Identify The Acid Associated With Each Conjugate Base.
Hey there, my curious friends! Ever find yourself staring at a chemical equation and thinking, "What's the deal with all these bases and their acidy buddies?" Well, get ready to have your mind gently blown, because we're about to dive into the wonderfully wacky world of conjugate bases and their associated acids. No need for a lab coat or a calculator here; we're keeping it light, fun, and totally accessible!
Think of it like this: every base has a secret identity, a shadowy alter ego that's an acid. And every acid has a partner in crime, a base that's just waiting to embrace it. It's a chemical romance, a dance of protons, and it's happening all around us, all the time! Pretty neat, right? It’s like uncovering a hidden subplot in your favorite story – suddenly, everything makes more sense, and it’s way more interesting!
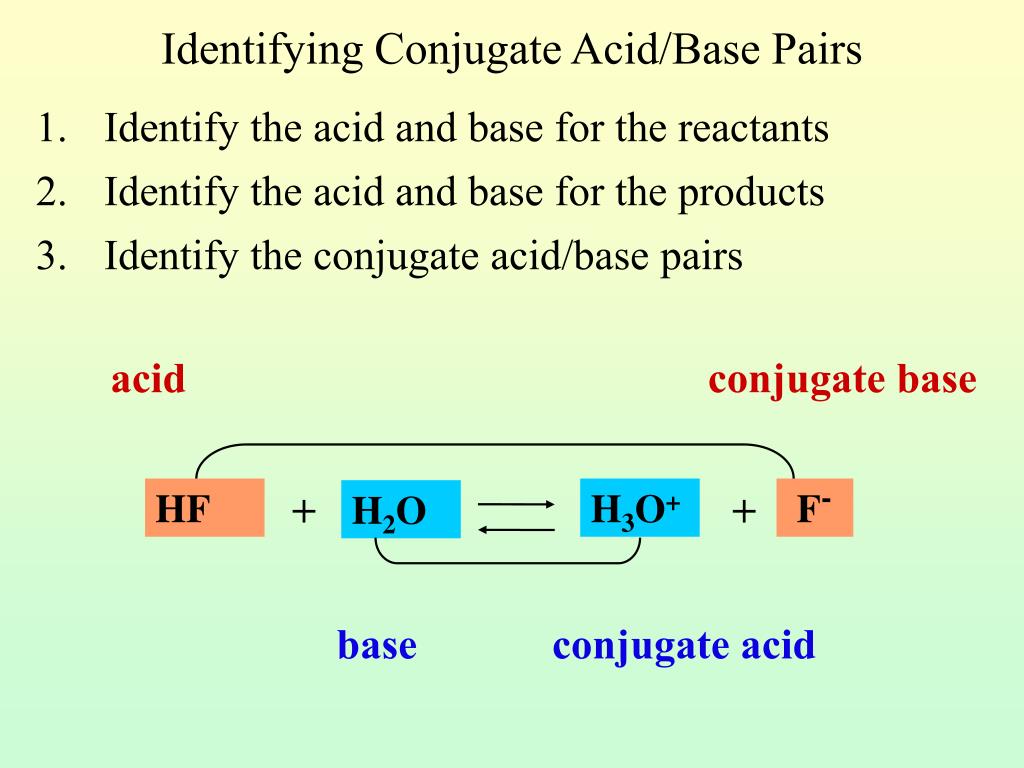
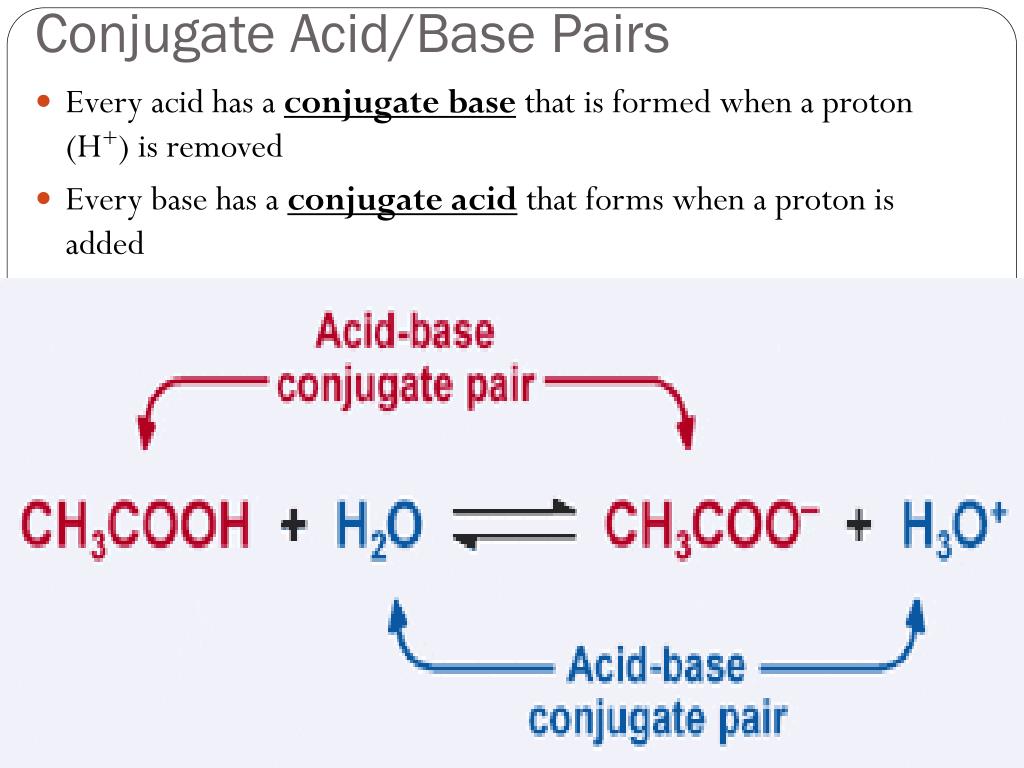
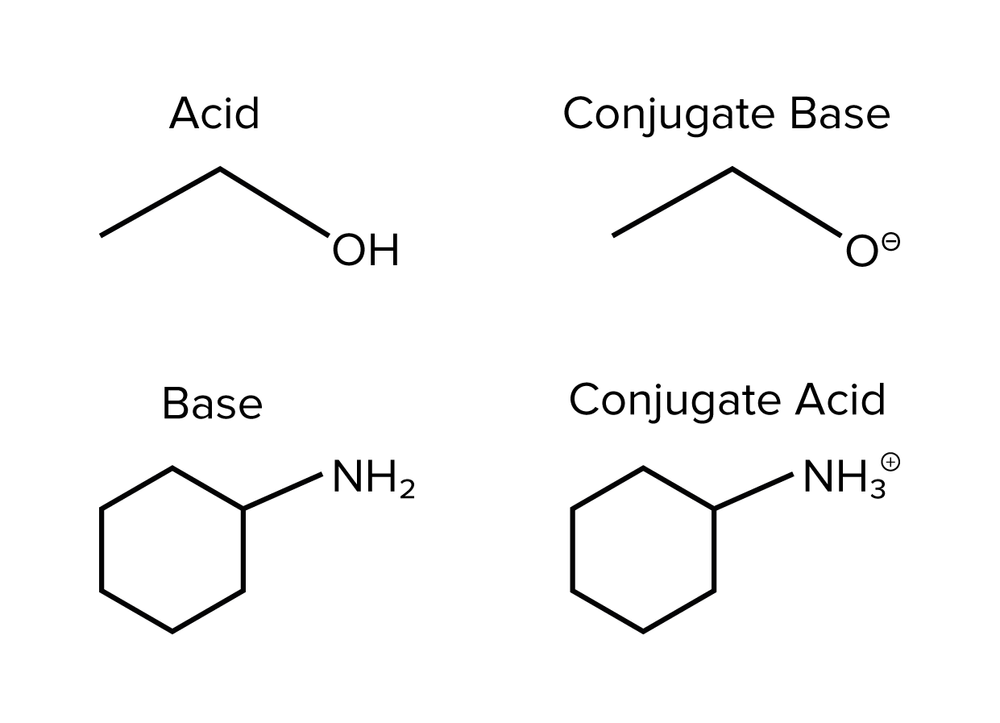
So, what exactly is a conjugate base? Imagine you have an acid, let’s call it a generous giver. Acids, as you might remember, are all about giving away a proton (that's a little positively charged particle, H+). When an acid happily hands over its proton, what’s left behind is its conjugate base. It's like the acid after a really good donation – a little lighter, a little different, but still very much related!
And guess what? That conjugate base is ready to accept a proton back! It's the flip side of the coin, the yin to the acid's yang. So, when you see a reaction happening, and you spot a base, you can bet your bottom dollar (or your mole of molecules!) that somewhere in that mix, or perhaps in a reversed reaction, there's an acid it came from, or an acid it's destined to become.
Let's Get Acquainted with Some Famous Pairs!
Now, to make this truly fun, let’s meet some of the usual suspects. We’ll look at a few common conjugate bases and then unmask their corresponding acids. It’s like a chemical detective game, and you, my friend, are the brilliant sleuth!
The Humble Hydroxide Ion (OH⁻): The Classic Callback!
Have you ever heard of water (H₂O)? Of course you have! It’s pretty essential, wouldn’t you say? Well, water can act as an acid. When water donates a proton, what’s left is the hydroxide ion, OH⁻. See? The OH⁻ is the conjugate base of water! So, the acid associated with OH⁻ is, drumroll please… water (H₂O)! It’s a perfect example of how common things in our lives have these fascinating chemical relationships.

Think about it: every time you see that OH⁻ floating around, you can picture its parent, the noble H₂O, having just performed a selfless act of proton donation. It’s a beautiful cycle, really. This little insight can make even the most mundane chemical observation feel like a secret handshake with nature!
The Chloride Ion (Cl⁻): A Halogen’s Helping Hand!
Let’s talk about hydrochloric acid (HCl). This is a strong acid, meaning it's very eager to give away its proton. When HCl loses its proton, it becomes the chloride ion, Cl⁻. So, the acid associated with the conjugate base Cl⁻ is none other than hydrochloric acid (HCl). It’s like the kid who’s always ready to share their toys, and what’s left after they share is their special toy-sharing identity!
Isn’t it cool how we can predict these things? Just knowing that Cl⁻ is the result of HCl’s generosity opens up a whole new way of looking at chemical reactions. It’s not just a jumble of atoms; it’s a story of give and take!
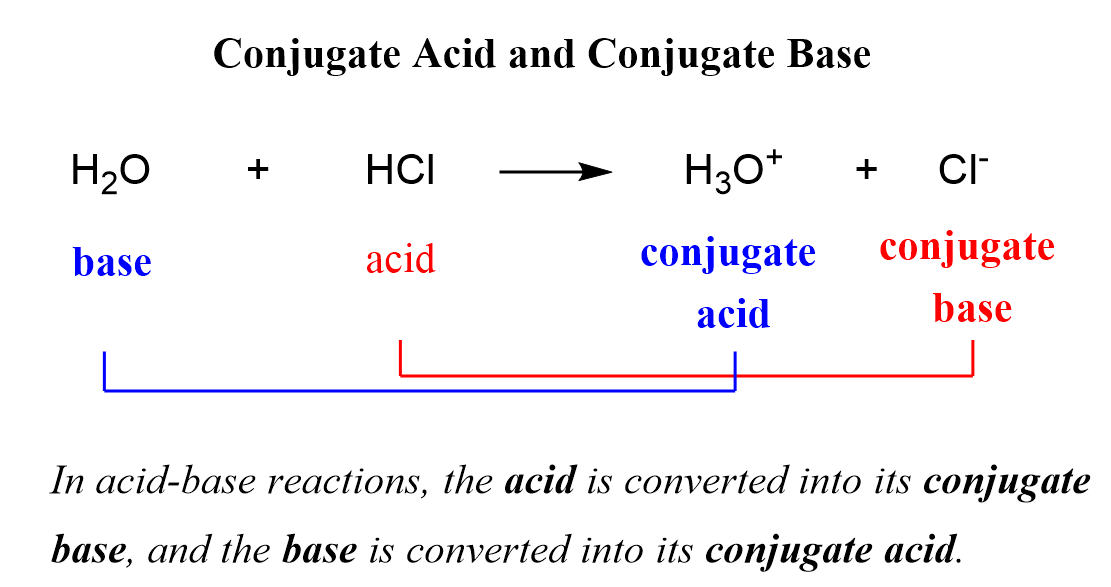
The Acetate Ion (CH₃COO⁻): The Vinegar Connection!
Now for something a little more… culinary! Ever used vinegar? That tangy stuff is primarily acetic acid (CH₃COOH). When acetic acid decides to be an acid and donate its proton, it forms the acetate ion, CH₃COO⁻. So, the acid associated with the conjugate base CH₃COO⁻ is acetic acid (CH₃COOH). See the pattern? It’s like a family tree, but with protons!
This is where it gets really fun. You can start to see these connections in everyday substances. That little bit of fizz in your soda? That's likely related to carbonic acid and its conjugate bases! It’s chemistry subtly at play everywhere you look.
The Bicarbonate Ion (HCO₃⁻): The Breath of Life!
Let’s tackle another familiar one: carbonic acid (H₂CO₃). This acid is crucial in our bodies, helping to regulate pH. When carbonic acid loses a proton, it becomes the bicarbonate ion, HCO₃⁻. So, the acid associated with the conjugate base HCO₃⁻ is carbonic acid (H₂CO₃). This is a super important relationship, both in chemistry and in biology!
Knowing this can make you appreciate the complex balance within your own body. It's not magic; it's chemistry, and you're understanding a part of it! How awesome is that?
Why This Matters (And Makes Life More Fun!)
You might be thinking, "Okay, this is interesting, but how does it make life more fun?" Well, my friends, it’s all about understanding the world around you! When you grasp the concept of conjugate pairs, you’re not just memorizing facts; you’re unlocking a new level of insight.
Suddenly, chemical reactions aren't random occurrences. They become predictable processes governed by these friendly acid-base relationships. It’s like learning a new language, and suddenly the world starts speaking to you in a way it never did before. You can look at a reaction and say, "Aha! I see what's happening here!"
This knowledge empowers you. It helps you understand everything from why certain medications work to how our bodies maintain essential balances. It’s a key to unlocking a deeper appreciation for the intricate workings of the universe. Plus, who doesn't love a little scientific superpower? Being able to understand the hidden chemical drama unfolding everywhere is pretty darn cool.
It adds a layer of wonder to the mundane. That bubbling pot on the stove? The way your laundry detergent works? It all has these underlying chemical stories. And you, my friend, are now privy to a secret chapter!
So, next time you encounter a chemical equation or hear about an acid or a base, take a moment to consider its conjugate partner. What acid does this base come from? What base will this acid become? It’s a fun little mental exercise that builds your understanding and makes chemistry feel less like a daunting subject and more like an exciting exploration.
Keep asking questions, keep exploring, and never underestimate the power of a curious mind. The world of chemistry is full of these delightful connections, waiting for you to discover them. And who knows? You might just find yourself inspired to delve even deeper into the fascinating world of acids, bases, and everything in between. The adventure is just beginning!
