How To Find Coordination Number And Oxidation Number

Alright, gather 'round, you magnificent science-curious humans! Ever stared at a chemical formula and felt like you were trying to decipher ancient hieroglyphs whispered by a particularly grumpy pigeon? Yeah, me too. But fear not, for today, we're going to demystify two of the most fundamental, yet oddly intimidating, concepts in chemistry: the Coordination Number and the Oxidation Number. Think of them as the VIP guest list and the mood ring of the molecular party, respectively. And trust me, once we crack these, you'll be strutting around like a Nobel laureate at a coffee tasting.
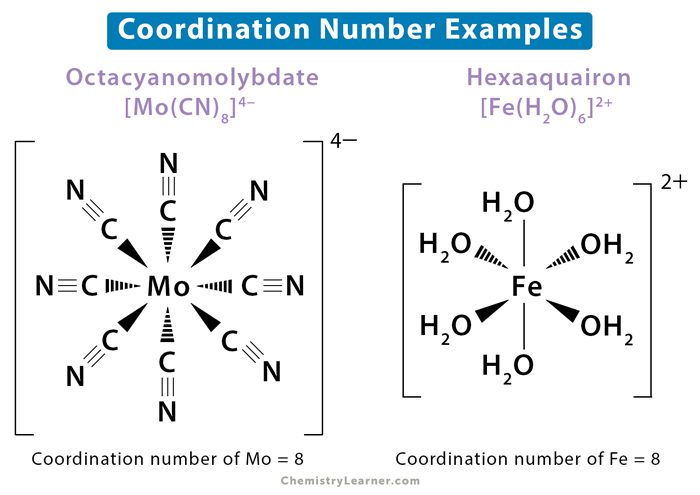
Let's kick things off with the Coordination Number. Imagine a dazzling, central atom, like a king or queen holding court. This regal atom loves to be surrounded by its loyal subjects – the surrounding atoms or molecules, affectionately known as ligands. The coordination number is simply the number of direct connections between our central atom and its devoted ligands. It's like counting how many hands are holding the king's hand. Simple, right? But oh, the drama it can hide!
Think of it this way: if you're at a buffet (the central atom, obviously, because who else gets all the attention?), your coordination number is how many different plates you're juggling simultaneously. Are you a sophisticated diner with just one perfectly balanced plate (coordination number 1)? Or are you a chaotic genius, piling on a dessert, a salad, a main course, and maybe even a rogue bread roll, all precariously balanced (a much higher coordination number)? The chemical world works similarly, just with less gravy stains.
A classic example is the humble ammonia molecule, NH₃. If we have a central nitrogen atom, it can bond to three hydrogen atoms. So, its coordination number is 3. Easy peasy. Now, let's go a bit fancier. Consider a complex ion like [Co(NH₃)₆]³⁺. Here, our central cobalt (Co) atom is chilling with six ammonia molecules (NH₃) hanging on for dear life. So, what's the coordination number? You guessed it: 6! It’s like that one friend who always has six people clinging to them at a party, just trying to get a word in edgewise. Poor cobalt.
Now, a little scientific gossip: the coordination number isn't always what you'd intuitively expect. Sometimes, ligands can be doubly or even triply attached to the central atom, acting like those clingy guests who bring their own security detail. These are called chelating ligands, and they can really inflate the coordination number by acting like a multi-pronged hug. It's a chemical embrace, people! Imagine a lobster claw trying to grab the central atom – that’s some serious coordination!

But here's a surprising fact to blow your tiny chemistry minds: some coordination numbers can be as high as 12! Twelve! That's like a central atom being the absolute most popular kid in school, with practically the entire graduating class wanting to be its lab partner. It’s pure chemical popularity, and frankly, a bit exhausting to visualize. I’m pretty sure my brain starts to sweat just thinking about it.
Alright, enough with the hand-holding and buffet-juggling. Let’s move on to our next star player: the Oxidation Number. This one is a bit more about the charge – specifically, the hypothetical charge an atom would have if all its bonds were completely ionic. Think of it as the atom's "emotional baggage" or its "reputation score" in the chemical world. Is it a generous electron-giver, a greedy electron-taker, or somewhere in the middle?
For individual atoms that aren't bonded to anything (like a lone sodium atom chilling by itself), their oxidation number is simply 0. Makes sense, right? No commitments, no charge. But the moment they start bonding, things get spicy. It's like getting a job – suddenly you have responsibilities and a whole new set of expectations!

Here’s where the rules come in, and they’re a bit like traffic laws for atoms: mostly sensible, but occasionally leading to a pile-up. The most important rule, the one you must tattoo on your brain (or at least put on a sticky note on your monitor), is that the sum of the oxidation numbers in a neutral compound is always 0. And in a polyatomic ion, the sum equals the charge of the ion. It's the ultimate chemical accounting principle.
Now, let’s talk about some common players. Oxygen, bless its electronegative heart, usually likes to be an electron-grabber. So, it typically sports an oxidation number of -2. It’s the undisputed champion of taking electrons, unless it’s bonded to fluorine (the ultimate electronegativity bully) or is in a peroxide, where it gets a slightly less grumpy -1. It’s like the boss who’s always demanding more, except when they’re talking to their boss.
![[SOLVED] Find coordination number, oxidation no. of metal and charge on](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/11/6380b7c6075e5_0376380b7c5b10ee.jpg)
Hydrogen, on the other hand, is a bit of a chameleon. When bonded to most non-metals (like in water, H₂O), it plays nice and takes on a +1 oxidation number. It’s like the eager intern, happy to help out. But when it decides to bond with a metal (like in sodium hydride, NaH), it flips the script and becomes a grumpy electron-hogger with an oxidation number of -1. It’s the intern who suddenly demands a corner office.
And the alkali metals (Group 1 on the periodic table – Lithium, Sodium, Potassium, etc.)? They are the most generous souls in the chemical universe. They always, and I mean ALWAYS, have an oxidation number of +1 when they're in a compound. They’re like those friends who are always lending you money, no questions asked. Just pure, unadulterated positive vibes.
Let's try a little practice, shall we? Consider water, H₂O. We know oxygen is usually -2. Since water is neutral (charge of 0), and we have two hydrogens, let's do some math: 2 * (oxidation number of H) + (-2) = 0. So, 2 * (oxidation number of H) = +2. Therefore, the oxidation number of hydrogen in water is +1. See? You're basically a mathlete now, but with way cooler molecules.
What about sulfuric acid, H₂SO₄? We know H is +1 and O is -2. The total charge is 0. So: 2(+1) + (oxidation number of S) + 4(-2) = 0. That simplifies to +2 + (oxidation number of S) - 8 = 0. So, (oxidation number of S) - 6 = 0. Boom! The oxidation number of sulfur is +6. It’s like a chemical detective solving a high-stakes case. You can practically hear the dramatic music!
A surprising fact about oxidation numbers? They can be fractional! Yes, you read that right. Sometimes, due to resonance or complex bonding structures, atoms can have oxidation numbers that aren't whole numbers. It’s like finding out your friend who always pays in exact change sometimes uses coupons and a gift card. Chemical chaos, but in a good way!
So, there you have it! The coordination number is all about who's holding hands with whom, and the oxidation number is about the hypothetical charge, or the atom's personality in the chemical limelight. They’re the dynamic duo that helps us understand how molecules interact and behave. Now go forth, armed with this newfound knowledge, and impress your friends, your pets, and maybe even a passing squirrel with your chemical prowess!
