Hydrogen Bonds Are Found In All Of The Following Except
Hey there, science adventurers! Ever feel like chemistry is this big, intimidating beast, all complicated formulas and reactions you can’t pronounce? Well, guess what? It’s actually a secret superpower, and today we’re going to uncover a little piece of that magic. Forget those late-night study sessions with dry textbooks. We're diving into something super cool, something that’s happening all around you, even as you’re reading this. We’re talking about hydrogen bonds!
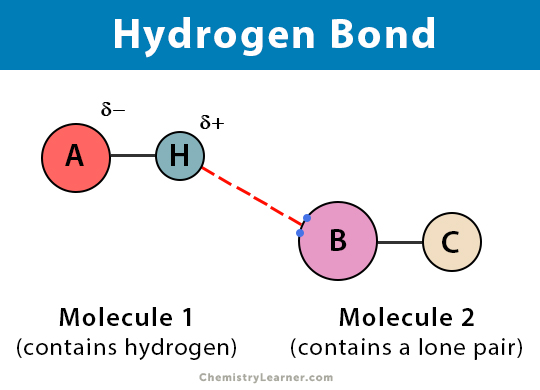
Now, you might be thinking, "Hydrogen bonds? Sounds… technical." But trust me, they’re less about beakers and more about besties. Think of them as the ultimate social butterflies of the molecular world. They're these special, super-weak, yet incredibly important attractions between certain atoms. Specifically, they happen when a hydrogen atom, already a little bit lonely because it’s bonded to something that really likes to hog electrons (like oxygen or nitrogen), gets drawn to another electron-hoarding atom nearby. It’s like a gentle nudge, a friendly wave, a whispered "hello" between molecules.
Why should you care about these little molecular handshakes? Oh, my friends, because they are the unsung heroes of so many amazing things! They are the glue that holds together the very fabric of life as we know it. Seriously! Ever wonder why water is so… well, watery? That amazing property of water, its ability to be a liquid at room temperature, to freeze into beautiful ice crystals, to climb up tiny plant stems – that’s all thanks to hydrogen bonds. They make water molecules stick together like a group of friends at a concert, even when things get a little wild.
Think about it: the dewdrop on a spiderweb, the steam rising from your morning coffee, the rain pattering on your windowpane – all these delightful spectacles are made possible by hydrogen bonds. They give water its surface tension, that invisible skin that allows tiny insects to walk on it. They’re responsible for the way your tea cools down, the way a river flows, and even the way your body stays hydrated. Pretty neat, right?
But it doesn’t stop there! These bonds are also the architects of our DNA! Yes, the blueprint of life itself is held together by these delicate attractions. Imagine the two strands of your DNA as a ladder. Hydrogen bonds are the rungs of that ladder, holding it all together in that iconic double helix shape. Without them, our genetic information would be all over the place, and well, we wouldn’t be here!
And proteins? Those amazing molecules that build our muscles, digest our food, and fight off diseases? They fold into incredibly specific shapes, and hydrogen bonds are a huge part of that folding process. It's like they're giving each protein a gentle hug to ensure it's in the perfect position to do its job. It’s truly mind-blowing stuff, happening constantly inside you, around you, everywhere!
So, we’ve sung the praises of where hydrogen bonds are. Now, for the fun part, the brain-tickler, the “aha!” moment: where are they not found? This is where we get to flex our understanding and appreciate the nuances of molecular interactions. It's a bit like a fun quiz, a gentle challenge to your newfound knowledge. The question is, "Hydrogen bonds are found in all of the following except..." This means we're looking for something that, by its very nature, doesn't have the right ingredients or the right setup for these special attractions.
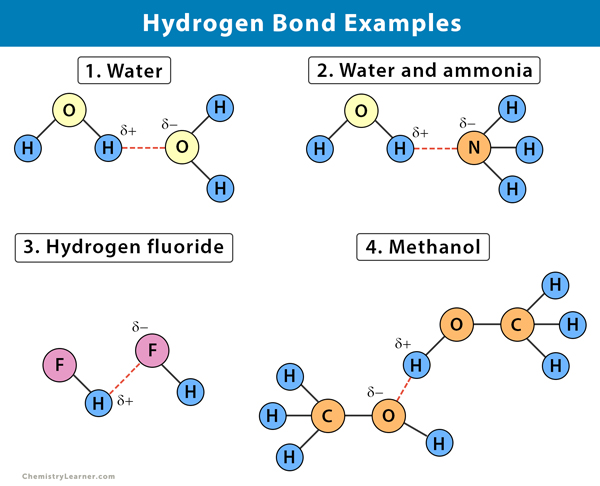
Let’s consider some common substances. You’ve got water (H₂O), right? Oxygen and hydrogen. Bingo! Perfect for hydrogen bonding. Then you have ammonia (NH₃). Nitrogen and hydrogen. Yep, another hydrogen bond enthusiast. Even things like alcohols, which have that special -OH group, are full of them. They make our life’s little joys possible, from the smoothness of lotion to the way sugar dissolves in our tea.

But what about something like, say, methane (CH₄)? Now, methane has hydrogen atoms, which is great. But the atom they're bonded to is carbon. And carbon, while it loves electrons, isn't quite as electronegative – meaning it doesn't pull electrons as strongly – as oxygen or nitrogen. Because of this, the hydrogen atoms in methane aren't polarized enough to be strongly attracted to another atom in a neighboring molecule. They just don't have that same "oomph" to form a significant hydrogen bond. It's like the molecules are polite acquaintances, nodding to each other across the room, but not quite grabbing each other’s hands for a dance.
Another example that would likely be an "except" is a simple alkane like ethane (C₂H₆). Again, you have hydrogen atoms, but they're attached to carbon atoms. The difference in electronegativity between carbon and hydrogen is too small to create the necessary polarity for substantial hydrogen bonding. These molecules are more likely to interact through weaker forces called London dispersion forces, which are more like fleeting glances than heartfelt greetings.

Or consider something like a metal oxide, like rust (iron oxide). While oxygen is present, the bonding with the metal is primarily ionic or covalent in a way that doesn't create the specific hydrogen-bonding scenario. The electrons are shared or transferred in a different manner, and the arrangement isn't conducive to those characteristic hydrogen-bond attractions we see in water or DNA.
So, the key to identifying where hydrogen bonds are not found is to look for molecules where the hydrogen atoms are bonded to atoms that are not sufficiently electronegative (like carbon in many organic molecules) or where the overall structure and bonding type don't facilitate these specific intermolecular attractions. It’s all about the right partners and the right dance moves, if you will!
Isn't it fascinating how these invisible forces play such a monumental role? They are the secret architects of our world, shaping everything from the mundane to the magnificent. And the best part? Understanding them doesn't require a PhD, just a little bit of curiosity and a willingness to see the wonder in the everyday. So next time you take a sip of water, admire a snowflake, or even just think about the amazing complexity of your own body, give a little nod to those incredible, ubiquitous hydrogen bonds. They’re the unsung heroes, and knowing about them makes the world a little more magical, and your own understanding a whole lot more powerful. Keep exploring, keep questioning, and you’ll find that science is an endless adventure waiting to be discovered!
