How To Determine Molar Concentration Of Chicken Egg Cytoplasm
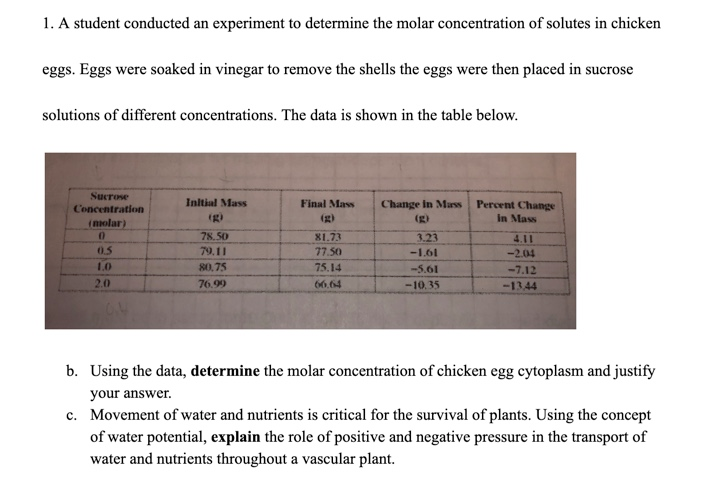
Ever stare at a perfectly cooked, sunny-side-up egg and wonder about the microscopic world buzzing within its golden yolk and pearly albumen? We're talking about the stuff that fuels life, folks! Today, we're diving into something a little more science-y than your average brunch prep, but trust me, it's way more fascinating than you might think. We're going to explore how to get a handle on the molar concentration of that chicken egg cytoplasm. Sounds intimidating, right? Like something out of a high-brow chemistry journal? Fear not, my friends, we're going to break it down into bite-sized, easy-going pieces, with a dash of culinary appreciation and maybe even a nod to your favorite sci-fi flick.
So, what exactly is molar concentration, and why should you care about it in relation to a humble chicken egg? Think of molar concentration, or molarity, as a way to measure how much "stuff" is dissolved in a given amount of liquid. It’s like knowing how many sugar cubes are in your iced coffee – a lot of cubes mean a sweet, potent brew; fewer cubes mean a lighter, less intense drink. In the egg's case, this "stuff" is made up of all sorts of essential molecules – proteins, salts, sugars, you name it – that keep that cell alive and kicking.
Why is this even a thing? Well, understanding molar concentration in biological systems, even as simple as an egg, helps scientists understand cellular processes. It’s the underlying chemistry that dictates how cells function, how nutrients are transported, and even how organisms develop. Think of it as the secret handshake of the cellular world. And who knows, maybe this knowledge could one day lead to the next big breakthrough in, say, artificial egg production or even advanced cellular therapies. Pretty neat, huh?
Deconstructing the Egg: It's More Than Just Breakfast!
Before we get our metaphorical lab coats on, let's appreciate the egg itself. It’s a complete package, a miniature marvel of nature. The yolk, rich and fatty, is the energy reserve, while the albumen, that clear, viscous fluid, is packed with proteins like albumin, which is what gives egg whites their structure when cooked (hello, meringue!). Both are essentially made up of cytoplasm – the jelly-like substance that fills the cell and surrounds the organelles. It's the bustling metropolis where all the cellular action happens.
When we talk about molar concentration in egg cytoplasm, we're essentially trying to quantify the total amount of dissolved particles within that cytoplasm. This isn't a single number; it's a complex blend of various ions, small molecules, and macromolecular components. But for a simplified overview, we can think about the major contributors to this "dissolved stuff."
The Usual Suspects: Ions and Small Molecules
Inside the egg cytoplasm, you’ve got a veritable cocktail of ions – charged particles that are super important for all sorts of cellular functions. Think of things like sodium (Na+), potassium (K+), chloride (Cl-), and phosphate (PO4^3-). These guys are the unsung heroes, involved in everything from nerve signaling (though less so in a chicken egg cell itself) to maintaining the pH balance. They’re like the background music at a party – you might not always notice them, but the whole atmosphere depends on them.
Then there are the small, non-ionic molecules. We're talking about things like glucose (sugar!), amino acids (the building blocks of proteins), and various metabolic intermediates. These are the fuel and the building materials of the cell. Imagine them as the snacks and the construction supplies on the cellular construction site. They’re essential for keeping things running and growing.

Measuring the precise molar concentration of each of these individually would be a Herculean task for a casual egg enthusiast. It requires sophisticated laboratory equipment and a whole lot of expertise. But for our easy-going exploration, we can think about the overall osmotic pressure, which is largely influenced by the total molar concentration of all these dissolved solutes.
The "Lab" at Home: Can We Really Do This?
Okay, let's be honest. Unless you have a well-equipped home science lab (and are perhaps a bit eccentric!), you're not going to be precisely determining the molar concentration of chicken egg cytoplasm with a beaker and a bunsen burner. The tools needed for accurate measurements are typically found in university research labs or specialized analytical facilities.
However, we can get a conceptual understanding and even perform some analogous experiments that illustrate the principles at play. Think of it like learning to cook by understanding the fundamental techniques, even if you don't have a Michelin-starred chef's arsenal. We can use readily available materials and some simple observations to get a feel for what's going on.
Osmosis: The Egg's Hidden Behavior
One of the most accessible ways to get a sense of molar concentration without fancy equipment is by observing osmosis. This is the movement of water across a semipermeable membrane from an area of lower solute concentration to an area of higher solute concentration. It’s like water trying to dilute the more concentrated side to achieve a balance – a natural equalizer.
You might have seen this phenomenon demonstrated with eggs in school science class. If you place a raw egg in vinegar for a day or two, the acid dissolves the calcium carbonate shell. What’s left is a semipermeable membrane surrounding the yolk and albumen. If you then place this de-shelled egg in pure water, water will rush into the egg, making it swell. This is because the cytoplasm inside the egg has a higher molar concentration of solutes than the pure water outside, so water moves in to try and equalize things.

Conversely, if you place that same de-shelled egg in a very salty or sugary solution, water will move out of the egg, causing it to shrink. The external solution is more concentrated than the cytoplasm, so water leaves the egg to dilute the outside solution. This is the principle behind food preservation techniques like salting fish or making jams – drawing water out of the food to inhibit microbial growth.
So, while you're not getting a precise molarity number, you're observing the consequences of that molarity. The degree to which the egg swells or shrinks gives you a qualitative idea of the relative concentration of the solutions you're using compared to the egg's internal environment.
The "Real" Science: What the Pros Do
If you were to actually measure the molar concentration of egg cytoplasm in a lab, you'd likely employ techniques that measure colligative properties. These are properties of a solution that depend solely on the ratio of the number of solute particles to the number of solvent molecules in the solution, and not on the specific nature of the chemical species present. Think of it like counting guests at a party – the number of people matters more than whether they're wearing a tuxedo or a t-shirt for some calculations.
Freezing Point Depression and Boiling Point Elevation
One common method is measuring freezing point depression. Pure water freezes at 0°C (32°F). However, when you add solutes to water, the freezing point lowers. The more solutes you have, the lower the freezing point. Scientists can carefully measure the freezing point of egg cytoplasm and compare it to the freezing point of pure water to estimate the total molar concentration of dissolved particles.
Similarly, adding solutes to water raises its boiling point (boiling point elevation). Pure water boils at 100°C (212°F). A solution will boil at a higher temperature. By measuring the boiling point of egg cytoplasm, scientists can infer its molar concentration. These methods are indirect but very effective.
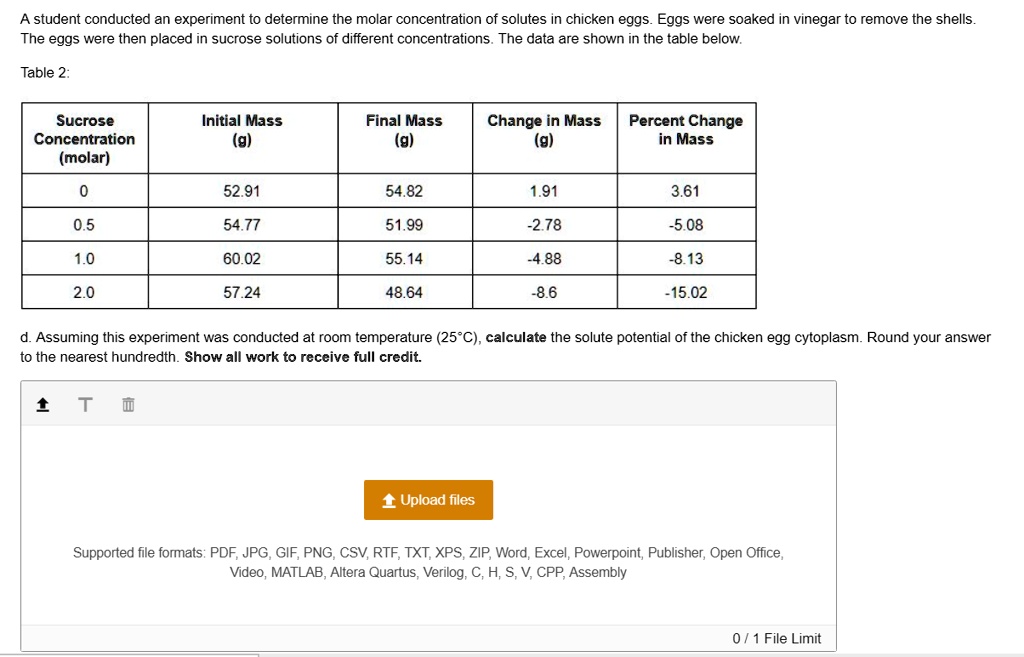
Osmometry
Another technique is using an osmometer. This is a device specifically designed to measure the osmotic pressure of a solution. By exposing the egg cytoplasm to a known solution and measuring the water movement across a semipermeable membrane, the osmometer can calculate the total molar concentration of the cytoplasm. It's a more direct approach, but still requires specialized equipment.
Spectrophotometry for Specific Solutes
If a researcher wanted to know the concentration of a specific solute, like glucose, they might use a spectrophotometer. This machine measures how much light a substance absorbs at a particular wavelength. By calibrating the spectrophotometer with known concentrations of glucose, they can then measure the absorbance of egg cytoplasm to determine the glucose concentration. This is a very precise method for individual components.
What's the Big Number? A Rough Estimate
So, what kind of numbers are we talking about? While it varies depending on the specific egg and the methods used, the total molar concentration of solutes in chicken egg cytoplasm is generally in the range of 0.3 to 0.4 M (moles per liter). This is roughly equivalent to the concentration of salts in seawater, which is why the egg membrane behaves the way it does in different solutions.
This value is crucial for maintaining the cell's internal environment, ensuring that water doesn't rush in or out too rapidly and that essential cellular processes can occur. It's the cell's way of saying, "This is my comfortable zone, and I need things to stay just so."
From Egg Cytoplasm to Your Daily Life
Now, you might be thinking, "This is all fascinating, but how does this relate to my Tuesday morning? I'm not exactly extracting cytoplasm for my cereal." And you're right! But the principles we've touched upon are everywhere.
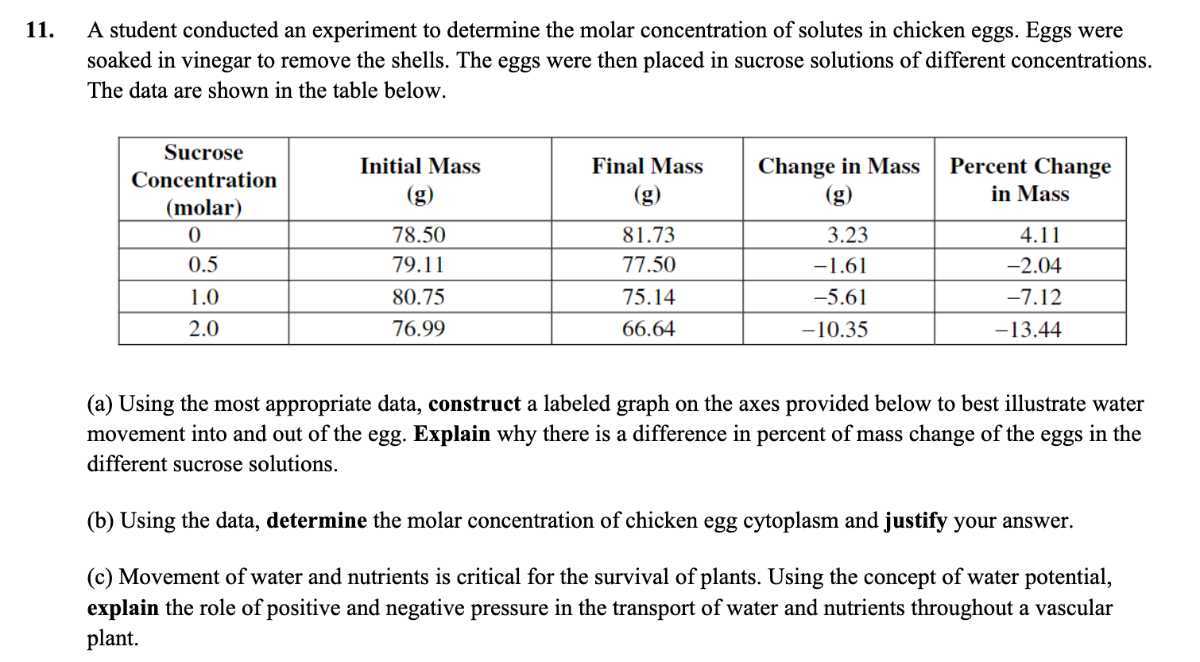
Think about drinking a sports drink after a workout. Why does it contain electrolytes like sodium and potassium? It’s to help replenish what you’ve lost and to maintain the right molar concentration in your body fluids, allowing for efficient rehydration and muscle function. It's the same idea as our egg – maintaining a balance.
Or consider cooking. When you brine chicken or marinate meat, you’re altering the molar concentration of the solution the food is in. The salt and other solutes draw water and flavor into the food, making it more tender and delicious. This is osmosis in action, driven by the differences in solute concentration, much like in our egg experiment.
Even the simple act of making tea or coffee involves diffusion and dissolution. The flavor molecules from the tea leaves or coffee grounds disperse into the hot water, increasing its molar concentration of those delicious compounds. The speed at which they disperse is influenced by temperature and the concentration gradient – another nod to fundamental chemical principles.
A Final Thought on Tiny Worlds
Looking at a chicken egg and pondering its molar concentration might seem like an exercise in academic curiosity. But it’s a beautiful reminder that even the most ordinary things are incredibly complex and governed by elegant scientific laws. From the microscopic world within an egg to the vast expanse of the universe, these fundamental principles of chemistry and physics are at play, shaping everything we see and experience.
So next time you crack an egg, take a moment to appreciate not just its culinary potential, but the miniature universe of molecules and membranes working in harmony within. It’s a tiny, fascinating world, and understanding even a little bit about its chemistry can add a whole new layer of wonder to your day. It’s proof that even the simplest things have stories to tell, if only we take the time to listen – or, in this case, to calculate!
