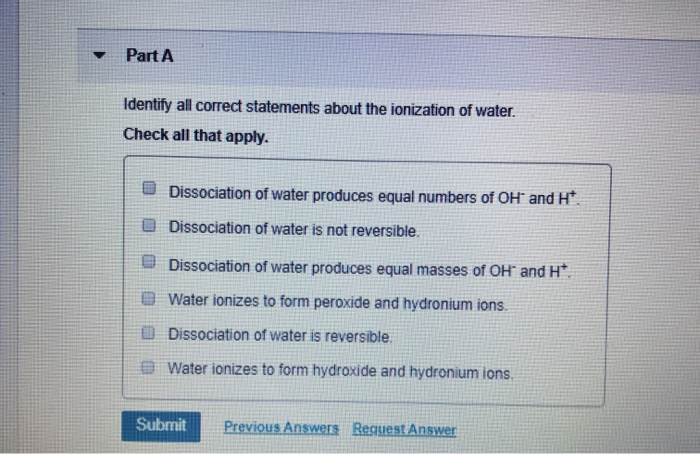
Identify All Correct Statements About The Ionization Of Water.

Hello there, science enthusiasts and curious minds! Today, we’re diving into something that might sound a little intimidating at first, but is actually incredibly fundamental to our existence and fascinating in its own right: the ionization of water. Think of it as water’s secret life, the hidden dance of its molecules that makes so much of our world possible. It’s not just for chemistry labs; understanding this process can illuminate how everything from your morning coffee to the oceans themselves operates.
So, why should we care about water’s ionization? The benefits and purpose it serves are truly everywhere. At its core, water ionization refers to the phenomenon where water molecules (H₂O) spontaneously break apart, or dissociate, into hydrogen ions (H⁺) and hydroxide ions (OH⁻). While a tiny fraction of water molecules do this at any given moment, this equilibrium is crucial. It’s what gives water its ability to act as a universal solvent, allowing it to dissolve a vast array of substances, which is essential for biological processes, chemical reactions, and countless industrial applications. Without this slight ionization, life as we know it simply wouldn't exist. Imagine trying to digest food or transport nutrients without water’s dissolving power!
You encounter the effects of water ionization constantly. When you drink water, its pH level, which is directly related to the balance of H⁺ and OH⁻ ions, determines if it's acidic, neutral, or alkaline. This impacts taste and our bodies' own delicate pH balance. Think about cooking: the way ingredients react and cook is influenced by the water you use. In nature, the pH of rivers and lakes affects aquatic life. Even cleaning your home involves water’s solvent properties, enhanced by its ionization. Every chemical reaction that occurs in an aqueous solution relies on this fundamental process.
Now, how can you “enjoy” understanding water ionization more effectively? It’s less about an activity and more about appreciating the science around you. Here are some practical tips:
Firstly, pay attention to pH. When you’re out and about, notice the pH of different beverages or even the water from your tap. Understanding that a neutral pH is around 7, with lower numbers being acidic and higher numbers alkaline, can be an interesting observation. You might even be surprised by the pH of certain foods and drinks!
Secondly, consider the applications. Next time you’re using cleaning products, remember that water’s ability to break down grime is powered by its ionization. When you’re enjoying a cup of tea or coffee, appreciate how the water dissolves the flavor compounds. These everyday moments become a little more profound when you connect them to the underlying science.

Finally, explore further. If you’re really intrigued, don’t hesitate to look up more information. There are fantastic educational resources online, from simple explanations to more detailed scientific articles, that can further illuminate the world of aqueous chemistry. Understanding this aspect of water can unlock a deeper appreciation for the complex and beautiful chemistry that shapes our planet.
So, while you can't exactly "do" water ionization for fun, you can certainly gain a richer understanding and appreciation for its vital role in everything around us. It’s a silent, constant force, and a truly fascinating topic to explore!
