How To Calculate The Energy Of One Mole Of Photons

Ever looked up at the sun on a bright, sunny day and felt that warmth on your skin? Or maybe you've marveled at how a tiny LED light can illuminate a whole room? It's all thanks to something incredibly fundamental, yet often overlooked: photons. These little packets of light energy are the building blocks of pretty much everything we see. And while we usually talk about light as a wave, it also behaves like a particle, a photon. Pretty neat, huh?
Now, you might be thinking, "Okay, photons, cool story. But what does that have to do with me? And how on earth do I calculate the energy of one mole of them?" Stick with me, because it’s actually more relatable than you might imagine, and understanding it can unlock a little bit of wonder about the world around us.
Think of it like baking. You know how you can't just throw a handful of flour into a recipe and expect a perfect cake? You need precise measurements. Similarly, when scientists are dealing with light and its effects, they often need to talk about a standard "batch" of photons. That's where the concept of a mole comes in.
A mole, in chemistry, is just a way of counting. It's a huge number – specifically, Avogadro's number, which is approximately 6.022 x 1023. Imagine counting every grain of sand on every beach in the world, and then multiplying that number by a few million. That's roughly how many particles are in one mole! So, when we talk about a mole of photons, we're talking about a massive, almost incomprehensible, amount of these tiny light particles.
So, why should we care about the energy of a mole of photons? Well, it’s like knowing the total power of a whole orchestra playing versus just one violin. Understanding the collective energy of a mole of photons helps us grasp the power behind things like:
- Solar Panels: How much energy can a solar panel truly capture from the sun? It's all about the photons hitting it!
- Photosynthesis: How do plants use sunlight to grow? They're literally soaking up photon energy.
- Medical Lasers: The precise energy of photons is crucial for treatments, from eye surgery to cancer therapy.
- Everyday Lighting: Even that little bedside lamp is delivering a torrent of photon energy.
It helps us understand how light interacts with matter, and that’s the foundation of so much science and technology. It’s the secret sauce behind why certain things happen when light shines on them.
The Magic Formula: E = hf
Alright, let's get to the good stuff – how do we actually figure out this energy? It’s surprisingly elegant, thanks to a couple of brilliant minds. The core idea comes from Max Planck and Albert Einstein.
They figured out that the energy of a single photon is directly related to its frequency. Frequency, in simple terms, is how many wave crests of light pass a point in one second. Think of it like the beat of a drum – a fast beat means high frequency, a slow beat means low frequency.
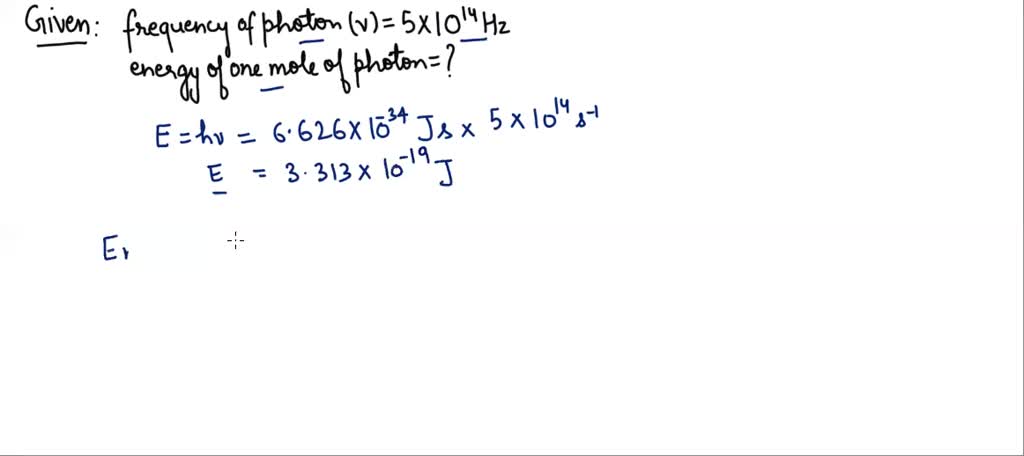
The formula for the energy of one photon is beautifully simple:
E = hf
Where:
- E is the energy of that single photon.
- h is Planck's constant. This is a fundamental number in physics, like a universal speed limit for information. It’s a tiny, tiny number: 6.626 x 10-34 joule-seconds (J·s). Think of it as the "cost" of a photon’s existence in terms of energy and time.
- f is the frequency of the light, measured in Hertz (Hz), which means cycles per second.
So, if you know the frequency of a light wave (say, the bright red light of a traffic signal), you can figure out the energy of one photon of that light. A higher frequency means a more energetic photon. Blue light, for example, has a higher frequency than red light, so a blue photon carries more punch!
From One Photon to a Mole of Photons
Now, we want the energy of a mole of photons. Remember that mole is just a very, very big number of things? So, to get the total energy of a mole of photons, we simply multiply the energy of one photon by the number of photons in a mole (Avogadro's number).
The formula looks like this:

Emole = NA * Ephoton
Which expands to:
Emole = NA * hf
Where:
- Emole is the energy of one mole of photons.
- NA is Avogadro's number (6.022 x 1023 mol-1).
- h is Planck's constant (6.626 x 10-34 J·s).
- f is the frequency of the light (Hz).
See how we've combined these key players? It's like putting together a recipe where Planck’s constant is your measuring spoon, Avogadro’s number is how many scoops you need, and the frequency tells you the intensity of the ingredient you're adding.

Let's Make it Tangible: A Little Story
Imagine you have a tiny, invisible firefly that flashes a specific color of light. Let's say its light has a frequency of 5 x 1014 Hz. That's a pretty common frequency for visible light.
First, we find the energy of one flash (one photon):
Ephoton = (6.626 x 10-34 J·s) * (5 x 1014 Hz)
Ephoton ≈ 3.313 x 10-19 Joules
That's an incredibly small amount of energy, right? It's hard to even imagine. That’s why we need the mole!
Now, let's imagine we have a whole swarm of these fireflies, so many that they represent one mole of flashes. To find the total energy of that swarm, we multiply the energy of one flash by Avogadro's number:

Emole = (6.022 x 1023) * (3.313 x 10-19 Joules)
Emole ≈ 199,545 Joules
Suddenly, we have a much larger, more understandable number! Around 200,000 Joules. That's roughly the energy needed to boil about half a liter of water. So, even though each individual photon is minuscule, a mole of them can add up to a significant amount of energy. It's like tiny raindrops – one drop doesn't do much, but a deluge can fill a river!
Why This Matters (Besides Being Cool!)
Understanding this calculation helps us appreciate the power of light in everyday ways. When you're basking in the sun, you're not just feeling warmth; you're experiencing the collective energy of countless photons. When you use a microwave, you’re using electromagnetic radiation (which is just photons of a specific frequency) to heat your food.
It also empowers us to understand scientific advancements. The development of lasers, which rely on precisely controlled photons, has revolutionized surgery, data transmission, and even entertainment. The efficiency of solar cells is constantly being improved by understanding how to capture more of those energetic photons.
So, the next time you see light, whether it's the gentle glow of a lamp or the dazzling brilliance of fireworks, take a moment to remember the photons. And if you’re feeling adventurous, grab a calculator and see if you can figure out the energy of a mole of photons for different colors of light. You might just discover a new appreciation for the energetic dance of the universe happening all around you, every single second!
