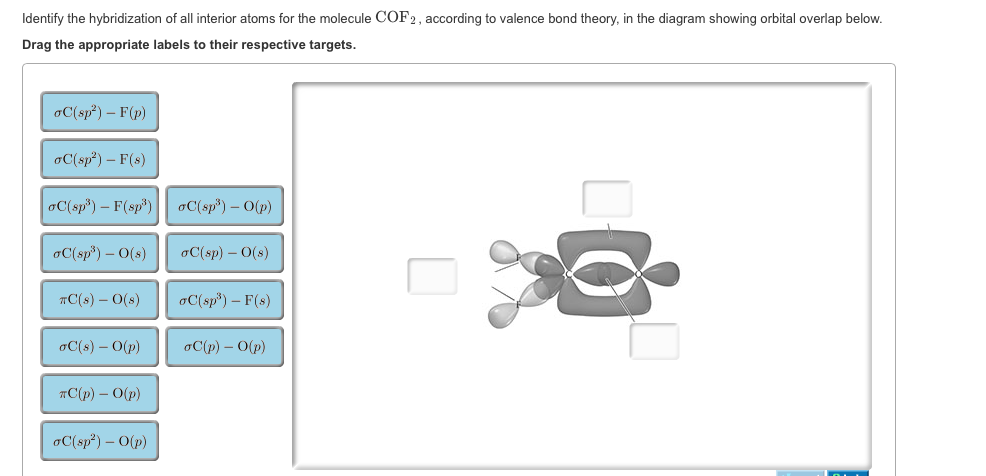
Hybridization Of All Interior Atoms For The Molecule Cof2
Hey there, science enthusiasts and curious minds! Ever look at a molecule and wonder what's really going on inside? Today, we're going to dive into the fascinating world of molecular bonding, specifically for a molecule you might have heard of, or maybe not: CoF₂. Now, before your eyes glaze over thinking about boring chemistry textbooks, stick with me! We're going to keep this super chill and explore why the way these atoms get together is actually pretty darn cool.
So, what exactly is CoF₂? Well, it's made up of one cobalt atom (that's the 'Co') and two fluorine atoms (that's the 'F₂'). Simple enough, right? But the real magic happens when we zoom in and think about how these atoms are actually holding hands, or rather, sharing electrons. This is where something called hybridization comes into play, and trust me, it's not as scary as it sounds. Think of it like a bunch of Lego bricks coming together to build something awesome!
What's the Big Deal About Hybridization?
Okay, so why do we even need this concept of hybridization? Imagine you have a bunch of perfectly shaped Lego bricks. You can build some cool stuff, sure. But what if you could melt those bricks down and reform them into totally new, custom shapes? That's kind of what hybridization does for atoms. It's a way for atomic orbitals – these are like the "rooms" where electrons hang out – to mix and match to create new, more suitable orbitals for bonding.
In simpler terms, the standard orbitals that atoms come with aren't always the best fit for forming strong, stable bonds. They might be the wrong shape, or point in the wrong direction. So, nature, in its infinite wisdom, allows these orbitals to get a little jumbled up and create new hybrid orbitals that are perfect for the job. It's like preparing the perfect dough for baking – you mix different flours and ingredients to get that ideal texture.
Diving into CoF₂: The Cobalt Story
Now, let's get back to our star, Cobalt (Co). Cobalt is a transition metal, which means it's got a bunch of electrons in its outer shells, and things can get a little more complex than with, say, your everyday elements like carbon or oxygen. When cobalt wants to bond with anything, it needs to figure out the best way to arrange its electrons and its orbitals.
.jpg)
For CoF₂, the cobalt atom is in the center, and it's got two fluorine atoms clinging to it. To make these bonds happen, the cobalt atom needs to offer up some of its electrons. But it's not just a simple donation. The orbitals on the cobalt atom actually mix. This is where hybridization becomes essential. It allows cobalt to create these perfectly shaped "arms" to reach out and grab onto the fluorine atoms.
Think of the cobalt atom's electron rooms. It has some spherical ones (s orbitals) and some dumbbell-shaped ones (p orbitals). To bond effectively with fluorine, these s and p orbitals might combine to form new orbitals. For CoF₂, a common hybridization scheme for transition metals like cobalt in this kind of bonding is sp³d hybridization. What does that even mean?
It means one 's' orbital, three 'p' orbitals, and one 'd' orbital from the cobalt atom all meld together to create five new, identical hybrid orbitals. These new orbitals are like custom-made tools, ready to form strong sigma bonds with the fluorine atoms. It's like going from a toolbox with a hammer, a screwdriver, and a wrench to a magical toolbox that instantly produces the exact tool you need for every single screw and bolt!

The Fluorine Friends
And what about our fluorine (F) friends? Fluorine is a highly electronegative element, meaning it loves to snatch electrons. When it comes to bonding, fluorine has its own set of orbitals. For simplicity, let's think about its valence electrons, the ones involved in bonding. Fluorine typically uses its 'p' orbitals to form bonds. In CoF₂, the fluorine atoms will be using these orbitals to overlap with the newly hybridized orbitals of cobalt.
So, you have these perfectly shaped hybrid orbitals from cobalt, and these ready-to-bond orbitals from fluorine, and bam! they overlap to create incredibly stable sigma bonds. These are the strong, direct connections between atoms. It's like two people shaking hands – a firm, direct grip.
Putting It All Together: The Geometry of CoF₂
Now, this hybridization isn't just about making bonds; it also dictates the shape of the molecule. With sp³d hybridization, the five hybrid orbitals on cobalt will arrange themselves in a specific way to be as far apart as possible, minimizing electron repulsion. This typically leads to a trigonal bipyramidal electron geometry. However, since cobalt is only bonding to two fluorine atoms and likely has some lone pairs of electrons (remember those electron "rooms" not used for bonding?), the overall molecular geometry can be different.

For CoF₂, you might expect a bent or V-shape, similar to water (H₂O). Imagine the cobalt atom at the point of a 'V', with the two fluorine atoms at the ends. This shape is a direct consequence of the electron arrangement dictated by the hybridization and the presence of lone pairs on the cobalt atom. It's like how the arrangement of your fingers affects the shape your hand makes when you're trying to pick something up.
The specific bond angles and the exact arrangement will depend on the interplay between the bonding pairs and any lone pairs on the cobalt. It’s a delicate dance of electron clouds pushing and pulling each other!
Why is This So Cool?
So, why should we care about hybridization in CoF₂? Well, it's a perfect example of how atoms aren't just static balls of matter. They are dynamic, constantly adjusting their electronic structures to achieve the most stable arrangement. This ability to hybridize is what allows for the incredible diversity of molecules we see in the world, from the simplest water molecule to complex DNA strands.
Understanding hybridization helps us predict how molecules will behave, what kind of reactions they'll undergo, and what properties they'll have. For CoF₂, the specific hybridization and geometry influence its reactivity, its potential applications in chemistry, and even its physical properties. It's the underlying reason why things are the way they are at the molecular level. It’s the secret blueprint that nature uses to build everything!
Think about it: the very fact that cobalt can form stable bonds with fluorine in this specific way is a testament to the power of these orbital mixing tricks. It's not just random; it's a highly optimized process. It’s like a chef skillfully blending ingredients to create a delicious dish – each component has its role, and the blending creates something far greater than the sum of its parts.
So, the next time you hear about a molecule like CoF₂, remember that behind those simple letters and numbers lies a complex and beautiful story of atomic orbitals getting together, mixing, and forming the bonds that create our world. It’s a reminder that even the most fundamental building blocks of matter are engaged in a constant, fascinating dance of attraction and arrangement. Pretty neat, huh?
