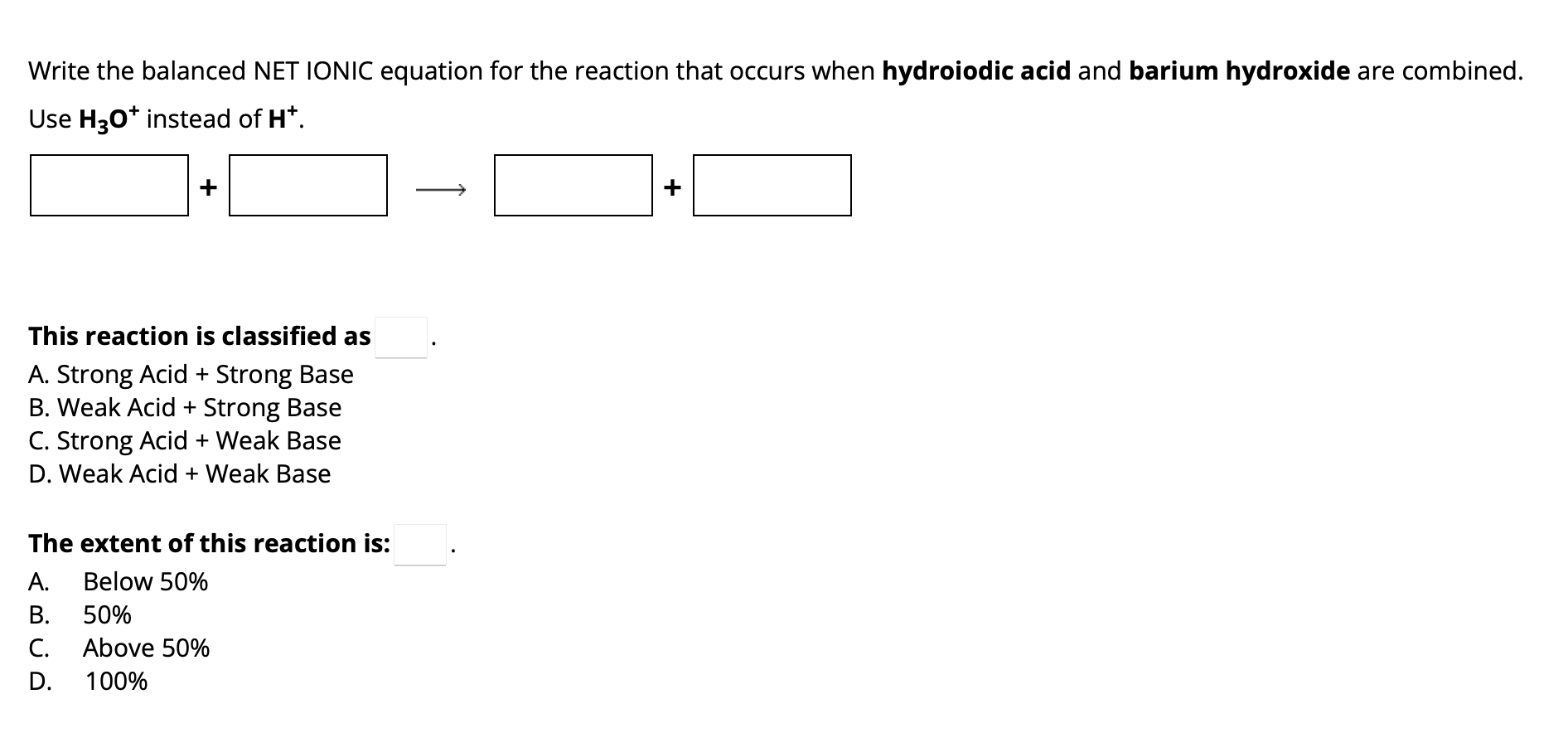
Hydroiodic Acid And Barium Hydroxide Balanced Equation
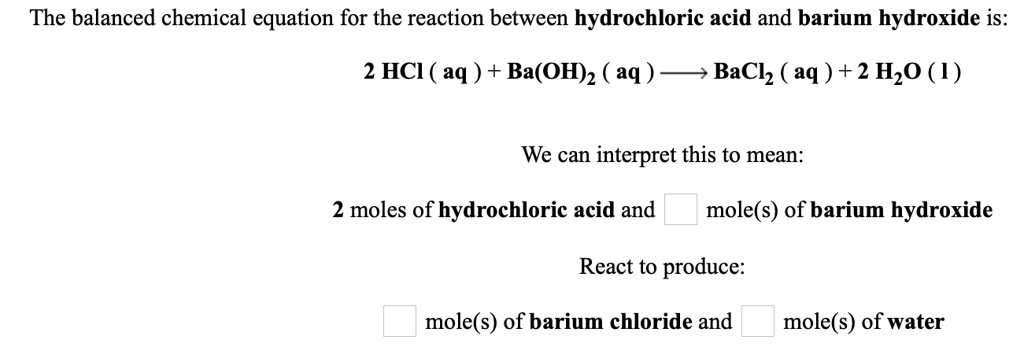
Let's talk about something that sounds super fancy and maybe a little intimidating: the balanced equation for hydroiodic acid and barium hydroxide. Yep, we're diving into the world of chemistry, but don't worry, we're keeping it fun. Think of it like a culinary experiment, but instead of making cookies, we’re making… well, we'll get to that.
So, we have hydroiodic acid. Sounds like something you'd find in a mad scientist's lab, right? It's basically an acid. And then we have barium hydroxide. This one sounds a bit like a fancy soap. It’s a base. Now, when acids and bases get together, they often have a little party. This party is called a reaction.
In our case, these two pals, hydroiodic acid and barium hydroxide, decide to shake hands. But in chemistry, shaking hands means something a bit more… dramatic. They transform into new things. It's like when you meet someone new and suddenly you're planning a road trip and learning how to bake sourdough. Things change!
The big secret, the part that might make you raise an eyebrow and say, "Really?", is that they don't just become any old things. They create barium iodide and water. Yes, water. That stuff we drink and shower in. And barium iodide. That sounds like it should be in a sci-fi movie, but it’s just a salt. A bit of a posh salt, perhaps.
Now, the "balanced equation" part. This is where things get a tiny bit nerdy, but stick with me. Imagine you're at a buffet. You want to make sure you have the right number of each food item. You don't want to end up with way too many mashed potatoes and not enough gravy, right? Chemistry equations are like that, but with atoms. We want to make sure that on one side of the equation (what you start with), you have the same number of each type of atom as you do on the other side (what you end up with).

So, we start with hydroiodic acid. Its formula is HI. Simple enough. And barium hydroxide. That’s Ba(OH)₂. See that little '2' there? That's important. It means there are two hydroxide bits for every barium bit. Like having two scoops of ice cream for one cone.
When they react, they make barium iodide. That's BaI₂. Again, two iodines for every barium. And water, which we all know is H₂O. Two hydrogens and one oxygen.
So, if we just wrote HI + Ba(OH)₂ → BaI₂ + H₂O, we’d have a problem. Let's count our atoms. On the left, we have one H, one I, one Ba, two O's, and two H's again (from the OH groups). That's a mess. On the right, we have one Ba, two I's, two H's, and one O. See how the iodines don't match? And the number of hydrogens and oxygens are a bit off too.
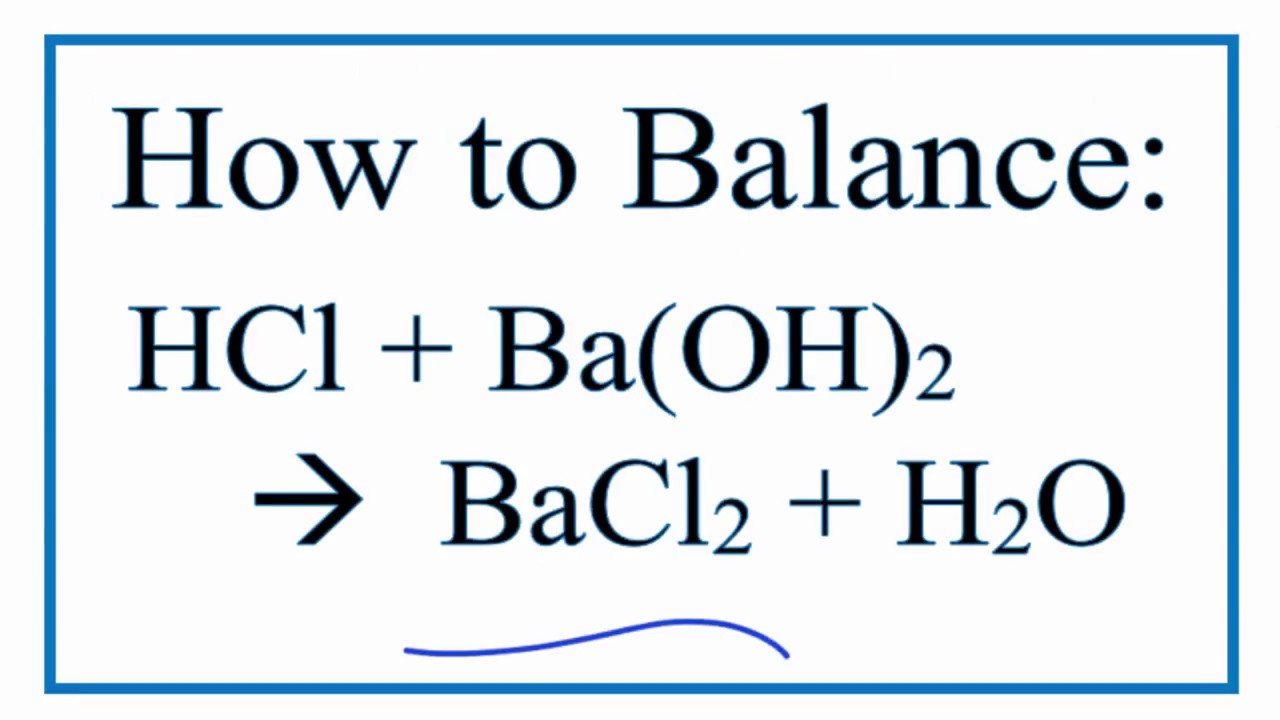
This is where my "unpopular opinion" comes in. I think balancing chemical equations is like trying to fold a fitted sheet perfectly. It’s supposed to be satisfying, but sometimes it just feels like a tangled mess that you eventually shove into a closet. Anyone else feel me on this?
I mean, who decided chemistry had to be so… precise? Can't atoms just be chill and hang out with whatever other atoms they feel like?
Write the balanced NET IONIC equation for the | Chegg.com
But alas, science marches on, and we must balance. So, to fix our HI and Ba(OH)₂ situation, we need more HI on the starting side. We need two molecules of hydroiodic acid. That’s where the big '2' comes in before the HI. So, we have 2HI + Ba(OH)₂.
Now let's count again. On the left: two H's from 2HI, two I's from 2HI, one Ba, two O's, and two H's from Ba(OH)₂. Total H's? Four. Total I's? Two. Total Ba's? One. Total O's? Two.
On the right: one Ba, two I's, two H's, and one O. Still not quite right. We have two iodines and one barium matching up nicely for barium iodide (BaI₂). But the hydrogens and oxygens from the hydroiodic acid and barium hydroxide need to make water. We have four hydrogens and two oxygens that need to form water (H₂O). How many H₂O molecules do we need to use up four hydrogens and two oxygens? That’s right, two molecules of water!
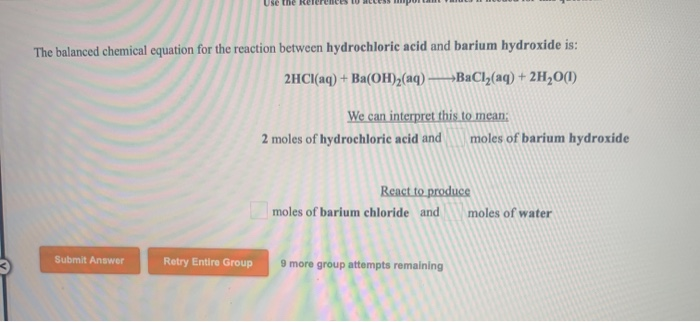
So, our balanced equation looks like this: 2HI + Ba(OH)₂ → BaI₂ + 2H₂O.
Let's do a final check. Left side: 2 H (from 2HI) + 2 H (from Ba(OH)₂) = 4 H. 2 I. 1 Ba. 2 O. Right side: 1 Ba. 2 I. 2 * 2 H (from 2H₂O) = 4 H. 2 * 1 O (from 2H₂O) = 2 O. Everything matches! It’s a perfect chemical symmetry. It’s like finding matching socks in the laundry, a rare and beautiful moment.
And there you have it. The balanced equation for hydroiodic acid and barium hydroxide. It might sound complex, but at its heart, it’s just about making sure everyone gets accounted for. A little bit of order in the chaotic universe of chemical reactions. It’s kind of like when you finally get your messy room clean – a sense of accomplishment, even if it’s temporary. Now, if you’ll excuse me, I need to go ponder the existential dread of folding fitted sheets.