, everyone wants to be comfy), each carbon atom has two attached hydrogens. One hydrogen sticks up, and the other sticks down. These are the axial hydrogens for that specific carbon. They're like the little antennas on top of a toy robot, pointing straight up or down from each joint. They’re parallel to each other along the “sides” of the chair, sort of like the legs of a chair pointing in opposite directions.
The other two hydrogens on each carbon atom are in the middle, sticking out to the sides. These are the equatorial hydrogens. They're like the arms of the chair, reaching out horizontally. They're not pointing straight up or down; they're kind of angled outwards, away from the main ring. Imagine each carbon in the cyclohexane ring as a tiny little person standing up. Two of their arms are pointing straight up and down (axial), and the other two are sticking out sideways (equatorial). They're in a constant state of molecular yoga, trying to find their balance.
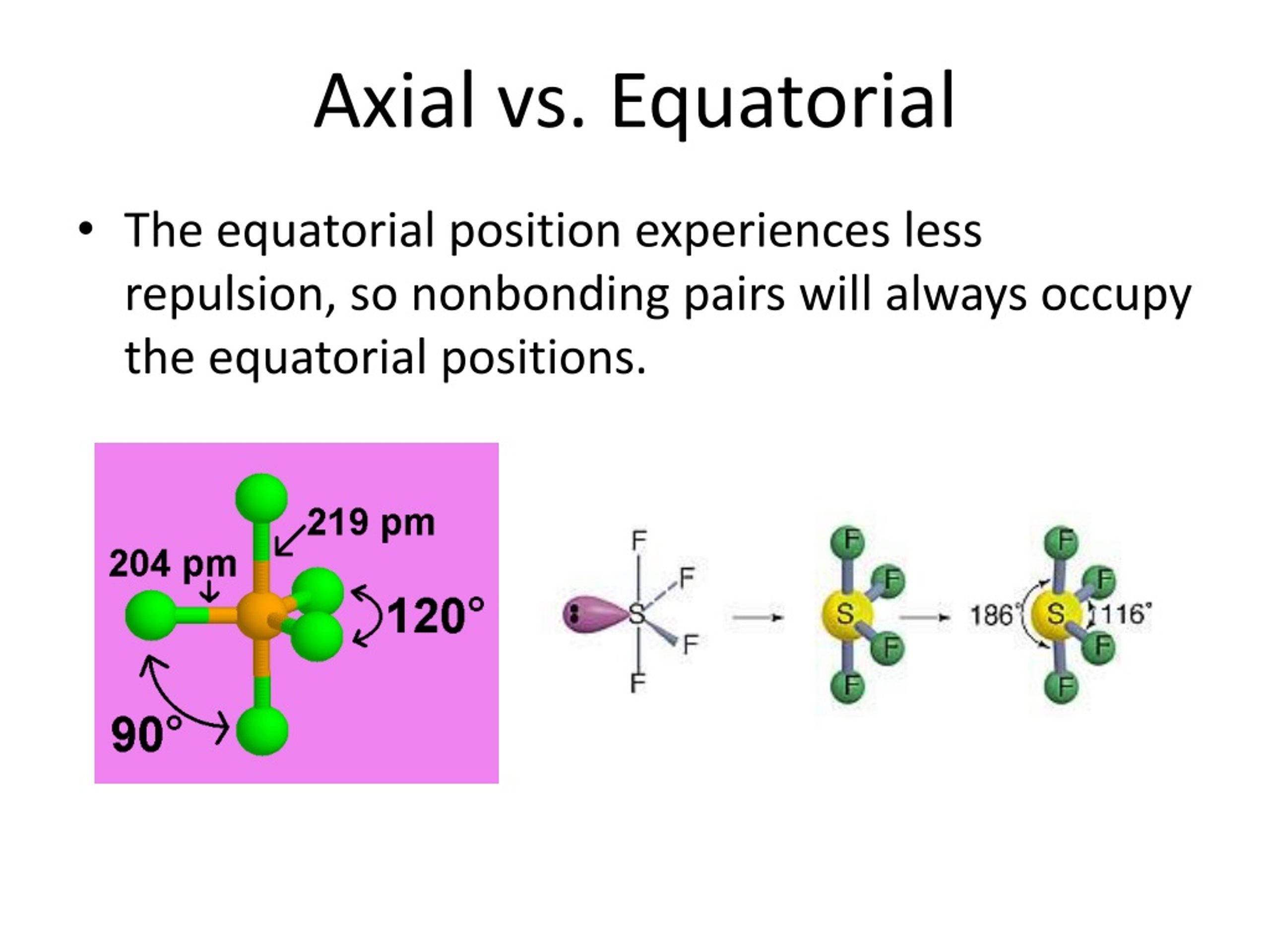
This whole axial and equatorial business becomes super important when we talk about how molecules react. Think of it like trying to park a big truck. It's easier to maneuver if you have plenty of space. Similarly, if a bulky group (like a giant, overstuffed suitcase) is in an axial position, it can bump into other bulky groups that are also in axial positions. This bumping is called steric hindrance, and it’s basically the molecular equivalent of two people trying to walk through a narrow doorway at the same time while carrying identical, unwieldy furniture.
So, these bulky groups often prefer to be in the equatorial position. Why? Because out there, they have more elbow room. They’re not crowding the axial “balcony” and they’re not directly above or below other axial groups. They’re more out of the way, like someone standing in the corner of a crowded room instead of right in the middle of the dance floor. This preference for equatorial positions helps to make the molecule more stable. It’s all about minimizing those awkward molecular bumps and bruises.
Let’s try another analogy. Imagine you have a merry-go-round. The center pole is the axis of rotation. The people sitting on the edge of the merry-go-round are like the equatorial atoms. They’re going around and around, with plenty of space between them as they spin. Now, imagine someone trying to stand on top of the center pole. That’s like an axial atom. They're kind of stuck there, and if the merry-go-round is crowded with other people on the edges, they might not have a lot of room to move their arms freely. If they have something big, like a giant inflatable flamingo, they're definitely going to have trouble holding it in that central position.
When we draw these molecules, especially in organic chemistry, we often use wedges and dashes to show the 3D structure. But when we're looking at specific arrangements like chair conformations, we might draw the ring flattened out a bit to see the axial and equatorial bonds more clearly. Think of it like flattening a crumpled piece of paper to see all the creases. You get a better sense of the overall structure, even though it’s not its natural, relaxed state.

So, how do you identify these axial and equatorial atoms? It’s not like they wear little name tags. You have to look at the geometry of the molecule. For cyclic molecules (those ring-shaped ones), you identify the "up" and "down" bonds relative to the plane of the ring. Those are your axial bonds, and the atoms attached to them are axial atoms. Then, the bonds that stick out to the sides, more or less parallel to the ring itself, are your equatorial bonds, and the atoms attached are equatorial atoms.
For non-cyclic molecules, like that methane example, it’s a bit more about visualizing the overall shape. If you’ve got a central atom with bonds radiating outwards, you might have bonds that are sticking out more directly opposite each other (axial-like) and others that are spread out around the middle (equatorial-like). It's all relative to the molecule's core. Think of the spokes on a bicycle wheel. Some might feel more "up" or "down" if the wheel is tilted, while others are clearly around the rim.
It’s a bit like looking at a family tree. You have the older generations at the top, the founders, so to speak. Then you have the main branches, and then the smaller twigs. The axial atoms are kind of like the people on the highest branches, pointing directly up or down from the main trunk. The equatorial atoms are the ones spreading out, making up the bulk of the tree's canopy. They're the ones you're more likely to bump into when you’re walking past.

This concept is super crucial in understanding why certain chemical reactions happen and why others don't. It dictates how molecules can get close enough to each other to react. If a reactive site is tucked away in a crowded equatorial spot, it might be a bit harder to access than if it's sticking out boldly in an axial position. It’s all about accessibility and steric effects. Imagine trying to offer someone a cookie. If you’re holding it out in an axial position, it’s pretty easy for them to grab. If you’re trying to reach around a bunch of people to offer it in an equatorial-ish way, it’s a bit more of a production.
So, next time you see a chemical structure, don't just see a jumble of lines and letters. Try to visualize that 3D dance. See those atoms in their axial balconies and their equatorial mingling spots. Understand that their positions aren't random; they're a fundamental part of the molecule's personality and its potential for interaction. It’s the hidden architecture that makes chemistry so wonderfully predictable, and sometimes, delightfully surprising. It’s the molecular equivalent of knowing that the couch is the best place to sit when you’re tired, and the table is where you put your food. Just with a lot more electrons and a lot less actual furniture.
Remember, even though we use these terms like "axial" and "equatorial," they're really just descriptive labels for positions in space. It’s about understanding the orientation. Is it pointing straight up or down relative to something? Or is it fanned out around the middle? Once you grasp that basic idea, you're well on your way to understanding a huge chunk of molecular behavior. It's like learning the difference between standing on the top rung of a ladder and sitting on the middle step. Both are on the ladder, but their experience is very different!
So, there you have it. The axial and equatorial atoms: they're not just fancy terms for chemists; they're the up-and-down VIPs and the around-and-about socialites of the molecular world. They dictate how molecules interact, how they fit together, and ultimately, how the world around us functions. Keep an eye out for them, and you’ll start seeing the 3D structure of molecules in a whole new, and hopefully, more smiling-inducing way.
