Hydrochloric Acid And Sodium Carbonate Balanced Equation
Ever wondered what happens when you mix a common household cleaner with baking soda? It’s more exciting than it sounds!
We're talking about a chemical reaction that's a little bit zesty. It’s like a tiny, fizzy party happening in your lab (or kitchen!).
The stars of this show are hydrochloric acid and sodium carbonate. Think of them as two friends who love to get together and create something new.
The Big Reveal: The Balanced Equation!
Now, in the world of chemistry, we like things to be fair. Everything that goes into a reaction needs to come out, just in a different form.
That's where the balanced equation comes in. It’s like a recipe that makes sure all the ingredients are accounted for. No cheating allowed in chemistry!
So, what’s the magic formula? It’s a bit of a mouthful, but stick with us!
Let's Meet Our Players
First up, we have hydrochloric acid. You might know it as HCl. It’s a strong acid, but when it’s diluted, it's actually used in some cleaning products.
It’s a bit of a drama queen, always ready to react. It loves to share its hydrogen atoms!
Next, we have sodium carbonate. This is also known as washing soda or soda ash. It’s what makes some detergents work so well.
Sodium carbonate is like a party host, bringing together a few different bits and pieces. It’s got sodium and carbonate ions just waiting for some excitement.

The Exciting Dance
When hydrochloric acid (HCl) meets sodium carbonate (Na₂CO₃), things get really interesting. It’s not just a simple hello.
They start to mingle and swap partners. It’s like a chemical square dance!
The hydrogen from the HCl decides to team up with the carbonate part of the sodium carbonate. And the sodium from the sodium carbonate finds a new friend in the chloride from the HCl.
The Fizz Factor
But the most entertaining part? The fizz! This reaction produces a gas called carbon dioxide.
That’s right, the same stuff that makes your soda bubbly! It’s like a spontaneous eruption of tiny gas bubbles.
This gas, CO₂, escapes from the mixture. It’s the fizzy evidence that a reaction has truly occurred. It’s like the universe is saying, "Ta-da! Something new has happened!"
The Other Products
So, we’ve got our fizzing carbon dioxide. But what else is left behind?
Remember how the sodium and chloride atoms swapped partners? They end up forming sodium chloride.

Yes, that's common table salt! It’s a surprising twist, isn't it? You start with a cleaning agent and baking soda, and you end up with salt and bubbles.
And there’s one more thing. Water! The hydrogen atoms from the acid and some of the oxygen from the carbonate form water (H₂O).
Putting It All Together
So, on one side of our equation, we have our reactants: hydrochloric acid and sodium carbonate.
On the other side, we have our products: carbon dioxide, sodium chloride, and water.
But here’s the trick: they have to be balanced. We need the same number of each type of atom on both sides.
The Magic Numbers: The Balanced Equation Revealed!
This is where the numbers, called coefficients, come into play. They ensure everything adds up perfectly.
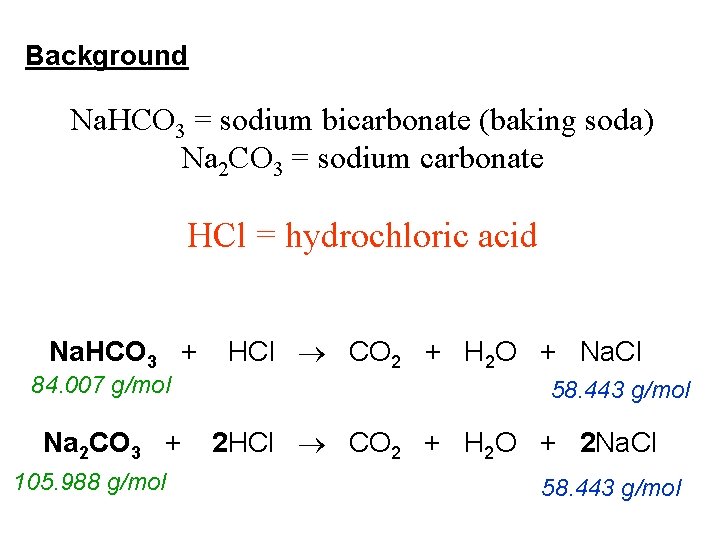
For our reaction, the balanced equation looks like this:

2 HCl + Na₂CO₃ → CO₂ + 2 NaCl + H₂O
Let's break down what those numbers mean.
Decoding the Numbers
The ‘2’ in front of HCl means we need two units of hydrochloric acid for every one unit of sodium carbonate.
Think of it as needing two partners for the sodium carbonate to feel truly balanced in this chemical dance. It's all about the right proportions!
The ‘1’ in front of Na₂CO₃ is usually invisible, but it’s there, signifying one unit of sodium carbonate. No need to write it!
On the other side, the ‘1’ in front of CO₂ and H₂O are also invisible. They mean one molecule of carbon dioxide and one molecule of water.
And that ‘2’ in front of NaCl? It means we get two units of sodium chloride (table salt).
Why Is This So Fun?
This whole process is like a tiny chemical magic show. You start with seemingly ordinary things and end up with a fizzing spectacle and even salt!

It’s the transformation that's so captivating. The way atoms rearrange themselves is pure chemistry theatre.
And the best part? You can often see this reaction happen in everyday life. Think of cleaning spills or even some DIY projects.
A Glimpse into the Chemical World
Understanding this balanced equation isn't just about memorizing symbols. It's about appreciating the order and precision of the universe.
Even though it seems simple, this equation represents a fundamental principle of chemistry. It shows us that matter is conserved. It never truly disappears.
It just changes its form. It’s a constant reminder of the cyclical nature of things.
Go Explore!
So, next time you see a fizz or hear a pop in a chemistry experiment, remember the humble equation of hydrochloric acid and sodium carbonate.
It’s a beautiful example of how chemistry can be both predictable and surprising. It's a tiny drama with a big payoff.
Why not try to find out more? The world of chemical reactions is full of these fascinating, balanced stories, just waiting to be discovered. It's a never-ending source of wonder!
