How Much Iron Can Be Recovered From 25.0g Of Fe2o3
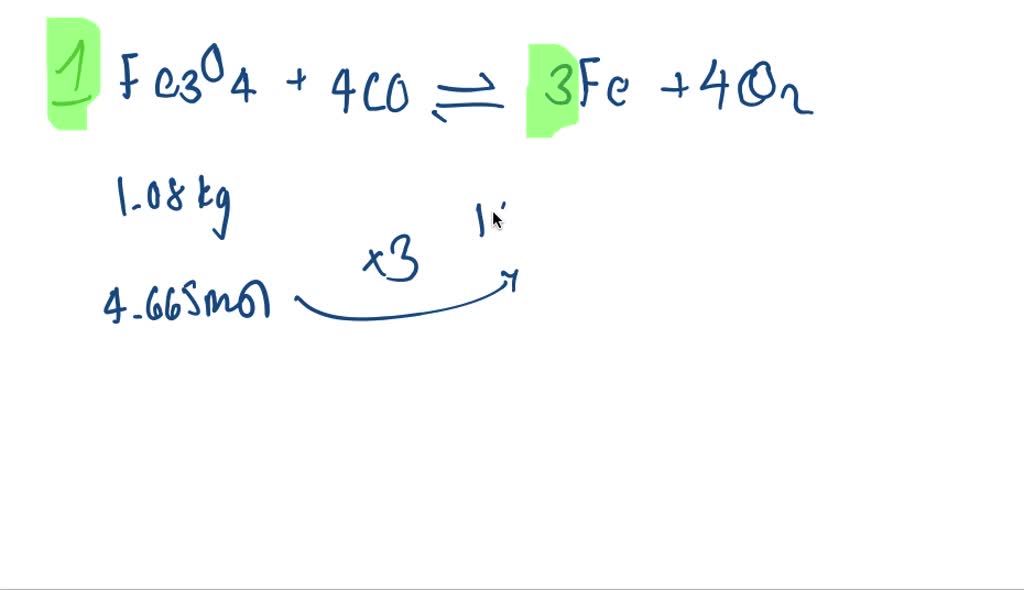
Hey there, science explorers! Ever looked at something and wondered, "What's really inside there?" Well, buckle up, because today we're diving into the fascinating world of iron, and trust me, it's more exciting than you might think! We're going to talk about rust – yes, that reddish-brown stuff that gets on your bike chain – but in a way that's actually pretty cool.
You see, that common old rust, chemically speaking, is mostly iron(III) oxide. Fancy name, right? It’s like saying "super-duper iron protector" instead of just "rust." And the amazing thing is, within this seemingly simple compound, there’s a whole lot of precious iron just waiting to be unleashed! Think of it like a hidden treasure chest, and we're about to find out how much treasure we can dig out.
So, let's get down to brass tacks – or should I say, iron tacks? We've got ourselves a neat little sample, precisely 25.0 grams of this iron(III) oxide (Fe2O3). Now, the big question is: how much of that is actual, usable iron? It’s like asking, if you have a bag of delicious cookies, how many cookies are really in there, not counting the chocolate chips or the sprinkles? Well, not exactly like that, but you get the drift!
Unlocking the Iron Within!
To figure this out, we need a little bit of chemistry magic. Don't worry, it's not complicated, and it’s kind of like following a recipe. First, we need to know the "ingredients" and their "weights." In chemistry, we call these atomic masses. These are like the individual weights of each atom that makes up our iron(III) oxide molecule.
We've got iron (Fe) and oxygen (O). Now, the formula Fe2O3 tells us we have two atoms of iron for every three atoms of oxygen. It's like a secret handshake between iron and oxygen! And these atoms have their own special weights:
- Iron (Fe) weighs about 55.845 grams per mole.
- Oxygen (O) weighs about 15.999 grams per mole.
A "mole" is just a chemist's way of counting a huge number of things – like a super-sized dozen! So, these are the weights for a whole bunch of atoms.

Calculating the Molecular Weight
Now, let's add up the weights for our entire Fe2O3 molecule. Remember, we have two irons and three oxygens:
- Weight of iron part: 2 * 55.845 g/mol = 111.69 g/mol
- Weight of oxygen part: 3 * 15.999 g/mol = 47.997 g/mol
So, the total weight of one mole of iron(III) oxide is: 111.69 g/mol + 47.997 g/mol = 159.687 g/mol. Let's round that to a nice, neat 159.7 grams per mole for easier calculations.
This means that out of every 159.7 grams of iron(III) oxide, 111.7 grams are pure, shiny iron! Isn't that neat? It's like finding out that a whole bunch of your cookie is actually made of yummy dough!
The Percentage Game!
To find out the percentage of iron in iron(III) oxide, we just do a simple division. It’s like figuring out what percentage of your pizza is pepperoni!

Percentage of Iron = (Weight of Iron in molecule / Total Weight of molecule) * 100%
Percentage of Iron = (111.7 g/mol / 159.7 g/mol) * 100%
And voilà! That gives us approximately 69.94%. So, almost 70% of iron(III) oxide is actually iron! That’s a pretty significant chunk, wouldn't you agree? It means that the rusty stuff isn't just a nuisance; it's a potential source of something incredibly useful!
Now, we started with 25.0 grams of Fe2O3. To find out how much iron we can recover, we just multiply our starting amount by the percentage of iron it contains.

Amount of Recoverable Iron = 25.0 g Fe2O3 * (69.94 / 100)
And the grand total? Drumroll, please… we can recover approximately 17.5 grams of iron! Wow! From just 25 grams of rust, we can get a good chunk of real iron. Isn’t that just amazing? It’s like finding a whole bunch of gold coins in a seemingly ordinary box!
Why Is This So Fun?
You might be thinking, "Okay, cool numbers, but how does this make life more fun?" Well, think about it! This isn't just about abstract chemistry. This is about understanding the building blocks of our world!
Every time you see a bridge, a car, or even your trusty frying pan, you're seeing the result of iron extraction and processing. The iron we use in our everyday lives often starts its journey in compounds like iron(III) oxide. By understanding how to extract it, we're understanding how we build and create!
It’s like being a detective, but instead of solving crimes, you're uncovering the secrets of matter. You're looking at something, calculating its potential, and seeing the hidden value within. This kind of thinking can be applied to so many things. See a pile of old electronics? There's probably valuable metal in there!
This knowledge empowers you. It makes you see the world with a little more curiosity and a lot more appreciation for the science that shapes our lives. You can look at a rusty nail and instead of just seeing decay, you can see a source of useful material, a testament to chemical transformations.
The Spark of Inspiration
So, the next time you encounter iron(III) oxide, or any chemical compound for that matter, remember this little adventure. Remember that even in the most ordinary-looking substances, there are often incredible possibilities waiting to be discovered. This is just the tip of the iceberg, my friends!
This is the magic of chemistry – it allows us to peek behind the curtain of the universe and understand how things work. It inspires us to ask "what if?" and to seek answers. So go forth, be curious, and never underestimate the fascinating science hidden all around you. Who knows what other amazing discoveries you’ll make!
