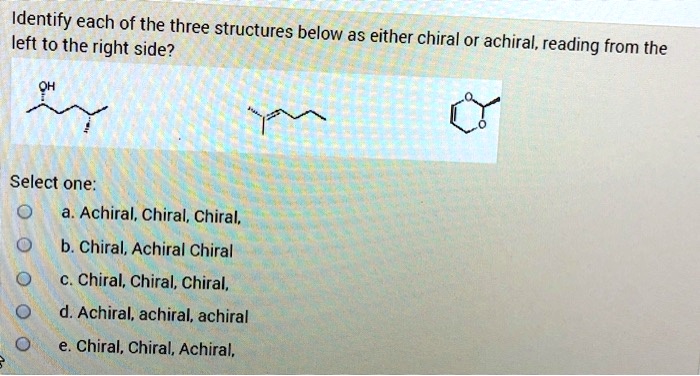
Identify Each Of The Following Structures As Chiral Or Achiral
Ever looked in the mirror and thought, "Hey, I'm pretty unique"? Well, the universe of molecules has its own versions of "unique" – some are perfectly balanced, like a well-made peanut butter sandwich, and others… well, they're a bit more like that one sock that always goes missing in the dryer. We're talking about chirality, folks, and it’s basically the molecular equivalent of having a left and a right hand.
Think about it. Your left hand and your right hand are pretty much identical, right? They have the same fingers, the same palm, the same general shape. But try to stack them perfectly on top of each other, palm to palm, fingers pointing the same way. You can't! They’re mirror images, but they’re not the same. They're like identical twins who refuse to wear the same outfit. This is the essence of chirality – a molecule that’s a non-superimposable mirror image of itself. Achiral molecules, on the other hand, are the perfectly symmetrical ones, like a perfectly round pizza or a flawless sphere. You can flip 'em, twist 'em, and they’ll land exactly the same way. No drama, no fuss.
So, how do we, mere mortals, figure out if a molecule is a lefty or a righty? It’s not like we can put a tiny lab coat on it and send it to a chirality-checking spa. We gotta look at its structure, specifically at the carbon atoms. Carbon is like the busy bee of the molecular world, often forming four bonds. When a carbon atom is bonded to four different things, that’s your red flag, your "caution, chirality ahead" sign. This carbon is called a chiral center. If that carbon is chilling with four identical buddies, or even just three different ones, then the whole molecule is probably just going to chill in the achiral camp. Boring, but stable!
Let’s dive into some examples, shall we? Because, let’s be honest, just talking about it is like describing a delicious meal without actually letting anyone taste it. We need some molecular appetizers!
The Case of the Ordinary Alkane
Imagine a simple molecule like methane (CH4). It's got one carbon atom smack-dab in the middle, and it’s hugging four hydrogen atoms. Now, all those hydrogen atoms are identical, right? So, if you were to try and make a mirror image of methane, you’d end up with… well, you’d still end up with methane. No matter how you spin it, it’s the same. It’s like trying to make a different reflection of a single, perfectly round gumball. It’s achiral. Pure, unadulterated symmetry. No duality, no special powers. Just a simple, straightforward molecule.
Now, let's spice things up a bit with propane (C3H8). It's a chain of three carbon atoms. Let's look at the carbons. The two outer carbons are each attached to two hydrogens and one other carbon. Not four different things. The middle carbon is attached to one hydrogen and two other carbons. Still not four different things. So, propane, in all its chain-like glory, is also achiral. It’s like a perfectly straight, unbent ruler. You can flip it over, and it looks exactly the same. No surprises, no existential crises for propane.
What about butane (C4H10)? Again, it's just a chain of carbons. The end carbons have two hydrogens and a carbon. The inner carbons have one hydrogen and two other carbons. None of these carbons are holding onto four distinct partners. So, butane is achiral. It's the molecular equivalent of a plain white t-shirt – functional, reliable, and utterly devoid of any distinguishing features that would make it a lefty or a righty. It just is.
When Things Get Interesting: The Chiral Centers Emerge
Alright, buckle up, buttercups, because things are about to get a little more… handy. Let's talk about a molecule called 2-butanol. The "2-" tells us something is attached to the second carbon in the chain. And the "butanol" tells us we have a four-carbon chain with an alcohol group (-OH). So, the structure looks something like this:
CH3 - CH(OH) - CH2 - CH3
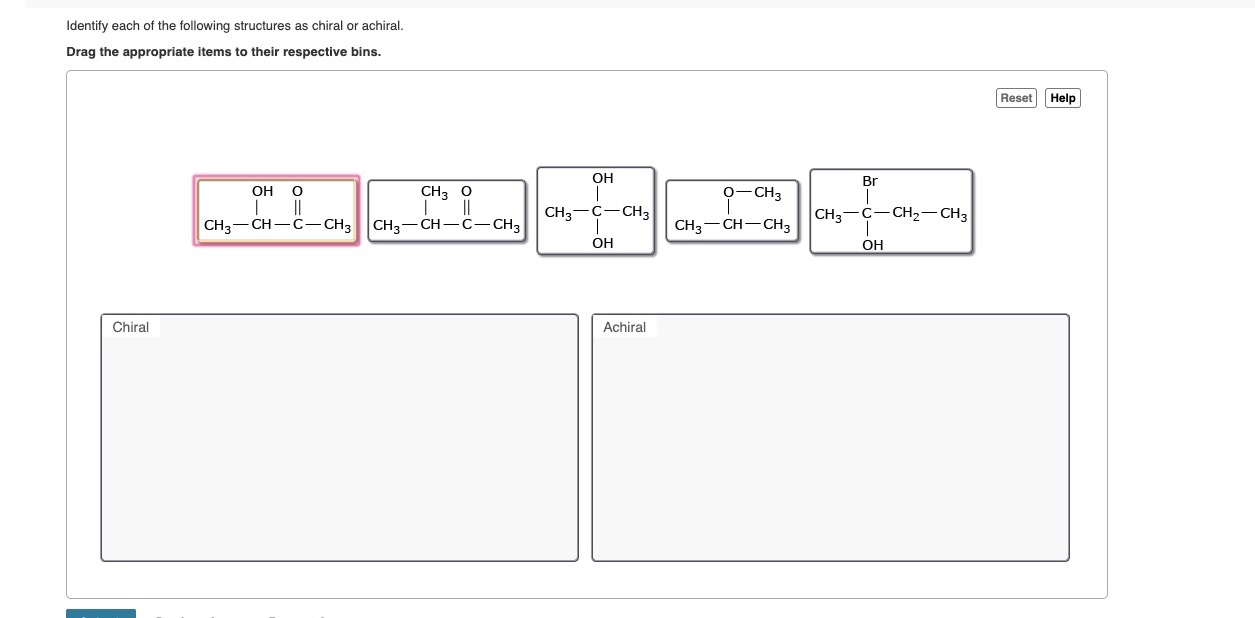
Now, let’s put our detective hats on and examine each carbon atom. The first carbon is a methyl group (CH3). It's got three hydrogens and a carbon. Not four different things. The third carbon (CH2) has two hydrogens and two carbons. Nope, not chiral. The fourth carbon (the end CH3) is similar to the first. Still not the mystery guest.
But, oh ho ho, look at the second carbon! It’s bonded to:
- A hydrogen atom (-H)
- A hydroxyl group (-OH)
- A methyl group (-CH3)
- An ethyl group (-CH2-CH3)
See that? Four completely different attachments! This carbon is our star player, our chiral center. Because this carbon is chiral, the entire 2-butanol molecule is chiral. It has a mirror image that’s not identical to itself. So, you'll have a "left-handed" 2-butanol and a "right-handed" 2-butanol. They'll behave almost identically in most situations, but when it comes to interacting with other chiral molecules, like enzymes in your body, they can be as different as chalk and cheese. This is super important in medicine, for instance. Sometimes only one "handedness" of a drug works, while the other is useless, or even harmful! It's like trying to put a left shoe on your right foot – it just doesn't feel right.
The Ever-So-Slightly-Different Cases
Let's consider another example: 2-chlorobutane. Same four-carbon chain, but instead of an -OH group, we have a chlorine atom (-Cl) attached to the second carbon.
CH3 - CH(Cl) - CH2 - CH3
Let’s check out that second carbon again. It’s bonded to:
- A hydrogen atom (-H)
- A chlorine atom (-Cl)
- A methyl group (-CH3)
- An ethyl group (-CH2-CH3)
Yep, you guessed it! Four different things. So, 2-chlorobutane is also chiral. It’s another example of a molecule that exists as a pair of mirror images, like those reversible jackets that look great on both sides, but are still distinctly different when you turn them inside out.
Now, what if we have a molecule that looks like it might be chiral, but then you zoom in and see a little trick? Take 2-methylpropane, also known as isobutane. It has a central carbon bonded to three methyl groups and one hydrogen.
CH3 - CH(CH3) - CH3
The central carbon has four attachments, but three of them are identical methyl groups. It’s like having four friends, but three of them are named Bob and wear the exact same shirt. You can’t really differentiate them in a way that creates a mirror image difference. So, even though the central carbon is bonded to four things, because three are the same, the molecule is achiral. It’s like a perfectly balanced seesaw. No matter how you look at it, it's the same. It’s the molecular equivalent of a perfectly symmetrical cloud formation – it looks impressive, but there's no inherent "left" or "right" to it.
The Ring Thing: Chirality in Circles
What about molecules arranged in rings? Things can get a bit more complicated, or surprisingly simpler, depending on the ring. Let's consider cyclohexane. It's a ring of six carbon atoms, each bonded to two hydrogens.
Each carbon in cyclohexane has two hydrogens and is bonded to two other carbons. Since those other carbons are part of the same ring and in a symmetrical arrangement, they don't create four different groups around any single carbon. Therefore, cyclohexane is achiral. It’s like a perfectly round Ferris wheel. Every seat is identical, and the whole structure is symmetrical. No matter where you stand, it looks the same.
Now, let’s make it interesting: methylcyclohexane. We attach a methyl group (CH3) to one of the carbons in the cyclohexane ring.
CH3
/
C - C
| |
C - C
\ /
C
Let's look at the carbon that has the methyl group attached. It's bonded to:
- A methyl group (-CH3)
- A hydrogen atom (-H) (which we don't always draw in skeletal structures but is there!)
- Two different paths around the cyclohexane ring. One path goes one way, and the other goes the other way, and because of the methyl group, these two paths are no longer identical.
This carbon is a chiral center! So, methylcyclohexane is chiral. It has a left-handed and a right-handed version. Think of it like a perfectly round clock face, but then you add a little decorative carving at the "12" position. Suddenly, the "12" spot is different from all the others, and the clock as a whole isn't perfectly symmetrical anymore. You can distinguish it!
The Takeaway: It's All About the Different Bits
So, the golden rule, the secret handshake of chirality identification, is this: find your carbon atoms. Look at what’s attached to them. If you find a carbon atom with four completely different groups attached, then congratulations, you've found a chiral center, and the molecule is chiral. It’s a molecule with personality, a molecule that can be two different, non-superimposable things. It’s the molecular equivalent of having a favorite mug that’s slightly chipped on one side – it’s still your favorite, but that chip makes it uniquely yours, and you can’t just flip it over to make that chip disappear.
If, however, all your carbons are rocking identical partners, or even just have a plane of symmetry that allows you to fold the molecule onto itself perfectly, then your molecule is achiral. It’s the reliable, straightforward type. Like a perfectly symmetrical snowflake or a freshly baked loaf of bread – beautiful in its own way, but not prone to existential hand-based dilemmas. It’s just… there. Solid and dependable.
Understanding chirality is like learning a new language for describing the world around us. It helps us appreciate why some things work the way they do, from how your body processes food to why certain perfumes smell the way they do. It’s a little bit of molecular magic, all thanks to the humble carbon atom and its ability to hold onto four different things!
