Hydrochloric Acid And Barium Hydroxide Balanced Equation
Alright, gather 'round, folks! Let's talk about a little something that sounds like it belongs in a mad scientist's lab, but it's actually way more down-to-earth. We're diving headfirst into the exciting (and slightly bubbly) world of hydrochloric acid and barium hydroxide. Now, I know what you're thinking: "Ooh, sciencey words! My brain might short-circuit!" But stick with me, because this is less about complex formulas and more about a spectacular chemical meet-cute, followed by a very tidy breakup.
Imagine this: you've got two characters, ready for their moment in the spotlight. On one side, we have Hydrochloric Acid. Think of it as the assertive, slightly edgy one. It's like that friend who always knows what they want and isn't afraid to get a little… acidic. Its scientific shorthand, for those who like to show off at parties, is HCl. It’s the stuff that helps your stomach digest that questionable burrito you probably shouldn't have eaten, so it’s a pretty useful character to have around, albeit a bit intense.
On the other side, strutting onto the chemical stage, is Barium Hydroxide. This one's a bit more of a gentle giant, but don't let that fool you. It's like a friendly, but very well-equipped bodyguard. Its name sounds a bit like a fancy European perfume, doesn't it? "Ah, yes, I'm wearing a hint of Barium Hydroxide today." Its chemical nickname is Ba(OH)₂. That little '2' at the end? It means it's got two of those hydroxyl groups, which are like little clinging arms ready to interact. Think of it as having a couple of extra hands for handshakes.
Now, when these two meet, it's not a messy brawl. Oh no, this is a choreographed dance of destruction… or rather, creation. They're going to react! And when chemicals react, they often do so to achieve a state of perfect, blissful balance. It's like they’re saying, "Okay, we’ve had our fun being individual, now let's form something stable and move on."
So, what happens when our edgy HCl and our bodyguard Ba(OH)₂ get together? It's a neutralization reaction, a classic in chemistry! Imagine HCl is a bit too eager, a bit too keen. It's got this extra hydrogen ion (H⁺) just itching to find a partner. Meanwhile, Ba(OH)₂ has these hydroxyl ions (OH⁻) just waiting for a hydrogen to latch onto. It's a match made in… well, a laboratory flask.
When they meet, the hydrogen from HCl and the hydroxyl from Ba(OH)₂ get together and form water (H₂O). Yep, plain old H₂O! It's like the ultimate peace treaty. They're so excited to form water that they practically high-five each other. This water is neutral, calm, and utterly unbothered by the previous drama. It's the chemical equivalent of taking a deep breath and saying, "Ahhh, much better."

But wait, there's more! We can't just leave our barium hanging around. Remember how HCl had that chlorine part, that Cl? And Barium Hydroxide had that leftover Ba? Well, these two also decide to pair up. They're like the kids who were stuck holding hands during a dance and realized they actually kind of like each other. So, Barium and Chlorine get together to form barium chloride.
Now, barium chloride (BaCl₂) is an interesting character. It's a salt. Not the kind you sprinkle on your fries, though it can look like crystals. This is a chemical salt, a product of this acid-base party. It's stable, it's happy, and it's perfectly content to chill in solution or sometimes precipitate out. It’s the quiet one who emerges from the lively gathering, looking perfectly put together.
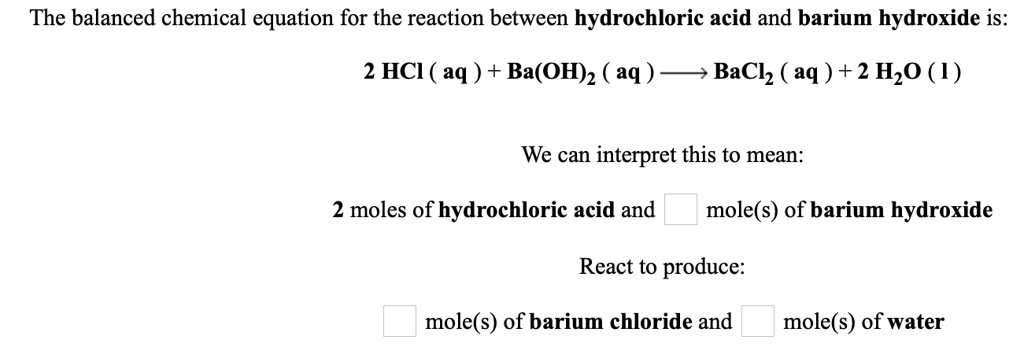
So, if we were to write this down as a chemical sentence, before balancing, it might look something like this: HCl + Ba(OH)₂ → H₂O + BaCl₂. Sounds simple enough, right? But here's where the "balancing" part comes in, which is basically chemistry's way of saying, "Okay, let's make sure everyone accounted for and nobody's trying to pull a fast one."
In our initial sentence, we have one hydrogen on the left (HCl) and two on the right (H₂O). That's a bit lopsided! And we have two hydroxyl groups on the left (Ba(OH)₂) and only one water molecule formed on the right. The universe, and chemistry, hates imbalance. It's like trying to have a party with more guests arriving than you have chairs; it just doesn't work.
So, we need to make sure the number of each type of atom is the same on both sides of the arrow. Think of it like counting your chickens before they hatch, but for atoms. We need the same number of H's, O's, Cl's, and Ba's on the reactant side (what you start with) as on the product side (what you end up with).
Let's look at Barium Hydroxide again: Ba(OH)₂. It has two hydroxide groups. Each hydroxide group (OH) has one oxygen and one hydrogen. So, Ba(OH)₂ is essentially saying, "I've got one Barium, two Oxygens, and two Hydrogens all bundled up." Meanwhile, Hydrochloric Acid, HCl, is just one Hydrogen and one Chlorine.
When HCl and Ba(OH)₂ react, we form water (H₂O) and barium chloride (BaCl₂). The barium chloride part is usually fine as is: one Ba and two Cl's. But our water formation? We've got two H's and one O in water. And where do those H's and O's come from? From our starting materials!
The Ba(OH)₂ gives us two OH groups, which means two O's and two H's. The HCl gives us one H and one Cl. Notice the mismatch? We have two O's and two H's available from the Ba(OH)₂, but only one H from the HCl. To make neutral water (H₂O), we need pairs of H's and O's.
This is where the real magic, or rather, the balancing act, happens. We need to make sure we have enough HCl to provide hydrogens for both of those hydroxide groups in Ba(OH)₂. If we only have one HCl, it can only "satisfy" one OH group, leaving the other one a bit lonely. We need two HCl molecules to give us two hydrogens, one for each of the two hydroxide groups in Ba(OH)₂.
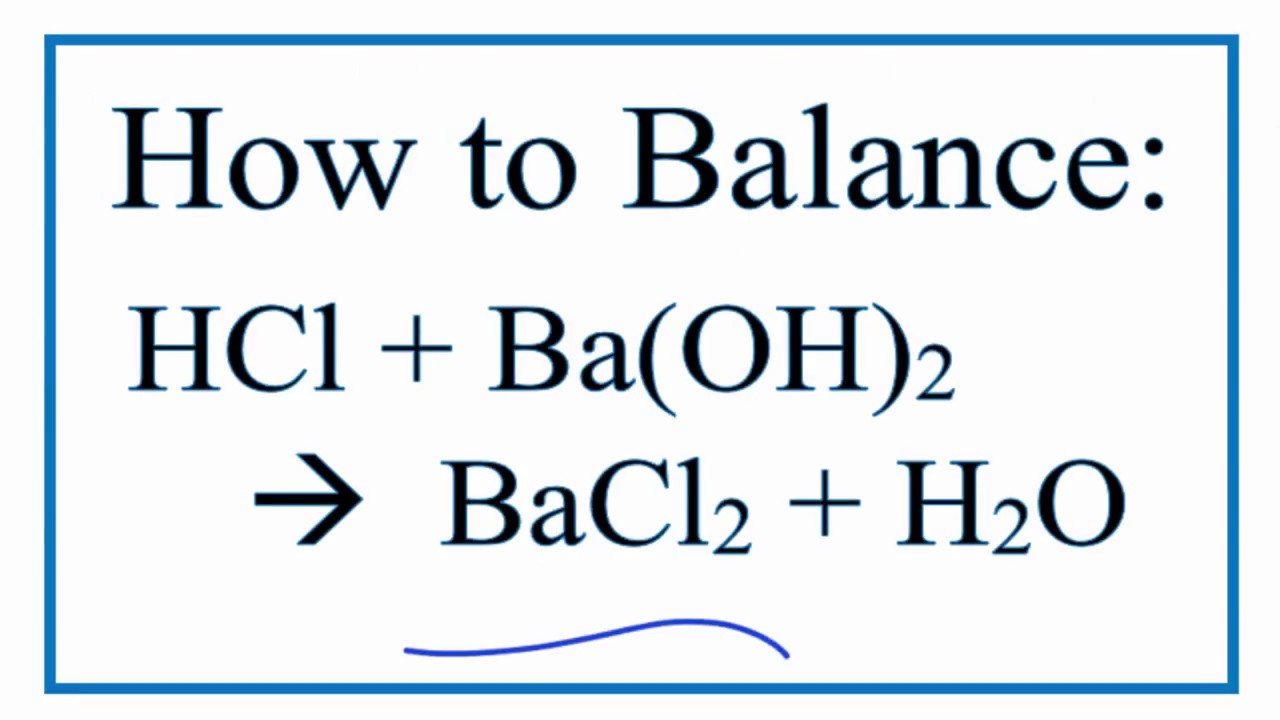
So, we put a big '2' in front of the HCl. Our equation now looks like this: 2 HCl + Ba(OH)₂ → H₂O + BaCl₂.
Let's check our counts: On the left: * Hydrogen (H): 2 (from 2 HCl) * Chlorine (Cl): 2 (from 2 HCl) * Barium (Ba): 1 (from Ba(OH)₂) * Oxygen (O): 2 (from Ba(OH)₂) On the right: * Water (H₂O): We need to see how many H's and O's we'll get. * Barium Chloride (BaCl₂): 1 Ba, 2 Cl. Okay, the Ba and Cl are balanced! We have one Ba on each side and two Cl's on each side. Now for the water. We have two OH groups from Ba(OH)₂. Each OH group will combine with one H from HCl to form water. So, we'll get two molecules of water! 2 H₂O. Let's rewrite our balanced equation: 2 HCl + Ba(OH)₂ → 2 H₂O + BaCl₂.
Now, let's recount: On the left: * Hydrogen (H): 2 (from 2 HCl) + 2 (from Ba(OH)₂) = 4 Hydrogens. Uh oh, something's still not quite right in my explanation there! Let's backtrack slightly in our explanation. The two HCl provide the H for the OH. Let's look at it this way: We have 2 HCl. That gives us 2 H⁺ ions and 2 Cl⁻ ions. We have Ba(OH)₂. That gives us 1 Ba²⁺ ion and 2 OH⁻ ions. When they meet: * The 2 H⁺ ions from HCl are going to combine with the 2 OH⁻ ions from Ba(OH)₂. Each H⁺ and OH⁻ pair forms H₂O. So, 2 H⁺ + 2 OH⁻ → 2 H₂O. This uses up all the H from HCl and all the OH from Ba(OH)₂. * What's left? The 2 Cl⁻ ions from HCl and the 1 Ba²⁺ ion from Ba(OH)₂. These will combine to form barium chloride: Ba²⁺ + 2 Cl⁻ → BaCl₂. So, the balanced equation is indeed: 2 HCl + Ba(OH)₂ → 2 H₂O + BaCl₂. Let's count atoms again, to be super sure: Left side: * H: 2 (from 2HCl) + 2 (from Ba(OH)₂) = 4 hydrogens. Wait! My explanation of where the hydrogens come from for water needs to be clearer. Let’s simplify. We start with: * Hydrochloric Acid: HCl * Barium Hydroxide: Ba(OH)₂ The reaction produces: * Water: H₂O * Barium Chloride: BaCl₂ Looking at the components: * Barium (Ba): There’s one Ba in Ba(OH)₂ and one Ba in BaCl₂. That's balanced already. * Chlorine (Cl): There’s one Cl in HCl and two Cl in BaCl₂. To balance this, we need two Cl atoms on the left. So, we put a 2 in front of HCl: 2 HCl. Now we have 2 Cl on the left. * Hydrogen (H): With 2 HCl, we now have 2 H atoms. Barium hydroxide, Ba(OH)₂, has two OH groups, meaning it has a total of 2 H atoms within those groups (one H in each OH). So, we have a total of 2 H from 2HCl and 2 H from Ba(OH)₂ on the left, making 4 H atoms. * Oxygen (O): Barium hydroxide, Ba(OH)₂, has two O atoms (one in each OH group). Now let's look at the products side with 2 HCl and Ba(OH)₂: * We form BaCl₂, which uses up our Ba and 2 Cl. * We have 2 H from the 2 HCl and 2 H from the Ba(OH)₂ (from the OH groups) and 2 O from the Ba(OH)₂ (from the OH groups). * These H's and O's combine to form water. Since we have 4 H atoms and 2 O atoms available in total from the reactants, and water is H₂O, we can form two molecules of water: 2 H₂O. Two H₂O molecules use up 4 H atoms and 2 O atoms. And voilà! Left side: * 2 x H (from 2HCl) = 2 * 2 x Cl (from 2HCl) = 2 * 1 x Ba (from Ba(OH)₂) = 1 * 2 x O (from Ba(OH)₂) = 2 * 2 x H (from Ba(OH)₂) = 2 Total on Left: 4 H, 2 Cl, 1 Ba, 2 O. Right side: * 2 x H (from 2H₂O) = 4 * 2 x O (from 2H₂O) = 2 * 1 x Ba (from BaCl₂) = 1 * 2 x Cl (from BaCl₂) = 2 Total on Right: 4 H, 2 O, 1 Ba, 2 Cl. See? Exactly the same number of each atom on both sides. It’s like a perfectly portioned meal. No leftovers, no shortages. Everyone is accounted for, and the reaction is complete, leaving us with neutral water and the salt, barium chloride.
So, the next time you hear about hydrochloric acid and barium hydroxide, don't picture a chaotic explosion. Picture a neat, tidy, and ultimately productive encounter that results in something everyone can agree on: water. And isn't that just a beautiful metaphor for life?
