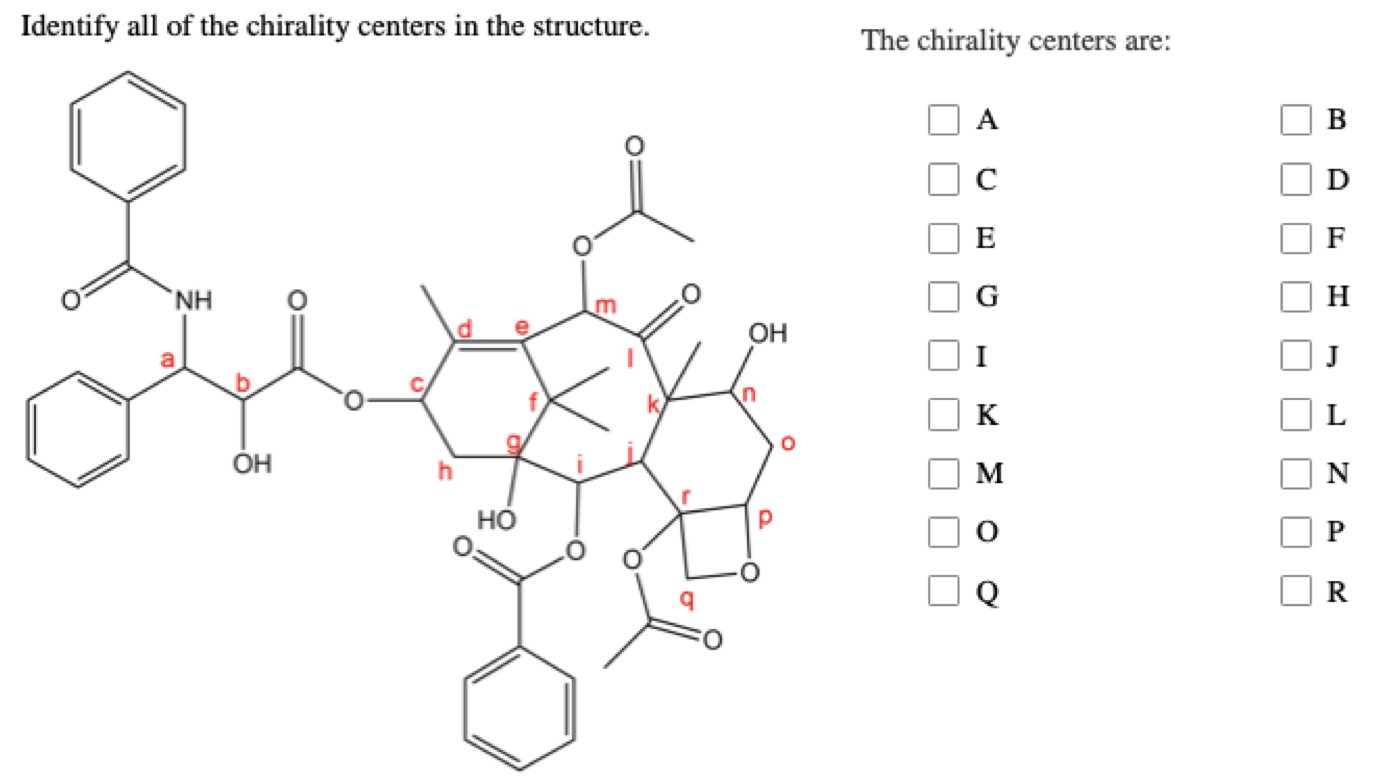
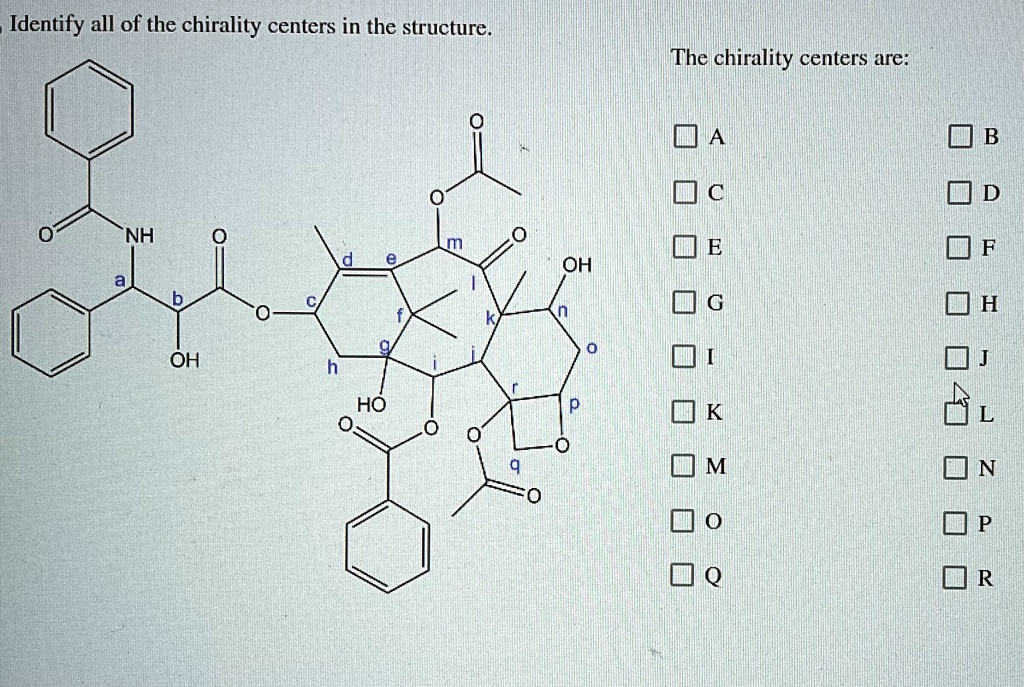
Identify All Of The Chirality Centers In The Structure
Hey there, fellow molecule admirer! Grab your favorite mug, let's dive into the wacky world of chirality. Ever feel like some molecules just have that extra something? That little twist that makes them… well, different? That's where our star player, the chirality center, swoops in. Think of it as the molecule's own personal disco ball, reflecting light in a special way.
So, what exactly is this elusive thing? Imagine your left and right hands. They look pretty darn similar, right? But try to put them on top of each other. Nope! They're mirror images, but they don't quite match up. Molecules can do the same thing. And the atom responsible for this fabulous mirroring act? That's our chiral center, also known as a stereocenter or, if you're feeling fancy, an asymmetric carbon atom. It’s like the universe's way of saying, "Here's a little puzzle for you!"
The main requirement for an atom to be a chirality center is pretty straightforward, really. It’s gotta be bonded to four different groups. Four! Not three, not five, but a perfect quartet of unique attachments. If it’s got any duplicates hanging around, well, then it’s just not that special. It's like showing up to a party with the same outfit as someone else – a bit of a bummer, right? We're looking for that individuality.
Let's break it down, because sometimes seeing is believing, or at least, describing is understanding. Imagine a carbon atom. It's usually the MVP when it comes to being a chiral center. Why? Because carbon loves to make four bonds. It's a social butterfly in the atomic world. Now, if those four bonds are reaching out to completely different molecules, or atoms, or even different parts of the same molecule, then BAM! You've got yourself a chiral center. It's like a four-way handshake with strangers.
Think of it like this: if you can't superimpose the molecule on its mirror image, then congratulations, you've found a chiral center. It's the molecular equivalent of a secret handshake. These little guys are responsible for a TON of stuff in the real world, from how your taste buds work to how medicines affect your body. Seriously, it’s a big deal!
Now, the tricky part, and where things can get a little hairy, is actually finding them in a given structure. It’s like a game of molecular I-Spy. You gotta be meticulous. No shortcuts here, my friends! We need to go atom by atom, checking out each carbon. And not just any carbon, mind you. We're focusing on carbons that are bonded to other things. The lone carbons chilling by themselves? Not our focus for now.
So, let's get down to business. You’ve got a structure in front of you, right? Don’t panic! We’ll tackle it together. First things first, let’s scan for any carbon atoms that look like they're part of the main chain or any branches. Ignore the carbons that are double or triple bonded. Those guys are already committed, they’ve got their dance partners sorted, and they can’t host a four-way handshake. They’re more like the couple already locked in a tango, no room for a new partner.
Okay, so you’ve identified your potential candidates – those carbons with single bonds only. Now, the real detective work begins. For each of these single-bonded carbons, you need to meticulously examine its neighbors. What's attached to it? You’re looking for four distinct entities. And I mean distinct. Not just slightly different, but genuinely different. Even a single hydrogen atom counts as one group!
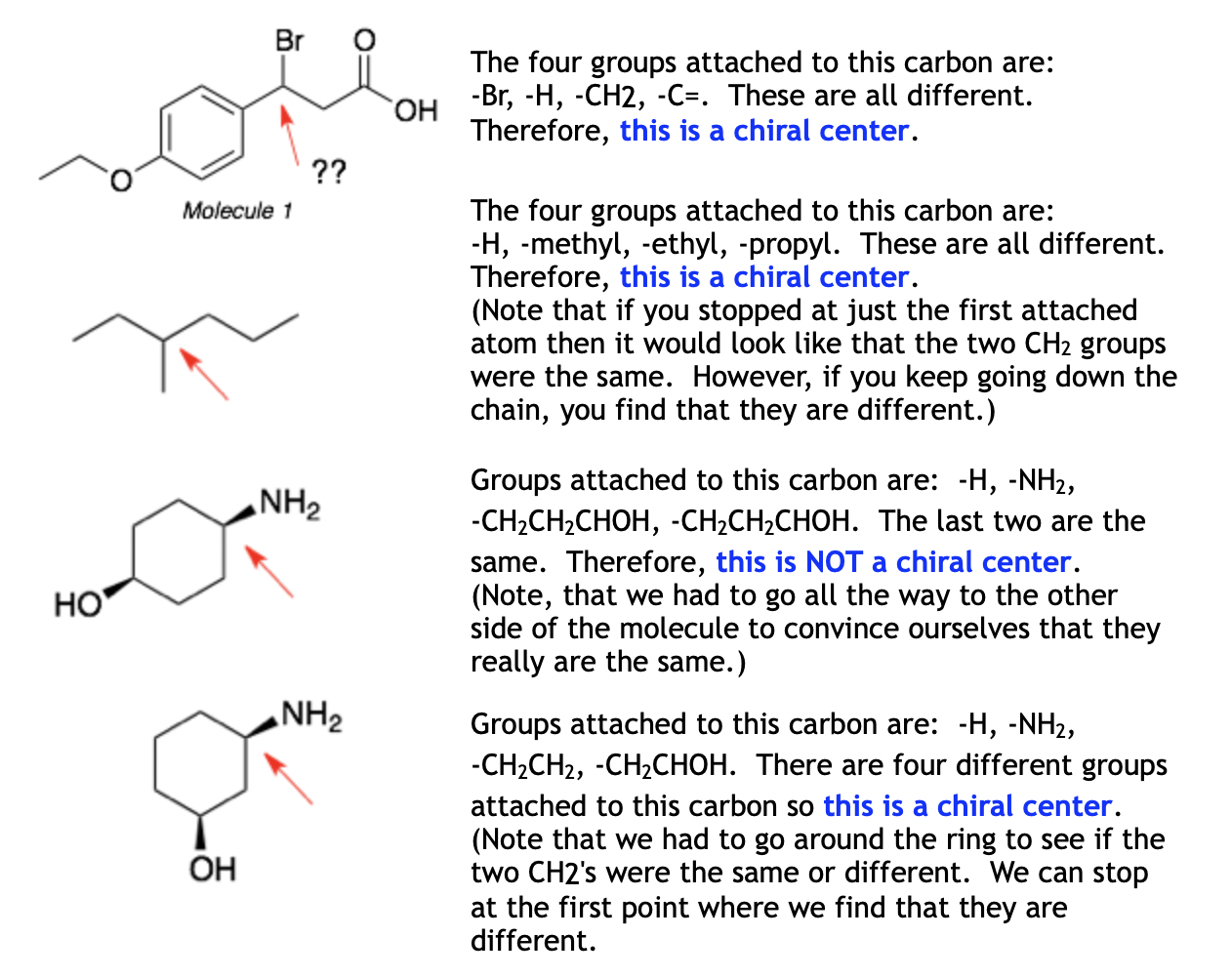
Let’s say you’re looking at a carbon atom. It's bonded to a hydrogen (that's group number one, easy peasy). Then, it's bonded to a methyl group (-CH3). That's group number two. Next, it’s bonded to an ethyl group (-CH2CH3). That’s group number three. Now, what’s the fourth group? If it’s something completely different, like a hydroxyl group (-OH), then congratulations, that carbon is a chirality center! You've found one! High fives all around!
But what if the fourth group looks suspiciously similar to one of the others? You gotta zoom in. Sometimes, the difference is subtle. Imagine you have a chain. A carbon might be bonded to a hydrogen, a methyl group, and then a longer carbon chain. You need to trace that chain out. Is the entire chain on one side different from the entire chain on the other side? That’s the question.
For instance, if a carbon is bonded to a hydrogen, a methyl group, and then a -CH2-CH2-CH3 chain, and on the other side, it's bonded to a -CH2-CH3 chain, then those two chains are different. One has three carbons, the other has two. So, that carbon would be a chiral center. See? It’s all about the detailed comparison.
What about those carbons in rings? Oh, they can be sneaky little devils! For a carbon in a ring to be a chiral center, the path around the ring in one direction has to be different from the path around the ring in the other direction, starting from that carbon. It's like walking a tightrope – the scenery on your left has to be different from the scenery on your right. If you come back to the same atoms and groups no matter which way you go, then it's not chiral. It's like walking in a perfect circle; you always see the same things.
Let’s take a cyclohexane ring for example. If all the carbons have two hydrogens and are identical in their surroundings, they’re not chiral. But if you start hanging stuff off that ring, things get interesting. If a carbon has a hydrogen, and then is connected to two different chains going around the ring, then it could be a chiral center. You gotta trace those paths carefully. Are the substituents along each path different? Are the lengths of the paths different before they rejoin or lead to another different part of the molecule?
Sometimes, you'll find carbons with a hydrogen, and then the rest of the molecule is the same on both sides. For example, if a carbon is bonded to a hydrogen, and then to two identical ethyl groups (-CH2CH3). In this case, the carbon is NOT chiral. It's got two identical friends, and that just won't do for our chiral club. It’s like trying to get into an exclusive club with a plus-one who is your twin. Not different enough!
And don't forget the functional groups! Sometimes, the attached groups themselves might contain chiral centers. This can get complicated, but for now, we're focusing on the central atom being attached to four different things. If one of those "things" is, say, a whole other molecule with its own chiral center, that still counts as one distinct group for the original carbon we're examining. It's like saying you brought a friend to a party; that friend is one entity, even if they have their own amazing stories.

Here’s a little tip: when you’re looking at a carbon, start by counting the hydrogens attached. If there are two or more hydrogens, that carbon cannot be a chiral center. Why? Because you already have two identical groups (the hydrogens)! So, you can immediately dismiss those. This saves you a lot of brainpower. Think of it as a quick elimination round.
Then, look for carbons with just one hydrogen. These are your prime suspects! Now you just need to check the other three groups. If they’re all different, then jackpot! You’ve found a chiral center. It’s like finding a four-leaf clover, but way more scientifically significant.
What about carbons with no hydrogens attached? These are often found in double or triple bonds (which we’ve already ruled out) or as part of aromatic rings, or carbons that are bonded to four other carbons. For these, you have to carefully examine the connectivity of everything attached. Are the carbon chains branching off in different ways? Are they leading to different functional groups? You really have to trace the whole path.
Let’s talk about symmetry. Molecules with a plane of symmetry are achiral. This is like a mirror reflecting a perfect twin; everything matches up. But if there’s no such plane, and you've got those four different groups, then that carbon is likely your chiral center. It’s the lack of internal mirroring that makes it special.

Sometimes, a molecule might have multiple chiral centers. This is where things get really exciting, and sometimes, a little mind-bending. Each chiral center can create different stereoisomers. Think of it as having multiple levers you can pull, each creating a slightly different outcome. The number of possible stereoisomers can be 2^n, where 'n' is the number of chiral centers. So, even with just a few chiral centers, you can have a whole family of related molecules!
And you know what's super cool? These chiral molecules, these enantiomers (that's what we call the mirror-image pairs), can behave completely differently in biological systems. One might be a lifesaver (like thalidomide, though sadly that was a disaster in its enantiomeric form), while its mirror image is ineffective or even harmful. It’s like having two keys that look identical, but only one opens the lock!
So, when you're asked to identify all the chirality centers, take a deep breath. Don't rush. Be methodical. Focus on those carbons. Check if they have four single bonds. Then, and this is the crucial part, check if all four of those attached groups are genuinely different. Trace out those chains. Look at the rings. Compare, compare, compare!
It's a skill that gets better with practice. The more structures you analyze, the faster you’ll become at spotting those elusive chiral centers. Soon, you’ll be able to do it almost instinctively, like recognizing a familiar face in a crowd. It’s all about training your molecular detective skills.
Remember the key rule: four different groups. If a carbon atom has that, and only single bonds, you've likely found yourself a chiral center. It's the heart of molecular handedness, the reason why some things interact with our bodies the way they do. Pretty amazing, right? So go forth, and find those chiral centers! Happy molecule hunting!
