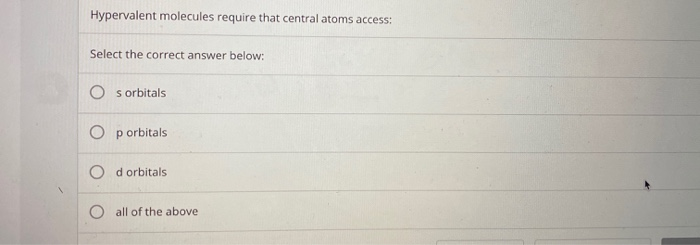
Hypervalent Molecules Require That Central Atoms Access
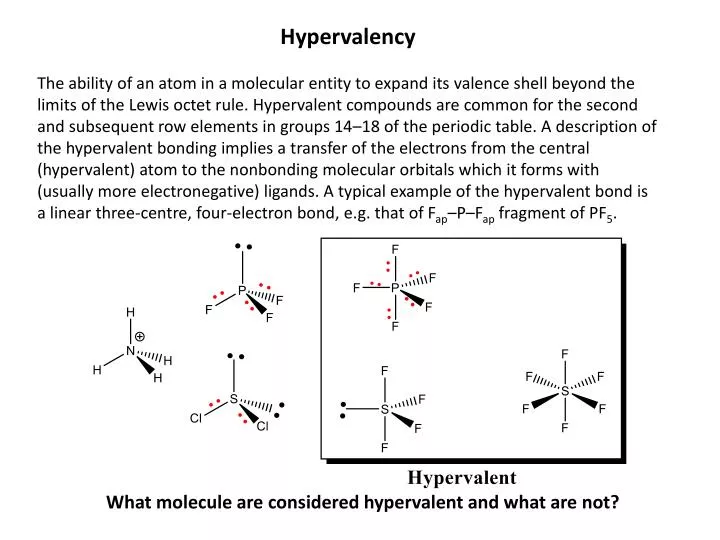
Okay, let's talk about atoms. You know, those tiny little things that make up everything. We all learned about them in school, right? They have a nucleus, and then electrons buzzing around like frantic bees. It's a whole miniature solar system in there. And usually, atoms are pretty chill. They like to have just enough electrons to feel complete. Like having a perfectly balanced bookshelf, you know? Not too many books, not too few. Just right.
But then, sometimes, you run into a molecule that's just… extra. It’s got way more electrons than it really needs. We’re talking about what scientists call hypervalent molecules. And honestly, I think it’s just a fancy way of saying these central atoms are having a bit of an identity crisis. They’re like that one friend who always tries to bring five extra people to a party that’s already at capacity. It’s a crowd!
You see, most atoms are content with their little electron shells. They’ve got their designated spots, their comfort zones. It’s all very neat and tidy. But a central atom in a hypervalent molecule? This guy is like, "You know what? Let’s just cram a few more in here!" It's a bit of a free-for-all. Like someone decided to open up a second, unauthorized snack bar in the middle of a perfectly good buffet. Chaos!
And what’s the deal with this "access" they need? Apparently, these central atoms have to reach out and grab electrons from their neighbors. It's like they’re reaching over the fence to borrow a cup of sugar, but instead of sugar, it's electron-y goodness. And they’re not just borrowing a little. They're taking a whole handful. It’s a bit of a power move, if you ask me. They're not asking nicely; they're demanding.
Think of it like this: Imagine you’re at a potluck. Everyone brought a dish. It’s all good. Then, one person arrives and just starts piling their plate higher and higher, taking the best bits from everyone else’s contributions. That’s kind of what these hypervalent molecules are doing. The central atom is the greedy guest, and the surrounding atoms are the innocent potluck contributors.

It's a little dramatic, I know. But honestly, isn't it kind of funny? These tiny, invisible things, behaving like characters in a sitcom. The central atom, feeling a bit insecure, decides the best way to feel powerful is to just hoover up all the available electrons. It’s the atomic equivalent of a power suit and a booming voice, all because it’s got… more.
And the neighbors? Well, they just have to deal with it. They’re the ones who have to share. They’re the ones who are a little bit depleted so the central atom can shine. It’s a bit of a one-sided relationship, isn't it? The central atom is all about its own expansion, its own electron party, while the others are left wondering where all their valuable bits went.

It’s like the central atom has a special VIP section for electrons, and everyone else has to stand outside. And this "access" they require? It’s just the key to get into that exclusive club. They’re not just accepting what they’re given; they’re actively seeking out more. They’re go-getters, I guess. Even if their getting involves a bit of electron-pilfering.
So next time you hear about a hypervalent molecule, just picture it. Picture that central atom, with its extra electron entourage, feeling all smug. It’s got more electrons than its neighbors, and it’s not afraid to show it. It’s a bit of a show-off, this hypervalent thing. And the rest of the molecule? They’re just along for the ride, probably a little bit tired from all the electron sharing.
It’s like having too many cousins show up at your small apartment for the holidays. Everyone’s crammed in, and the main person in charge (the central atom) is just trying to make room for everyone, even if it means stacking people on top of each other. It’s not ideal, but hey, it’s a party, right? A very, very crowded party.
And the funny thing is, these molecules are everywhere! They're not some rare, exotic specimens. They're out there, doing their hypervalent thing, making the world a little more crowded, electron-wise. It’s a quirky universe, isn't it?
So, while the scientists might use all sorts of big words to describe it, I like to think of it as just atoms being a little too enthusiastic about collecting. It's like they've discovered a new hobby: electron hoarding. And the central atoms? They're the undisputed champions of this particular game. They've got the access, they've got the electrons, and they've definitely got the bragging rights. It's a bit of an "unpopular opinion," but I'm convinced these hypervalent molecules are just misunderstood hoarders. And honestly, who can blame them? Electrons are pretty cool.
