Hydrogen Peroxide + Manganese Dioxide Balanced Equation
Hey there, science explorers and curious minds! Ever been chilling in your kitchen and wondered about that brown bottle of stuff labeled "hydrogen peroxide"? You know, the one you use to clean scrapes or make spooky Halloween fog? Well, today we're going to dive into a little behind-the-scenes magic that happens when hydrogen peroxide meets a super cool catalyst. Think of it as a backstage pass to a chemical reaction that's way more interesting than you might think. We're talking about hydrogen peroxide + manganese dioxide, and it's a pretty neat little show.
So, what even is hydrogen peroxide? Imagine water (H₂O), but with an extra oxygen atom hanging out. That's essentially what it is – H₂O₂. This extra oxygen makes it a bit unstable, a bit eager to, well, do something. It’s like a tightly wound spring, just waiting for a nudge.
And what about manganese dioxide? This is where things get really interesting. Manganese dioxide (MnO₂) is a bit like a helpful friend, or maybe a really good conductor at a concert. It doesn't actually get used up in the reaction itself. It just hangs around, speeding things up and making the whole process much easier. Scientists call this kind of helper a catalyst. Pretty neat, right? It's like having a referee who doesn't play the game but makes sure it runs smoothly.
The Big Unveiling: What Happens?
Okay, so picture this: you've got your hydrogen peroxide, that eager-to-react molecule. Then, you toss in a pinch of that manganese dioxide. What’s the result? Poof! You get a whole lot of oxygen gas and some good old-fashioned water.
Think of it like a chemical party. Hydrogen peroxide arrives, feeling a little restless. Manganese dioxide is already there, setting up the dance floor and playing some upbeat music. Suddenly, the hydrogen peroxide molecules decide, "You know what? I've had enough of this extra oxygen hanging around!" They break apart, and the oxygen (O₂) floats off as a gas, like tiny bubbles of freedom. And what's left? Just regular water (H₂O), all calm and collected. It’s like the oxygen was the energetic guest who eventually leaves, and everyone else can just relax.
Why All the Fuss About a Balanced Equation?
Now, you might be thinking, "Okay, it makes oxygen and water. Big deal." But in the world of chemistry, the details are everything. And that's where the concept of a balanced chemical equation comes in. It’s like a recipe or a meticulous accounting ledger for atoms.
You see, in the universe, matter isn't created or destroyed. It just changes form. This fundamental rule is called the Law of Conservation of Mass. So, when our hydrogen peroxide breaks down, all the atoms that were in the original hydrogen peroxide have to end up somewhere in the products – in this case, oxygen gas and water. A balanced equation makes sure that the number of each type of atom is exactly the same on both sides of the arrow.
It’s kind of like sorting LEGO bricks. If you start with 10 red bricks and 5 blue bricks, at the end, you should still have exactly 10 red bricks and 5 blue bricks, even if you've built something completely different with them. The balanced equation is our way of proving that we’ve accounted for every single brick.
The Star of the Show: The Balanced Equation Itself!
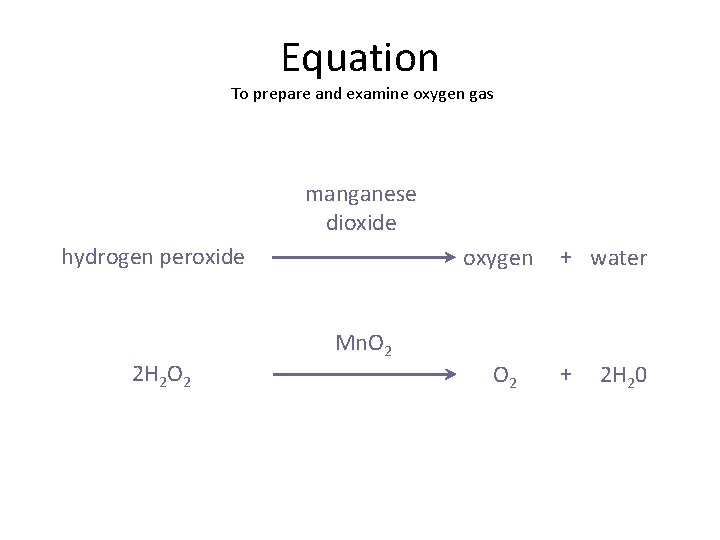
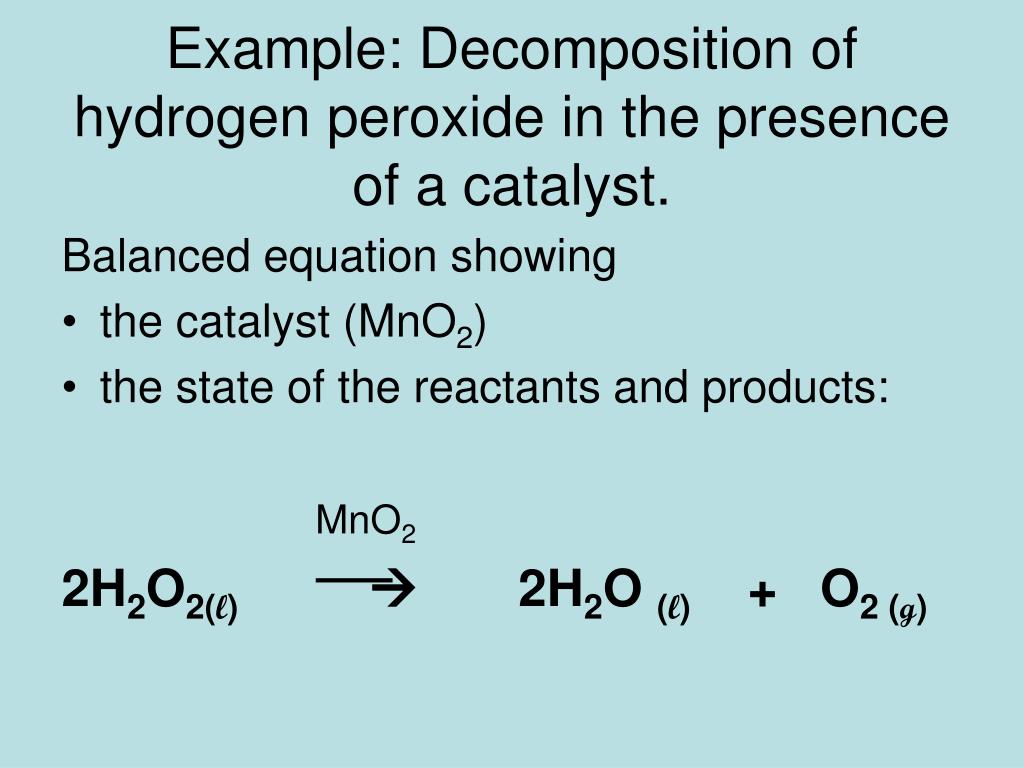
So, what does this fancy balancing act look like on paper? Here it is, in all its glory:
2 H₂O₂ (aq) → 2 H₂O (l) + O₂ (g)
Let's break this down, piece by piece, like a detective examining clues.
First, we have 2 H₂O₂. This tells us we start with two molecules of hydrogen peroxide. Why two? We'll get to that!
Then, we have the arrow (→). This is the universal symbol for "transforms into" or "yields." It’s the grand finale, the moment of change.
On the other side of the arrow, we see 2 H₂O. This means we end up with two molecules of water. See how the hydrogen and oxygen atoms from the original hydrogen peroxide have rearranged themselves into water?
And finally, we have + O₂. This is the exciting part – the oxygen gas that bubbles away! It’s like the fizzy pop you get when you open a soda.
The little letters in parentheses are also important. (aq) means "aqueous," which is just a fancy way of saying it's dissolved in water. (l) means "liquid," and (g) means "gas." So, we start with hydrogen peroxide dissolved in water, and we end up with liquid water and oxygen gas.
Why the "2"s? The Balancing Act Explained
Now, let's get back to those pesky "2"s. They are the key to making sure our equation is balanced. Let's count the atoms on each side:
- Left Side (Reactants): We have 2 molecules of H₂O₂. Each molecule has 2 hydrogen atoms and 2 oxygen atoms. So, in total, we have 2 x 2 = 4 hydrogen atoms and 2 x 2 = 4 oxygen atoms.
- Right Side (Products): We have 2 molecules of H₂O. Each molecule has 2 hydrogen atoms and 1 oxygen atom. So, that's 2 x 2 = 4 hydrogen atoms and 2 x 1 = 2 oxygen atoms. THEN, we have the O₂ molecule, which has 2 oxygen atoms. So, in total for oxygen, we have 2 + 2 = 4 oxygen atoms.
Voila! 4 hydrogen atoms on both sides. 4 oxygen atoms on both sides. The equation is perfectly balanced! It’s like a perfectly choreographed dance where every dancer has a partner and everyone ends up in the right place.

And where does our catalyst, manganese dioxide (MnO₂), fit in? It’s so crucial for making this reaction happen at a decent speed, but it doesn't actually get chemically changed. So, in the balanced equation itself, we don't include it as a reactant or product. However, it's often written above the arrow to show that it's present and doing its catalytic magic. Think of it as the stage manager who's essential but not part of the play’s script.
Why is This So Cool?
Beyond just being a neat chemical trick, this reaction is incredibly useful! Remember that bubbling you see when you pour hydrogen peroxide on a cut? That's the oxygen gas being released, and it's a sign that the hydrogen peroxide is doing its antiseptic work. While manganese dioxide isn't usually found in your first-aid kit, it's a common ingredient in experiments that create those impressive oxygen fountains or the "elephant's toothpaste" effect (which is basically a super-charged version of this reaction!).
It’s a simple yet powerful example of how a little catalyst can dramatically change the pace of a chemical reaction. It shows us that even everyday substances have complex and fascinating processes happening within them. It’s a reminder that the world around us is constantly buzzing with activity, even if we can’t always see it.
So, the next time you see that brown bottle of hydrogen peroxide, give it a little nod. It’s more than just a cleaner; it’s a molecule with potential, just waiting for a little help from a friend like manganese dioxide to show its true colors – or in this case, to turn into harmless water and a burst of life-giving oxygen. Pretty cool, right?
