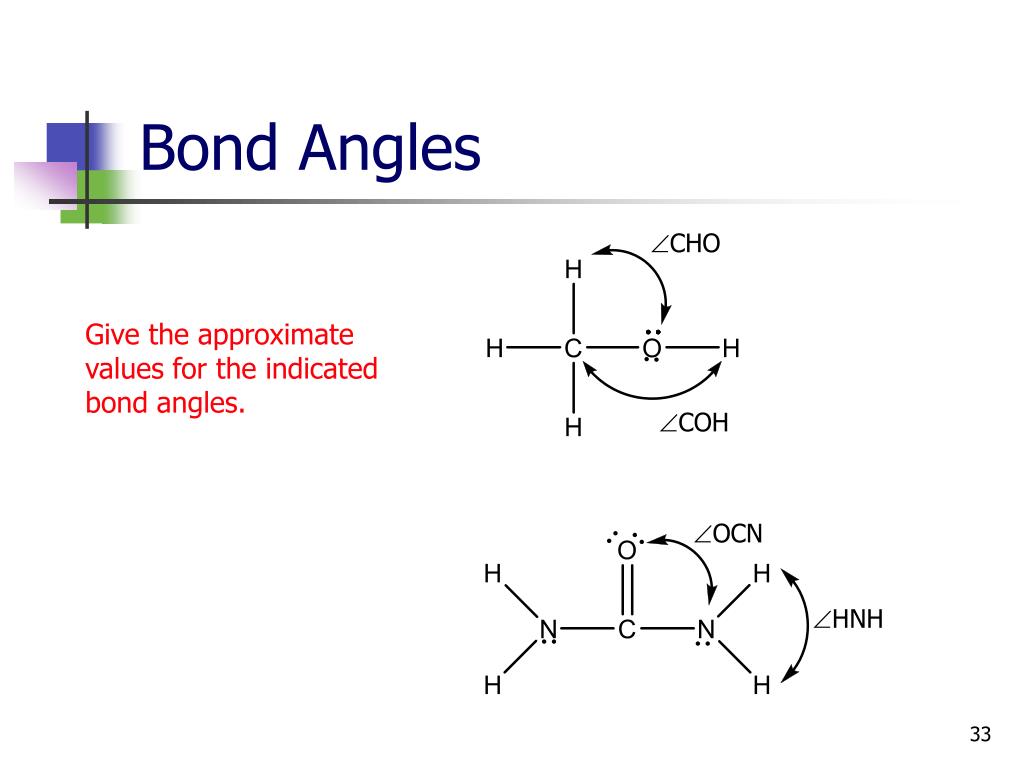
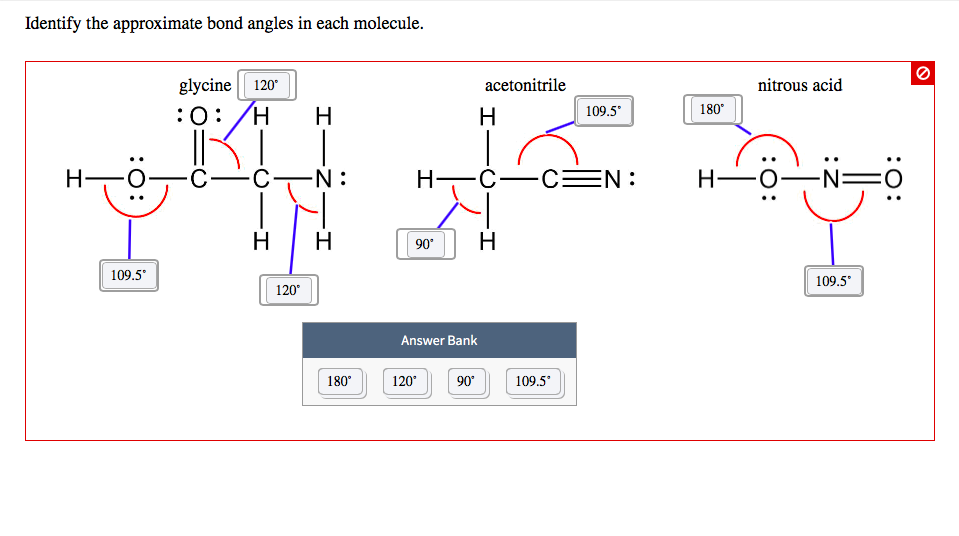
Identify The Approximate Bond Angles In Each Molecule.

Hey there, science curious folks! Ever looked at a water molecule and wondered why it’s not just a straight line? Or maybe you’ve seen those cool 3D models of molecules and thought, “Huh, they’re not all perfectly symmetrical, are they?” Well, you’ve stumbled upon something super neat: bond angles! And guess what? Understanding them is actually way less scary and way more fun than you might think. In fact, it’s like figuring out why your favorite comfy chair is shaped the way it is – it all comes down to how things are arranged!
Think about it like building with LEGOs. You can snap bricks together in a straight line, sure. But to build something sturdy, something that actually works, like a little car or a miniature house, you need to connect them at different angles. Molecules are kind of like that. The atoms are the LEGO bricks, and the bonds are the connections. The bond angle is simply the angle formed by these connections between three atoms. It’s the geometry of the molecule, if you want to get fancy!
So, why should you, a perfectly normal person who probably has better things to do than memorize molecular shapes, even care about bond angles? Well, it turns out these little angles have a huge impact on… well, everything! They dictate how molecules interact with each other, how they dissolve in different liquids, how they smell, how they taste, and even how they can be used in everything from medicines to the materials we use every day.
Imagine trying to shake hands with someone, but your arms were locked at a weird, awkward angle. It wouldn't work very well, right? Molecules are a bit like that. Their shape, determined by bond angles, dictates how they can “shake hands” with other molecules. This is super important for how drugs work in our bodies, for example. If a drug molecule has the wrong shape (because of its bond angles), it might not be able to latch onto the right target in your body, and poof – it’s useless!
Let’s dive into some common shapes and their approximate bond angles. We'll keep it light, I promise!
The Straight Shooter: Linear Molecules
Our first guest is the simplest: the linear molecule. Think of a perfectly straight string of beads. In this case, you’ll have two atoms bonded to a central atom, and they’ll be stretched out in opposite directions. The bond angle here is a tidy 180 degrees. It’s like a perfectly straight arrow!
A classic example is carbon dioxide (CO2). You know, the stuff we exhale? It looks like O=C=O. Those oxygen atoms are perfectly opposite the carbon. It’s a very efficient way to arrange things when you’ve got just two attachments.
Think of a perfectly stretched clothesline. That's 180 degrees. No wobbling, no bending, just pure straightness. It’s the ultimate in minimalist molecular design!

The Happy Triangle: Trigonal Planar Molecules
Now, let’s add a bit more flair. Meet the trigonal planar family. Here, you have a central atom bonded to three other atoms, and they all spread out as far apart as possible in the same plane (like a flat sheet of paper). The best way for them to do this is to form three equal angles.
If you divide a circle into three equal parts, you get 120 degrees. So, the approximate bond angles in a trigonal planar molecule are around 120 degrees. Imagine a three-leaf clover, or the spokes of a bicycle wheel radiating from the center. That’s the vibe!
A common example is boron trifluoride (BF3). The boron is in the middle, and the three fluorines are spread out like a little fan. This shape is quite stable and efficient for packing things in a flat way.
Think of a pizza cut into three equal slices. The angle at the center of each slice is roughly 120 degrees. It’s a balanced and spread-out arrangement.
The Cozy Pyramid: Trigonal Pyramidal Molecules
Sometimes, things aren't perfectly flat. Enter the trigonal pyramidal shape. This happens when you have a central atom bonded to three other atoms, AND you have an extra lone pair of electrons hanging around the central atom. These lone pairs, while not atoms, have electron “clouds” that take up space and push the bonded atoms away from each other.
Instead of being perfectly flat at 120 degrees, the angles get a little squeezed. They’re typically a bit less than 120 degrees, often around 107 degrees. Think of a little three-sided pyramid, like the tip of a party hat.
The most famous example of this is ammonia (NH3). The nitrogen is in the middle, bonded to three hydrogens, and there’s a lone pair on the nitrogen. That lone pair pushes the hydrogens down, creating that pyramid shape. It’s like having three friends sitting around you, and one extra friend hovering nearby, making everyone huddle a bit closer together.
The bond angle in ammonia is really important for its chemical properties, like its ability to dissolve in water and act as a base. It's that slight deviation from perfect flatness that makes all the difference!
The Classic Bent Shape: Bent or Angular Molecules
Now, for a molecule we all know and love (or at least use every day!): water (H2O). Water is a bent or angular molecule. Here, you have a central atom bonded to two other atoms, and importantly, two lone pairs of electrons on the central atom.
Just like in the trigonal pyramidal case, those lone pairs push the bonded atoms closer together. Instead of a straight 180 degrees, the hydrogen atoms in water are at an angle of about 104.5 degrees. It’s not a sharp angle, but it’s definitely not straight!
This bent shape is absolutely critical for water’s amazing properties. It makes water a polar molecule, meaning it has a slight positive charge on one end and a slight negative charge on the other. This is why water is such a great solvent – it can dissolve so many different things! It's like a tiny, two-sided magnet.
Imagine trying to drink from a perfectly straight straw versus a slightly bent one. The bent straw might be more comfortable to hold and direct the liquid. Similarly, the bent shape of water allows it to interact with other molecules in ways that a straight molecule couldn't.
The Four-Square Champ: Tetrahedral Molecules
Let’s move on to a very common and stable shape: tetrahedral. Here, you have a central atom bonded to four other atoms. To spread out as much as possible in three dimensions, these four atoms arrange themselves at the corners of a tetrahedron, which is like a pyramid with a triangular base.
The bond angles in a perfect tetrahedron are all equal and are approximately 109.5 degrees. Think of the legs of a tripod, or the points of a star if you were to connect them. It’s a very symmetrical and stable arrangement.
Methane (CH4), the main component of natural gas, is a classic example. The carbon atom is in the center, and the four hydrogen atoms are at the corners of the tetrahedron. This shape makes methane a very stable molecule.
Imagine holding a perfectly balanced four-legged stool. That’s kind of the stability we're talking about with tetrahedral molecules. It's a solid, well-distributed shape.
Why Does This Even Matter?
So, we’ve seen some basic shapes and their approximate bond angles. But why is this knowledge useful outside of a chemistry lab? Well, it’s the foundation for understanding so much about the world around us!
Drug Design: As mentioned earlier, the precise shape of a drug molecule (determined by its bond angles) is crucial for it to bind to its target in the body. A tiny change in a bond angle can make a drug effective or completely useless, or even harmful.
Materials Science: The way molecules are arranged and bonded affects the properties of materials. Think about the difference between a soft, squishy polymer and a hard, crystalline solid. Bond angles play a role in that!
Biology: DNA, proteins, enzymes – all these incredibly complex biological molecules have specific 3D shapes that are determined by their bond angles. These shapes are what allow them to carry out their essential functions in our bodies.
Smell and Taste: Believe it or not, the shape of a molecule is what allows it to interact with the receptors in your nose and on your tongue, triggering your sense of smell and taste. The same molecule with a slightly different bond angle might smell completely different, or not smell at all!
It’s like having a key that perfectly fits a lock. The shape of the key (the molecule) and the shape of the lock (the receptor) have to match. Bond angles are a big part of that key’s shape!
Don’t Stress, Just Observe!
The main takeaway here isn’t to memorize every single bond angle. It’s to appreciate that molecules aren’t just random collections of atoms. They have specific, often beautiful, three-dimensional shapes, and these shapes are dictated by the angles at which the atoms are connected.
Next time you look at a glass of water, or take a whiff of a flower, or even just marvel at a sturdy piece of furniture, remember that the tiny, invisible world of molecules and their bond angles is silently working its magic to make it all possible. It’s a fascinating peek into the fundamental architecture of our universe!
